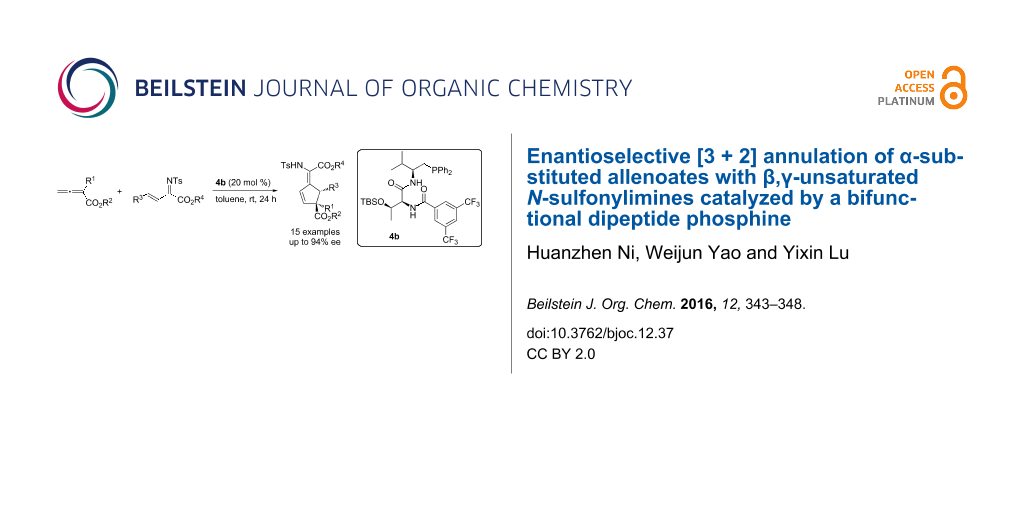
Abstract
The first enantioselective [3 + 2] annulation of α-substituted allenoates with β,γ-unsaturated N-sulfonylimines is described. In the presence of a dipeptide phosphine catalyst, a wide range of highly functionalized cyclopentenes bearing an all-carbon quaternary center were obtained in moderate to good yields and with good to excellent enantioselectivities.
Graphical Abstract
Introduction
Over the past decade, chiral phosphine catalysts have been utilized extensively for the construction of a broad range of synthetically useful molecular structures [1-13]. Since the initial discovery of phosphine-catalyzed [3 + 2] annulation of allenoates and activated alkenes by Lu in 1995, this type of annulation reaction has received considerable attention due to its high efficiency and versatility in creating five-membered ring systems [14-33]. However, most of the earlier examples make use of allenoates without an α-substitution. As demonstrated by Yu, Kwon and their co-workers [34-36], this is due to the requirement of a hydrogen atom at the α-position for a proton shift during the reaction cycle. Instead, α-substituted allenoates were shown to interact with phosphine in different reaction modes and undergo [4 + 2] annulations with suitable reaction partners to afford six-membered ring structures [37-47]. Recently, He and co-workers disclosed that the reaction between α-substituted allenoates and β,γ-unsaturated N-sulfonylimines proceeded in an unexpected [3 + 2] annulation mode to afford a cyclopentene ring with an all-carbon quaternary center (Scheme 1) [48]. In recent years our group has developed a family of amino acid-derived bifunctional phosphines and has intensively investigated related asymmetric transformations [49-63]. We became interested in developing an asymmetric variant of the above transformation by utilizing our amino acid-derived bifunctional phosphine catalysts.
Scheme 1: The [3 + 2] annulation of α-substituted allenoates reported by He.
Scheme 1: The [3 + 2] annulation of α-substituted allenoates reported by He.
Results and Discussion
We chose the [3 + 2] annulation between α-benzyl-substituted allenoate 1a and β,γ-unsaturated N-sulfonylimine 2a as a model reaction and evaluated a number of amino acid based bifunctional phosphines as catalyst. As shown in Table 1, simple L-valine-derived phosphines 3a–c were found to be effective in promoting the reaction, and products were obtained in moderate to good yields and with good E/Z ratios, and amide–phosphine 3b worked best (Table 1, entries 2–4). L-Alanine-based phosphine 3d and L-threonine-derived catalysts 3e and 3f did not provide better results (Table 1, entries 5–7). By employing L-threonine-derived catalyst 3g, the enantioselectivity of the reaction was improved to 68%. To further improve the reaction results, we next utilized dipeptide phosphine catalysts, which are more structurally diverse and tunable. The L-thr-L-thr-derived catalyst 4a was a poor catalyst, on the other hand, L-val-L-thr-derived catalyst 4b led to adequately improved enantioselectivity of the reaction and was chosen for further investigations.
