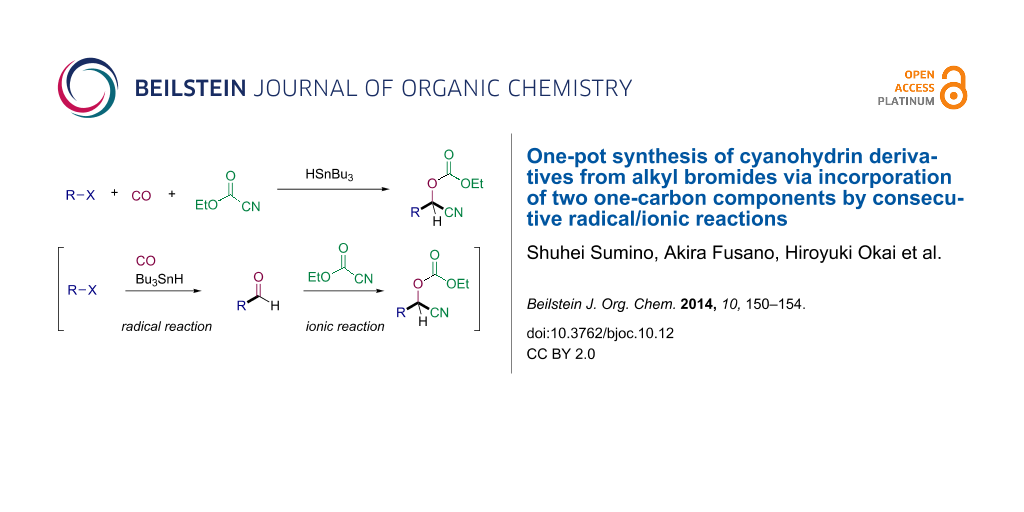
Abstract
The consecutive radical/ionic reaction consisting of radical formylation of alkyl bromides and nucleophilic addition of a cyanide ion was investigated, which gave moderate to good yields of cyanohydrin derivatives in one-pot.
Graphical Abstract
Introduction
Radical carbonylation reactions have been recognized as a versatile tool for the synthesis of a wide variety of carbonyl compounds [1-4]. In 1990, we demonstrated that aldehydes can be prepared from alkyl or aromatic halides and CO under typical radical chain reaction conditions using tributyltin hydride and AIBN [5,6]. Under the reaction conditions where a catalytic amount of fluorous tin hydride and an excess amount of sodium cyanoborohydride were used, initially formed aldehydes can be converted into hydroxymethylated compounds in one-pot [7-9], since borohydride acts not only as the reagent for the regeneration of tin hydride [10-13] but also as the reagent for aldehyde reduction. Later on we found that borohydride reagents can also serve as radical mediator delivering hydrogen to the radical centre [14], thus we developed a hydroxymethylation method using Bu4NBH4 and a radical initiator [15-17]. Recent work in collaboration with Dennis Curran has revealed that, with the use of NHC-borane [18], hydroxymethylation of aromatic iodides can be attained [19]. All these reactions consist of the combination of radical formylation with CO and ionic hydride reduction by hydride reagents (Scheme 1, reaction 1). During the course of our study on borohydride-mediated radical hydroxymethylation of alkyl halides with CO, we found that cyanohydrin was obtained as a byproduct when Bu4NBH3CN was used as a radical mediator [15], which led us to investigate the one-pot synthesis of cyanohydrins based on radical formylation. Thus, we thought that the two step radical/ionic reactions can be extended to the consecutive C–C bond forming reactions.
Scheme 1: Sequential radical formylation and derivatization.
Scheme 1: Sequential radical formylation and derivatization.
Cyanohydrins are important subunits frequently found in biologically active compounds and are also versatile building blocks for further synthetic transformations [20,21]. The common method to obtain cyanohydrins is the reaction of aldehydes with a cyanide sources such as TMSCN [22,23], ethyl cyanoformate [24-26] or acyl cyanide [27,28]. We provide here an efficient one-pot method for the synthesis of cyanohydrin derivatives via consecutive radical/ionic C–C bond forming reaction of alkyl bromides, CO and ethyl cyanoformate (Scheme 1, reaction 2).