Table 1: Screening of different amino acid-based bifunctional phosphine catalysts.
|
|
||||
| Entry | Catalyst | E/Z ratioa | Yield (%)b | ee (%)c |
|---|---|---|---|---|
| 1 | MePPh2 | 85:15 | 67 | – |
| 2 | 3a | 83:17 | 60 | 10 |
| 3 | 3b | 88:12 | 70 | 48 |
| 4 | 3c | 80:20 | 65 | 32 |
| 5 | 3d | 89:11 | 72 | 35 |
| 6 | 3e | 87:13 | 64 | 36 |
| 7 | 3f | 85:15 | 73 | 47 |
| 8 | 3g | 88:12 | 74 | 68 |
| 9 | 4a | 86:14 | 71 | 60 |
| 10 | 4b | 89:11 | 72 | 76 |
aDetermined by 1H NMR analysis of the crude reaction mixture. bIsolated yield of the E-isomers. cDetermined by HPLC analysis on a chiral stationary phase.
With the optimized conditions established, the substrate scope of this [3 + 2] annulation was explored by varying α-substituted allenoates 1 and imines 2 (Table 2). Firstly, different ester groups at the allenoates were examined (Table 2, entries 1–3). An allenoate bearing a tert-butyl ester group (1b) was found to be the best substrate, and the annulation products were obtained in good E/Z ratio, high yield and an ee of 84% (Table 2, entry 2). Allenoate substrates having different substitutions at the α-position were well tolerated, and the employment of various α-benzyl allenoates led to the formation of the products in consistently high E/Z ratios and enantioselectivities (Table 2, entries 4–6). It seemed that the presence of the ortho substituent in allenoates led to better enantioselectivity and decreased chemical yield (Table 2, entry 6). The utilization of 1-naphthyl substituted allenoate 1g resulted in poor yield but excellent enantioselectivity (Table 2, entry 7). Notably, the electronic properties of the benzyl groups in allenoates did not have much effect on the reaction outcome (Table 2, entries 8 and 9). Furthermore, methoxycarbonylmethyl-substituted allenoate 1j also proved to be a suitable substrate (Table 2, entry 10). The scope of β,γ-unsaturated N-sulfonylimines was subsequently examined by employing a number of differently substituted imines (Table 2, entries 11–15). In general, all the reactions worked well and afforded the annulation products in good E/Z ratios, moderate to good yields, and high enantioselectivities. Notably, imine 2e bearing an electron rich aryl substituent was found to be a superior substrate; higher yield and ee value were attainable (Table 2, entry 14).
Table 2: Enantioselective [3 + 2] annulation of α-substituted allenoates with β,γ-unsaturated N-sulfonylimines catalyzed by dipeptide catalyst 4b.a
|
|
||||||
| Entry | 1 (R1/R2) | 2 (R3/R4) | 5 | E/Zb | Yield (%)c | ee (%)d |
|---|---|---|---|---|---|---|
| 1 | 1a (Ph/Me) | 2a (Ph/Me) | 5a | 89:11 | 76 | 76 |
| 2 | 1b (Ph/t-Bu) | 2a (Ph/Me) | 5b | 83:17 | 70 | 84 |
| 3 | 1c (Ph/Bn) | 2a (Ph/Me) | 5c | 85:15 | 72 | 78 |
| 4 | 1d (4-ClPh/t-Bu) | 2a (Ph/Me) | 5d | 80:20 | 69 | 86 |
| 5 | 1e (3-ClPh/t-Bu) | 2a (Ph/Me) | 5e | 81:19 | 60 | 89 |
| 6 | 1f (2-ClPh/t-Bu) | 2a (Ph/Me) | 5f | 78:22 | 45 | 94 |
| 7 | 1g (1-naphthyl/t-Bu) | 2a (Ph/Me) | 5g | 80:20 | 43 | 93 |
| 8 | 1h (4-MePh/t-Bu) | 2a (Ph/Me) | 5h | 83:17 | 65 | 86 |
| 9 | 1i (4-NO2Ph/t-Bu) | 2a (Ph/Me) | 5i | 81:19 | 73 | 92 |
| 10 | 1j (CO2Me/Bn) | 2a (Ph/Me) | 5j | 90:10 | 72 | 82 |
| 11 | 1b (Ph/t-Bu) | 2b (Ph/Et) | 5k | 80:20 | 68 | 85 |
| 12 | 1b (Ph/t-Bu) | 2c (4-FPh/Et) | 5l | 83:17 | 55 | 86 |
| 13 | 1b (Ph/t-Bu) | 2d (4-ClPh/Et) | 5m | 78:22 | 58 | 82 |
| 14 | 1b (Ph/t-Bu) | 2e (4-MeOPh/Et) | 5n | 88:12 | 70 | 90 |
| 15 | 1b (Ph/t-Bu) | 2f (2-Thienyl/Et) | 5o | 80:20 | 67 | 86 |
aReactions were performed with 1 (0.15 mmol), 2 (0.1 mmol) and 4b (0.02 mmol) in toluene (0.5 mL) at room temperature. bDetermined by 1H NMR analysis of the crude reaction mixture. cYield of isolated product. dDetermined by HPLC analysis on a chiral stationary phase.