Results and Discussion
We examined AIBN-induced radical formylation of 1-bromooctane (1a) with Bu3SnH under 80 atm of CO pressure in the presence of a cyanide source (Scheme 2). Under the employed conditions, the reaction using TMSCN (2a') was slow, which gave 16% of 3a' and 51% of nonanal. The use of AcCN (2a'') also gave 3a'' but only in 12% yield. However, when ethyl cyanoformate (2a) was used together with Et3N [29], the cyanohydrin 3a was obtained in 62% yield. When we used higher CO pressure such as 120 atm, the yield of 3a increased to 79%.
We examined various alkyl bromides 1 in the present radical/ionic three-component coupling reaction (Table 1). Primary alkyl bromides 1b–e containing a chlorine atom, an ester group, a cyano group, or a phenyl group worked well to give the corresponding cyanohydrin derivatives 3b–e in good yields (Table 1, entries 2–5). The reaction of secondary and tertiary alkyl bromides 1f–i also proceeded well to give the corresponding cyanohydrins 3f–i in good yields (Table 1, entries 6–9). The reaction using cyclopropylmethyl bromide (1j) afforded the lowest yield of cyanohydrin 3j, which possessed an olefin structure arising from the ring-opening of a cyclopropylcarbinyl radical (Table 1, entry 10) [30,31].
Table 1: Three-component coupling reaction leading to cyanohydrin derivatives.
|
|
||||
| entry | alkyl bromide | CO (atm) | product | yielda (%) |
|---|---|---|---|---|
| 1b |
1a |
120 |
3a |
79 |
| 2 |
1b |
80 |
3b |
60 |
| 3 |
1c |
80 |
3c |
83 |
| 4 |
1d |
120 |
3d |
76 |
| 5 |
1e |
120 |
3e |
61 |
| 6 |
1f |
120 |
3f |
61 |
| 7 |
1g |
120 |
3g |
74 |
| 8 |
1h |
120 |
3h |
73 |
| 9 |
1i |
110 |
3i |
82 |
| 10 |
1j |
110 |
3j |
45 |
aIsolated yield after flash chromatography on SiO2. b0.03 M.
Conclusion
In summary, we have demonstrated a three-component coupling reaction comprising alkyl bromides 1, CO and ethyl cyanoformate (2a) in the presence of Bu3SnH, AIBN, and Et3N, which gave moderate to good yields of cyanohydrin derivatives 3. This protocol represents a one-pot method [32,33] based on radical carbonylation and ionic cyanation.
Experimental
Typical procedure for radical/ionic three-component coupling reaction leading to cyanohydrin derivatives 1-cyanononyl ethyl carbonate (3a) [34] (Table 1, entry 1): A mixture of 1-bromooctane (1a, 96.6 mg, 0.5 mmol), ethyl cyanoformate (2a, 79.3 mg, 0.8 mmol), tributyltin hydride (174.6 mg, 0.6 mmol), triethylamine (13.2 mg, 0.13 mmol), and AIBN (24.6 mg, 0.15 mmol) in C6H6 (17 mL) were placed in a 100 mL stainless steel autoclave. The reaction mixture was degassed 3 times with 10 atm of CO and charged with 90 atm of CO at −40 °C (MeCN–dry ice bath). Then the autoclave was allowed to warm to room temperature, which caused the pressure gauge to indicate 120 atm. Then the reaction was conducted at 80 °C for 3 h. After cooling to room temperature, the reaction mixture was concentrated and purified by silica gel flash chromatography (hexane/EtOAc 97:3) to afford 3a (95.3 mg, 79%). 1H NMR (CDCl3, 500 MHz) δ 5.18 (t, J = 6.8 Hz, 1H), 4.4–4.2 (m, 2H), 2.0–1.9 (m, 2H), 1.6–1.5 (m, 2H), 1.4–1.2 (m, 13H), 0.88 (t, J = 6.9 Hz, 3H); 13C NMR (CDCl3, 125 MHz) δ 153.56, 116.51, 65.27, 64.66, 32.31, 31.68, 29.12, 28.99, 28.71, 24.34, 22.53, 14.05, 13.93.
References
-
Ryu, I.; Sonoda, N. Angew. Chem., Int. Ed. Engl. 1996, 35, 1050. doi:10.1002/anie.199610501
Return to citation in text: [1] -
Ryu, I.; Sonoda, N.; Curran, D. P. Chem. Rev. 1996, 96, 177. doi:10.1021/cr9400626
Return to citation in text: [1] -
Chatgilialoglu, C.; Crich, D.; Komatsu, M.; Ryu, I. Chem. Rev. 1999, 99, 1991. doi:10.1021/cr9601425
Return to citation in text: [1] -
Ryu, I. Chem. Soc. Rev. 2001, 30, 16. doi:10.1039/a904591k
Return to citation in text: [1] -
Ryu, I.; Kusano, K.; Ogawa, A.; Kambe, N.; Sonoda, N. J. Am. Chem. Soc. 1990, 112, 1295. doi:10.1021/ja00159a088
Return to citation in text: [1] -
Ryu, I.; Kusano, K.; Masumi, N.; Yamazaki, H.; Ogawa, A.; Sonoda, N. Tetrahedron Lett. 1990, 31, 6887. doi:10.1016/S0040-4039(00)97198-3
Return to citation in text: [1] -
Gupta, V.; Kahne, D. Tetrahedron Lett. 1993, 34, 591. doi:10.1016/S0040-4039(00)61627-1
Return to citation in text: [1] -
Ryu, I.; Niguma, T.; Minakata, S.; Komatsu, M.; Hadida, S.; Curran, D. P. Tetrahedron Lett. 1997, 38, 7883. doi:10.1016/S0040-4039(97)10076-4
Return to citation in text: [1] -
Matsubara, H.; Yasuda, S.; Sugiyama, H.; Ryu, I.; Fujii, Y.; Kita, K. Tetrahedron 2002, 58, 4071. doi:10.1016/S0040-4020(02)00256-9
Return to citation in text: [1] -
Corey, E. J.; Suggs, W. J. Org. Chem. 1975, 40, 2554. doi:10.1021/jo00905a039
Return to citation in text: [1] -
Giese, B.; González-Gómez, J. A.; Witzel, T. Angew. Chem., Int. Ed. Engl. 1984, 23, 69. doi:10.1002/anie.198400691
Return to citation in text: [1] -
Stork, G.; Sher, P. M. J. Am. Chem. Soc. 1986, 108, 303. doi:10.1021/ja00262a024
Return to citation in text: [1] -
Curran, D. P.; Hadida, S.; Kim, S.-Y.; Luo, Z. J. Am. Chem. Soc. 1999, 121, 6607. doi:10.1021/ja990069a
Return to citation in text: [1] -
Ryu, I.; Uehara, S.; Hirao, H.; Fukuyama, T. Org. Lett. 2008, 10, 1005. doi:10.1021/ol7031043
Return to citation in text: [1] -
Kobayashi, S.; Kawamoto, T.; Uehara, S.; Fukuyama, T.; Ryu, I. Org. Lett. 2010, 12, 1548. doi:10.1021/ol1002847
Return to citation in text: [1] [2] -
Kobayashi, S.; Kinoshita, T.; Kawamoto, T.; Wada, M.; Kuroda, H.; Masuyama, A.; Ryu, I. J. Org. Chem. 2011, 76, 7096. doi:10.1021/jo201064h
Return to citation in text: [1] -
Kawamoto, T.; Ryu, I. Chimia 2012, 66, 372.
Return to citation in text: [1] -
Curran, D. P.; Solovyev, A.; Makhlouf Brahmi, M.; Fensterbank, L.; Malacria, M.; Lacôte, E. Angew. Chem., Int. Ed. 2011, 50, 10294. doi:10.1002/anie.201102717
See for a review on NHC-borane.