A possible reaction mechanism rationalizing the formation of the [3 + 2] annulation product is shown in Scheme 2 [34-36,48]. The reaction is initiated by the activation of the allenoate through a nucleophilic attack of the phosphine, generating zwitterionic intermediate 6, which undergoes a [3 + 2] annulation with imine 2 to furnish intermediate 8. Due to the lack of a hydrogen atom at the α-position, the normal proton shift in a typical [3 + 2] annulation cannot occur. Instead, this intermediate undergoes a proton shift to generate intermediate 9, where a [1,4]-proton shift can occur to yield intermediate 10. Lastly, elimination of the phosphine catalyst furnishes the final [3 + 2] annulation product 5.
Conclusion
In conclusion, we have described the first enantioselective [3 + 2] cycloaddition of α-substituted allenoates with β,γ-unsaturated N-sulfonylimines, catalyzed by amino acid-derived bifunctional phosphines. The [3 + 2] annulation reactions yielded highly functionalized cyclopentenes with an all-carbon quaternary center in moderate to good yields and good to excellent enantioselectivities. Further extension of the reaction reported herein and mechanistic studies are ongoing in our laboratory.
Experimental
General procedure for the [3 + 2] annulation
Into a flame-dried round bottle flask with a magnetic stirring bar under N2 at room temperature were added allenoate 1 (0.15 mmol) and β,γ-unsaturated N-sulfonylimine 2 (0.1 mmol), followed by the addition of anhydrous toluene (0.5 mL). Catalyst 4b (0.02 mmol, 14.5 mg) was then introduced, and the reaction mixture was stirred at room temperature for 24 h. After complete consumption of the β,γ-unsaturated N-sulfonylimine, monitored by TLC, the solvent was removed under reduced pressure and the residue was purified by column chromatography on silica gel to afford annulation adducts 5.
Supporting Information
| Supporting Information File 1: Additional material. | ||
| Format: PDF | Size: 2.8 MB | Download |
References
-
Lu, X.; Zhang, C.; Xu, Z. Acc. Chem. Res. 2001, 34, 535–544. doi:10.1021/ar000253x
Return to citation in text: [1] -
Methot, J. L.; Roush, W. R. Adv. Synth. Catal. 2004, 346, 1035–1050. doi:10.1002/adsc.200404087
Return to citation in text: [1] -
Lu, X.; Du, Y.; Lu, C. Pure Appl. Chem. 2005, 77, 1985–1990. doi:10.1351/pac200577121985
Return to citation in text: [1] -
Ye, L.-W.; Zhou, J.; Tang, Y. Chem. Soc. Rev. 2008, 37, 1140–1152. doi:10.1039/b717758e
Return to citation in text: [1] -
Cowen, B. J.; Miller, S. J. Chem. Soc. Rev. 2009, 38, 3102–3116. doi:10.1039/b816700c
Return to citation in text: [1] -
Wei, Y.; Shi, M. Acc. Chem. Res. 2010, 43, 1005–1018. doi:10.1021/ar900271g
Return to citation in text: [1] -
Marinetti, A.; Voituriez, A. Synlett 2010, 174–194. doi:10.1055/s-0029-1219157
Return to citation in text: [1] -
Wang, S.-X.; Han, X.; Zhong, F.; Wang, Y.; Lu, Y. Synlett 2011, 2766–2778. doi:10.1055/s-0031-1289538
Return to citation in text: [1] -
Zhao, Q.-Y.; Lian, Z.; Wei, Y.; Shi, M. Chem. Commun. 2012, 48, 1724–1732. doi:10.1039/c1cc15793k
Return to citation in text: [1] -
Fan, Y. C.; Kwon, O. Chem. Commun. 2013, 49, 11588–11619. doi:10.1039/c3cc47368f
Return to citation in text: [1] -
Wang, Z.; Xu, X.; Kwon, O. Chem. Soc. Rev. 2014, 43, 2927–2940. doi:10.1039/c4cs00054d
Return to citation in text: [1] -
Xiao, Y.; Sun, Z.; Guo, H.; Kwon, O. Beilstein J. Org. Chem. 2014, 10, 2089–2121. doi:10.3762/bjoc.10.218
Return to citation in text: [1] -
Wei, Y.; Shi, M. Chem. – Asian J. 2014, 9, 2720–2734. doi:10.1002/asia.201402109
Return to citation in text: [1] -
Zhang, C.; Lu, X. J. Org. Chem. 1995, 60, 2906–2908. doi:10.1021/jo00114a048
Return to citation in text: [1] -
Zhu, G.; Chen, Z.; Jiang, Q.; Xiao, D.; Cao, P.; Zhang, X. J. Am. Chem. Soc. 1997, 119, 3836–3837. doi:10.1021/ja9644687
Return to citation in text: [1] -
Du, Y.; Lu, X.; Yu, Y. J. Org. Chem. 2002, 67, 8901–8905. doi:10.1021/jo026111t
Return to citation in text: [1] -
Du, Y.; Lu, X. J. Org. Chem. 2003, 68, 6463–6465. doi:10.1021/jo034281f
Return to citation in text: [1] -
Pham, T. Q.; Pyne, S. G.; Skelton, B. W.; White, A. H. J. Org. Chem. 2005, 70, 6369–6377. doi:10.1021/jo050827h
Return to citation in text: [1] -
Lu, X.; Lu, Z.; Zhang, X. Tetrahedron 2006, 62, 457–460. doi:10.1016/j.tet.2005.09.100
Return to citation in text: [1] -
Wilson, J. E.; Fu, G. C. Angew. Chem., Int. Ed. 2006, 45, 1426–1429. doi:10.1002/anie.200503312
Return to citation in text: [1] -
Henry, C. E.; Kwon, O. Org. Lett. 2007, 9, 3069–3072. doi:10.1021/ol071181d
Return to citation in text: [1] -
Cowen, B. J.; Miller, S. J. J. Am. Chem. Soc. 2007, 129, 10988–10989. doi:10.1021/ja0734243
Return to citation in text: [1] -
Voituriez, A.; Panossian, A.; Fleury-Brégeot, N.; Retailleau, P.; Marinetti, A. J. Am. Chem. Soc. 2008, 130, 14030–14031. doi:10.1021/ja806060a
Return to citation in text: [1] -
Jones, R. A.; Krische, M. J. Org. Lett. 2009, 11, 1849–1851. doi:10.1021/ol900360h
Return to citation in text: [1] -
Voituriez, A.; Pinto, N.; Neel, M.; Retailleau, P.; Marinetti, A. Chem. – Eur. J. 2010, 16, 12541–12544. doi:10.1002/chem.201001791
Return to citation in text: [1] -
Xiao, H.; Chai, Z.; Zheng, C.-W.; Yang, Y.-Q.; Liu, W.; Zhang, J.-K.; Zhao, G. Angew. Chem., Int. Ed. 2010, 49, 4467–4470. doi:10.1002/anie.201000446
Return to citation in text: [1] -
Fujiwara, Y.; Fu, G. C. J. Am. Chem. Soc. 2011, 133, 12293–12297. doi:10.1021/ja2049012
Return to citation in text: [1] -
Han, X.; Wang, Y.; Zhong, F.; Lu, Y. J. Am. Chem. Soc. 2011, 133, 1726–1729. doi:10.1021/ja1106282