Return to citation in text: [1] -
Kawamoto, T.; Okada, T.; Curran, D. P.; Ryu, I. Org. Lett. 2013, 15, 2144. doi:10.1021/ol4006294
Return to citation in text: [1] -
Gregory, R. J. H. Chem. Rev. 1999, 99, 3649. doi:10.1021/cr9902906
Return to citation in text: [1] -
Brunel, J.-M.; Holmes, I. P. Angew. Chem., Int. Ed. 2004, 43, 2752. doi:10.1002/anie.200300604
Return to citation in text: [1] -
Lidy, W.; Sundermeyer, W. Chem. Ber. 1973, 106, 587. doi:10.1002/cber.19731060224
Return to citation in text: [1] -
Evans, D. A.; Truesdale, L. K.; Carroll, G. L. J. Chem. Soc., Chem. Commun. 1973, 55. doi:10.1039/c39730000055
Return to citation in text: [1] -
Poirier, D.; Berthiaume, D.; Boivin, R. P. Synlett 1999, 1423. doi:10.1055/s-1999-2859
Return to citation in text: [1] -
Berthiaume, D.; Poirier, D. Tetrahedron 2000, 56, 5995. doi:10.1016/S0040-4020(00)00535-4
Return to citation in text: [1] -
Tian, S.-K.; Deng, L. J. Am. Chem. Soc. 2001, 123, 6195. doi:10.1021/ja010690m
Return to citation in text: [1] -
Hoffmann, H. M. R.; Ismail, Z. M.; Hollweg, R.; Zein, A. R. Bull. Chem. Soc. Jpn. 1990, 63, 1807. doi:10.1246/bcsj.63.1807
Return to citation in text: [1] -
Okimoto, M.; Chiba, T. Synthesis 1996, 1188. doi:10.1055/s-1996-4361
Return to citation in text: [1] -
Baeza, A.; Nájera, C.; de Garcia Retamosa, M.; Sansano, J. M. Synthesis 2005, 2787. doi:10.1055/s-2005-872096
Return to citation in text: [1] -
Bowry, V. W.; Ingold, K. U. J. Am. Chem. Soc. 1991, 113, 5699. doi:10.1021/ja00015a025
Return to citation in text: [1] -
Newcomb, M. Tetrahedron 1993, 49, 1151. doi:10.1016/S0040-4020(01)85808-7
Return to citation in text: [1] -
Suga, S.; Yamada, D.; Yoshida, J.-i. Chem. Lett. 2010, 39, 404. doi:10.1246/cl.2010.404
Return to citation in text: [1] -
Yoshida, J.-i.; Saito, K.; Nokami, T.; Nagaki, A. Synlett 2011, 1189. doi:10.1055/s-0030-1259946
Return to citation in text: [1] -
Khan, N. H.; Agrawal, S.; Kureshy, R. I.; Abdi, S. H. R.; Sadhukhan, A.; Pillai, R. S.; Bajaj, H. C. Catal. Commun. 2010, 11, 907. doi:10.1016/j.catcom.2010.04.005
Return to citation in text: [1]
| 34. | Khan, N. H.; Agrawal, S.; Kureshy, R. I.; Abdi, S. H. R.; Sadhukhan, A.; Pillai, R. S.; Bajaj, H. C. Catal. Commun. 2010, 11, 907. doi:10.1016/j.catcom.2010.04.005 |
| 1. | Ryu, I.; Sonoda, N. Angew. Chem., Int. Ed. Engl. 1996, 35, 1050. doi:10.1002/anie.199610501 |
| 2. | Ryu, I.; Sonoda, N.; Curran, D. P. Chem. Rev. 1996, 96, 177. doi:10.1021/cr9400626 |
| 3. | Chatgilialoglu, C.; Crich, D.; Komatsu, M.; Ryu, I. Chem. Rev. 1999, 99, 1991. doi:10.1021/cr9601425 |
| 4. | Ryu, I. Chem. Soc. Rev. 2001, 30, 16. doi:10.1039/a904591k |
| 14. | Ryu, I.; Uehara, S.; Hirao, H.; Fukuyama, T. Org. Lett. 2008, 10, 1005. doi:10.1021/ol7031043 |
| 30. | Bowry, V. W.; Ingold, K. U. J. Am. Chem. Soc. 1991, 113, 5699. doi:10.1021/ja00015a025 |
| 31. | Newcomb, M. Tetrahedron 1993, 49, 1151. doi:10.1016/S0040-4020(01)85808-7 |
| 10. | Corey, E. J.; Suggs, W. J. Org. Chem. 1975, 40, 2554. doi:10.1021/jo00905a039 |
| 11. | Giese, B.; González-Gómez, J. A.; Witzel, T. Angew. Chem., Int. Ed. Engl. 1984, 23, 69. doi:10.1002/anie.198400691 |