Return to citation in text: [1] -
Zhang, X.-C.; Cao, S.-H.; Wei, Y.; Shi, M. Chem. Commun. 2011, 47, 1548–1550. doi:10.1039/c0cc04289g
Return to citation in text: [1] -
Zhao, Q.; Han, X.; Wei, Y.; Shi, M.; Lu, Y. Chem. Commun. 2012, 48, 970–972. doi:10.1039/c2cc16904e
Return to citation in text: [1] -
Dakas, P.-Y.; Parga, J. A.; Höing, S.; Schöler, H. R.; Sterneckert, J.; Kumar, K.; Waldmann, H. Angew. Chem., Int. Ed. 2013, 52, 9576–9581. doi:10.1002/anie.201302045
Return to citation in text: [1] -
Marco-Martínez, J.; Marcos, V.; Reboredo, S.; Filippone, S.; Martín, N. Angew. Chem., Int. Ed. 2013, 52, 5115–5119. doi:10.1002/anie.201301292
Return to citation in text: [1] -
Wang, D.; Wang, G.-P.; Sun, Y.-L.; Zhu, S.-F.; Wei, Y.; Zhou, Q.-L.; Shi, M. Chem. Sci. 2015, 6, 7319–7325. doi:10.1039/c5sc03135d
Return to citation in text: [1] -
Xia, Y.; Liang, Y.; Chen, Y.; Wang, M.; Jiao, L.; Huang, F.; Liu, S.; Li, Y.; Yu, Z.-X. J. Am. Chem. Soc. 2007, 129, 3470–3471. doi:10.1021/ja068215h
Return to citation in text: [1] [2] -
Mercier, E.; Fonovic, B.; Henry, C.; Kwon, O.; Dudding, T. Tetrahedron Lett. 2007, 48, 3617–3620. doi:10.1016/j.tetlet.2007.03.030
Return to citation in text: [1] [2] -
Liang, Y.; Liu, S.; Xia, Y.; Li, Y.; Yu, Z.-X. Chem. – Eur. J. 2008, 14, 4361–4373. doi:10.1002/chem.200701725
Return to citation in text: [1] [2] -
Zhu, X.-F.; Lan, J.; Kwon, O. J. Am. Chem. Soc. 2003, 125, 4716–4717. doi:10.1021/ja0344009
Return to citation in text: [1] -
Tran, Y. S.; Kwon, O. Org. Lett. 2005, 7, 4289–4291. doi:10.1021/ol051799s
Return to citation in text: [1] -
Tran, Y. S.; Kwon, O. J. Am. Chem. Soc. 2007, 129, 12632–12633. doi:10.1021/ja0752181
Return to citation in text: [1] -
Tran, Y. S.; Martin, T. J.; Kwon, O. Chem. – Asian J. 2011, 6, 2101–2106. doi:10.1002/asia.201100190
Return to citation in text: [1] -
Wurz, R. P.; Fu, G. C. J. Am. Chem. Soc. 2005, 127, 12234–12235. doi:10.1021/ja053277d
Return to citation in text: [1] -
Wang, T.; Ye, S. Org. Lett. 2010, 12, 4168–4171. doi:10.1021/ol101762z
Return to citation in text: [1] -
Baskar, B.; Dakas, P.-Y.; Kumar, K. Org. Lett. 2011, 13, 1988–1991. doi:10.1021/ol200389p
Return to citation in text: [1] -
Xiao, H.; Chai, Z.; Wang, H.-F.; Wang, X.-W.; Cao, D.-D.; Liu, W.; Lu, Y.-P.; Yang, Y.-Q.; Zhao, G. Chem. – Eur. J. 2011, 17, 10562–10565. doi:10.1002/chem.201100850
Return to citation in text: [1] -
Chen, X.-Y.; Ye, S. Eur. J. Org. Chem. 2012, 5723–5728. doi:10.1002/ejoc.201200747
Return to citation in text: [1] -
Zhong, F.; Han, X.; Wang, Y.; Lu, Y. Chem. Sci. 2012, 3, 1231–1234. doi:10.1039/c2sc00963c
Return to citation in text: [1] -
Yu, H.; Zhang, L.; Li, Z.; Liu, H.; Wang, B.; Xiao, Y.; Guo, H. Tetrahedron 2014, 70, 340–348. doi:10.1016/j.tet.2013.11.063
Return to citation in text: [1] -
Tian, J.; He, Z. Chem. Commun. 2013, 49, 2058–2060. doi:10.1039/c3cc38264h
Return to citation in text: [1] [2] -
Wang, T.; Yu, Z.; Hoon, D. L.; Huang, K.-W.; Lan, Y.; Lu, Y. Chem. Sci. 2015, 6, 4912–4922. doi:10.1039/c5sc01614b
Return to citation in text: [1] -
Wang, T.; Hoon, D. L.; Lu, Y. Chem. Commun. 2015, 51, 10186–10189. doi:10.1039/c5cc03289j
Return to citation in text: [1] -
Yao, W.; Dou, X.; Lu, Y. J. Am. Chem. Soc. 2015, 137, 54–57. doi:10.1021/ja5109358
Return to citation in text: [1] -
Gandi, V. R.; Lu, Y. Chem. Commun. 2015, 51, 16188–16190. doi:10.1039/c5cc06197k
Return to citation in text: [1] -
Han, X.; Yao, W.; Wang, T.; Tan, Y. R.; Yan, Z.; Kwiatkowski, J.; Lu, Y. Angew. Chem., Int. Ed. 2014, 53, 5643–5647. doi:10.1002/anie.201311214
Return to citation in text: [1] -
Wang, T.; Yao, W.; Zhong, F.; Pang, G. H.; Lu, Y. Angew. Chem., Int. Ed. 2014, 53, 2964–2968. doi:10.1002/anie.201307757
Return to citation in text: [1] -
Zhong, F.; Dou, X.; Han, X.; Yao, W.; Zhu, Q.; Meng, Y.; Lu, Y. Angew. Chem., Int. Ed. 2013, 52, 943–947. doi:10.1002/anie.201208285
Return to citation in text: [1] -
Lee, R.; Zhong, F.; Zheng, B.; Meng, Y.; Lu, Y.; Huang, K.-W. Org. Biomol. Chem. 2013, 11, 4818–4824. doi:10.1039/C3OB40144H
Return to citation in text: [1] -
Zhong, F.; Luo, J.; Chen, G.-Y.; Dou, X.; Lu, Y. J. Am. Chem. Soc. 2012, 134, 10222–10227. doi:10.1021/ja303115m
Return to citation in text: [1] -
Zhong, F.; Chen, G.-Y.; Han, X.; Yao, W.; Lu, Y. Org. Lett. 2012, 14, 3764–3767. doi:10.1021/ol301647g
Return to citation in text: [1] -
Han, X.; Zhong, F.; Wang, Y.; Lu, Y. Angew. Chem., Int. Ed. 2012, 51, 767–770. doi:10.1002/anie.201106672
Return to citation in text: [1] -
Zhong, F.; Wang, Y.; Han, X.; Huang, K.-W.; Lu, Y. Org. Lett. 2011, 13, 1310–1313. doi:10.1021/ol103145g
Return to citation in text: [1] -
Zhong, F.; Han, X.; Wang, Y.; Lu, Y. Angew. Chem., Int. Ed. 2011, 123, 7983–7987. doi:10.1002/ange.201102094
Return to citation in text: [1] -
Han, X.; Wang, Y.; Zhong, F.; Lu, Y. Org. Biomol. Chem. 2011, 9, 6734–6740. doi:10.1039/c1ob05881a
Return to citation in text: [1] -
Han, X.; Wang, S.-X.; Zhong, F.; Lu, Y. Synthesis 2011, 1859–1864. doi:10.1055/s-0030-1260460
Return to citation in text: [1]
| 1. | Lu, X.; Zhang, C.; Xu, Z. Acc. Chem. Res. 2001, 34, 535–544. doi:10.1021/ar000253x |
| 2. | Methot, J. L.; Roush, W. R. Adv. Synth. Catal. 2004, 346, 1035–1050. doi:10.1002/adsc.200404087 |
| 3. | Lu, X.; Du, Y.; Lu, C. Pure Appl. Chem. 2005, 77, 1985–1990. doi:10.1351/pac200577121985 |
| 4. | Ye, L.-W.; Zhou, J.; Tang, Y. Chem. Soc. Rev. 2008, 37, 1140–1152. doi:10.1039/b717758e |
| 5. | Cowen, B. J.; Miller, S. J. Chem. Soc. Rev. 2009, 38, 3102–3116. doi:10.1039/b816700c |