| 12. | Stork, G.; Sher, P. M. J. Am. Chem. Soc. 1986, 108, 303. doi:10.1021/ja00262a024 |
| 13. | Curran, D. P.; Hadida, S.; Kim, S.-Y.; Luo, Z. J. Am. Chem. Soc. 1999, 121, 6607. doi:10.1021/ja990069a |
| 32. | Suga, S.; Yamada, D.; Yoshida, J.-i. Chem. Lett. 2010, 39, 404. doi:10.1246/cl.2010.404 |
| 33. | Yoshida, J.-i.; Saito, K.; Nokami, T.; Nagaki, A. Synlett 2011, 1189. doi:10.1055/s-0030-1259946 |
| 7. | Gupta, V.; Kahne, D. Tetrahedron Lett. 1993, 34, 591. doi:10.1016/S0040-4039(00)61627-1 |
| 8. | Ryu, I.; Niguma, T.; Minakata, S.; Komatsu, M.; Hadida, S.; Curran, D. P. Tetrahedron Lett. 1997, 38, 7883. doi:10.1016/S0040-4039(97)10076-4 |
| 9. | Matsubara, H.; Yasuda, S.; Sugiyama, H.; Ryu, I.; Fujii, Y.; Kita, K. Tetrahedron 2002, 58, 4071. doi:10.1016/S0040-4020(02)00256-9 |
| 27. | Hoffmann, H. M. R.; Ismail, Z. M.; Hollweg, R.; Zein, A. R. Bull. Chem. Soc. Jpn. 1990, 63, 1807. doi:10.1246/bcsj.63.1807 |
| 28. | Okimoto, M.; Chiba, T. Synthesis 1996, 1188. doi:10.1055/s-1996-4361 |
| 5. | Ryu, I.; Kusano, K.; Ogawa, A.; Kambe, N.; Sonoda, N. J. Am. Chem. Soc. 1990, 112, 1295. doi:10.1021/ja00159a088 |
| 6. | Ryu, I.; Kusano, K.; Masumi, N.; Yamazaki, H.; Ogawa, A.; Sonoda, N. Tetrahedron Lett. 1990, 31, 6887. doi:10.1016/S0040-4039(00)97198-3 |
| 29. | Baeza, A.; Nájera, C.; de Garcia Retamosa, M.; Sansano, J. M. Synthesis 2005, 2787. doi:10.1055/s-2005-872096 |
| 15. | Kobayashi, S.; Kawamoto, T.; Uehara, S.; Fukuyama, T.; Ryu, I. Org. Lett. 2010, 12, 1548. doi:10.1021/ol1002847 |
| 22. | Lidy, W.; Sundermeyer, W. Chem. Ber. 1973, 106, 587. doi:10.1002/cber.19731060224 |
| 23. | Evans, D. A.; Truesdale, L. K.; Carroll, G. L. J. Chem. Soc., Chem. Commun. 1973, 55. doi:10.1039/c39730000055 |
| 19. | Kawamoto, T.; Okada, T.; Curran, D. P.; Ryu, I. Org. Lett. 2013, 15, 2144. doi:10.1021/ol4006294 |
| 24. | Poirier, D.; Berthiaume, D.; Boivin, R. P. Synlett 1999, 1423. doi:10.1055/s-1999-2859 |
| 25. | Berthiaume, D.; Poirier, D. Tetrahedron 2000, 56, 5995. doi:10.1016/S0040-4020(00)00535-4 |
| 26. | Tian, S.-K.; Deng, L. J. Am. Chem. Soc. 2001, 123, 6195. doi:10.1021/ja010690m |
| 18. |
Curran, D. P.; Solovyev, A.; Makhlouf Brahmi, M.; Fensterbank, L.; Malacria, M.; Lacôte, E. Angew. Chem., Int. Ed. 2011, 50, 10294. doi:10.1002/anie.201102717
See for a review on NHC-borane. |
| 15. | Kobayashi, S.; Kawamoto, T.; Uehara, S.; Fukuyama, T.; Ryu, I. Org. Lett. 2010, 12, 1548. doi:10.1021/ol1002847 |
| 16. | Kobayashi, S.; Kinoshita, T.; Kawamoto, T.; Wada, M.; Kuroda, H.; Masuyama, A.; Ryu, I. J. Org. Chem. 2011, 76, 7096. doi:10.1021/jo201064h |
| 17. | Kawamoto, T.; Ryu, I. Chimia 2012, 66, 372. |
| 20. | Gregory, R. J. H. Chem. Rev. 1999, 99, 3649. doi:10.1021/cr9902906 |
| 21. | Brunel, J.-M.; Holmes, I. P. Angew. Chem., Int. Ed. 2004, 43, 2752. doi:10.1002/anie.200300604 |
© 2014 Sumino et al; licensee Beilstein-Institut.
This is an Open Access article under the terms of the Creative Commons Attribution License (http://creativecommons.org/licenses/by/2.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
The license is subject to the Beilstein Journal of Organic Chemistry terms and conditions: (http://www.beilstein-journals.org/bjoc)