| 6. | Wei, Y.; Shi, M. Acc. Chem. Res. 2010, 43, 1005–1018. doi:10.1021/ar900271g |
| 7. | Marinetti, A.; Voituriez, A. Synlett 2010, 174–194. doi:10.1055/s-0029-1219157 |
| 8. | Wang, S.-X.; Han, X.; Zhong, F.; Wang, Y.; Lu, Y. Synlett 2011, 2766–2778. doi:10.1055/s-0031-1289538 |
| 9. | Zhao, Q.-Y.; Lian, Z.; Wei, Y.; Shi, M. Chem. Commun. 2012, 48, 1724–1732. doi:10.1039/c1cc15793k |
| 10. | Fan, Y. C.; Kwon, O. Chem. Commun. 2013, 49, 11588–11619. doi:10.1039/c3cc47368f |
| 11. | Wang, Z.; Xu, X.; Kwon, O. Chem. Soc. Rev. 2014, 43, 2927–2940. doi:10.1039/c4cs00054d |
| 12. | Xiao, Y.; Sun, Z.; Guo, H.; Kwon, O. Beilstein J. Org. Chem. 2014, 10, 2089–2121. doi:10.3762/bjoc.10.218 |
| 13. | Wei, Y.; Shi, M. Chem. – Asian J. 2014, 9, 2720–2734. doi:10.1002/asia.201402109 |
| 37. | Zhu, X.-F.; Lan, J.; Kwon, O. J. Am. Chem. Soc. 2003, 125, 4716–4717. doi:10.1021/ja0344009 |
| 38. | Tran, Y. S.; Kwon, O. Org. Lett. 2005, 7, 4289–4291. doi:10.1021/ol051799s |
| 39. | Tran, Y. S.; Kwon, O. J. Am. Chem. Soc. 2007, 129, 12632–12633. doi:10.1021/ja0752181 |
| 40. | Tran, Y. S.; Martin, T. J.; Kwon, O. Chem. – Asian J. 2011, 6, 2101–2106. doi:10.1002/asia.201100190 |
| 41. | Wurz, R. P.; Fu, G. C. J. Am. Chem. Soc. 2005, 127, 12234–12235. doi:10.1021/ja053277d |
| 42. | Wang, T.; Ye, S. Org. Lett. 2010, 12, 4168–4171. doi:10.1021/ol101762z |
| 43. | Baskar, B.; Dakas, P.-Y.; Kumar, K. Org. Lett. 2011, 13, 1988–1991. doi:10.1021/ol200389p |
| 44. | Xiao, H.; Chai, Z.; Wang, H.-F.; Wang, X.-W.; Cao, D.-D.; Liu, W.; Lu, Y.-P.; Yang, Y.-Q.; Zhao, G. Chem. – Eur. J. 2011, 17, 10562–10565. doi:10.1002/chem.201100850 |
| 45. | Chen, X.-Y.; Ye, S. Eur. J. Org. Chem. 2012, 5723–5728. doi:10.1002/ejoc.201200747 |
| 46. | Zhong, F.; Han, X.; Wang, Y.; Lu, Y. Chem. Sci. 2012, 3, 1231–1234. doi:10.1039/c2sc00963c |
| 47. | Yu, H.; Zhang, L.; Li, Z.; Liu, H.; Wang, B.; Xiao, Y.; Guo, H. Tetrahedron 2014, 70, 340–348. doi:10.1016/j.tet.2013.11.063 |
| 34. | Xia, Y.; Liang, Y.; Chen, Y.; Wang, M.; Jiao, L.; Huang, F.; Liu, S.; Li, Y.; Yu, Z.-X. J. Am. Chem. Soc. 2007, 129, 3470–3471. doi:10.1021/ja068215h |
| 35. | Mercier, E.; Fonovic, B.; Henry, C.; Kwon, O.; Dudding, T. Tetrahedron Lett. 2007, 48, 3617–3620. doi:10.1016/j.tetlet.2007.03.030 |
| 36. | Liang, Y.; Liu, S.; Xia, Y.; Li, Y.; Yu, Z.-X. Chem. – Eur. J. 2008, 14, 4361–4373. doi:10.1002/chem.200701725 |
| 14. | Zhang, C.; Lu, X. J. Org. Chem. 1995, 60, 2906–2908. doi:10.1021/jo00114a048 |
| 15. | Zhu, G.; Chen, Z.; Jiang, Q.; Xiao, D.; Cao, P.; Zhang, X. J. Am. Chem. Soc. 1997, 119, 3836–3837. doi:10.1021/ja9644687 |
| 16. | Du, Y.; Lu, X.; Yu, Y. J. Org. Chem. 2002, 67, 8901–8905. doi:10.1021/jo026111t |
| 17. | Du, Y.; Lu, X. J. Org. Chem. 2003, 68, 6463–6465. doi:10.1021/jo034281f |
| 18. | Pham, T. Q.; Pyne, S. G.; Skelton, B. W.; White, A. H. J. Org. Chem. 2005, 70, 6369–6377. doi:10.1021/jo050827h |
| 19. | Lu, X.; Lu, Z.; Zhang, X. Tetrahedron 2006, 62, 457–460. doi:10.1016/j.tet.2005.09.100 |
| 20. | Wilson, J. E.; Fu, G. C. Angew. Chem., Int. Ed. 2006, 45, 1426–1429. doi:10.1002/anie.200503312 |
| 21. | Henry, C. E.; Kwon, O. Org. Lett. 2007, 9, 3069–3072. doi:10.1021/ol071181d |
| 22. | Cowen, B. J.; Miller, S. J. J. Am. Chem. Soc. 2007, 129, 10988–10989. doi:10.1021/ja0734243 |
| 23. | Voituriez, A.; Panossian, A.; Fleury-Brégeot, N.; Retailleau, P.; Marinetti, A. J. Am. Chem. Soc. 2008, 130, 14030–14031. doi:10.1021/ja806060a |
| 24. | Jones, R. A.; Krische, M. J. Org. Lett. 2009, 11, 1849–1851. doi:10.1021/ol900360h |
| 25. | Voituriez, A.; Pinto, N.; Neel, M.; Retailleau, P.; Marinetti, A. Chem. – Eur. J. 2010, 16, 12541–12544. doi:10.1002/chem.201001791 |
| 26. | Xiao, H.; Chai, Z.; Zheng, C.-W.; Yang, Y.-Q.; Liu, W.; Zhang, J.-K.; Zhao, G. Angew. Chem., Int. Ed. 2010, 49, 4467–4470. doi:10.1002/anie.201000446 |
| 27. | Fujiwara, Y.; Fu, G. C. J. Am. Chem. Soc. 2011, 133, 12293–12297. doi:10.1021/ja2049012 |
| 28. | Han, X.; Wang, Y.; Zhong, F.; Lu, Y. J. Am. Chem. Soc. 2011, 133, 1726–1729. doi:10.1021/ja1106282 |
| 29. | Zhang, X.-C.; Cao, S.-H.; Wei, Y.; Shi, M. Chem. Commun. 2011, 47, 1548–1550. doi:10.1039/c0cc04289g |
| 30. | Zhao, Q.; Han, X.; Wei, Y.; Shi, M.; Lu, Y. Chem. Commun. 2012, 48, 970–972. doi:10.1039/c2cc16904e |
| 31. | Dakas, P.-Y.; Parga, J. A.; Höing, S.; Schöler, H. R.; Sterneckert, J.; Kumar, K.; Waldmann, H. Angew. Chem., Int. Ed. 2013, 52, 9576–9581. doi:10.1002/anie.201302045 |
| 32. | Marco-Martínez, J.; Marcos, V.; Reboredo, S.; Filippone, S.; Martín, N. Angew. Chem., Int. Ed. 2013, 52, 5115–5119. doi:10.1002/anie.201301292 |
| 33. | Wang, D.; Wang, G.-P.; Sun, Y.-L.; Zhu, S.-F.; Wei, Y.; Zhou, Q.-L.; Shi, M. Chem. Sci. 2015, 6, 7319–7325. doi:10.1039/c5sc03135d |
| 34. | Xia, Y.; Liang, Y.; Chen, Y.; Wang, M.; Jiao, L.; Huang, F.; Liu, S.; Li, Y.; Yu, Z.-X. J. Am. Chem. Soc. 2007, 129, 3470–3471. doi:10.1021/ja068215h |
| 35. | Mercier, E.; Fonovic, B.; Henry, C.; Kwon, O.; Dudding, T. Tetrahedron Lett. 2007, 48, 3617–3620. doi:10.1016/j.tetlet.2007.03.030 |
| 36. | Liang, Y.; Liu, S.; Xia, Y.; Li, Y.; Yu, Z.-X. Chem. – Eur. J. 2008, 14, 4361–4373. doi:10.1002/chem.200701725 |
| 48. | Tian, J.; He, Z. Chem. Commun. 2013, 49, 2058–2060. doi:10.1039/c3cc38264h |
| 49. | Wang, T.; Yu, Z.; Hoon, D. L.; Huang, K.-W.; Lan, Y.; Lu, Y. Chem. Sci. 2015, 6, 4912–4922. doi:10.1039/c5sc01614b |
| 50. | Wang, T.; Hoon, D. L.; Lu, Y. Chem. Commun. 2015, 51, 10186–10189. doi:10.1039/c5cc03289j |
| 51. | Yao, W.; Dou, X.; Lu, Y. J. Am. Chem. Soc. 2015, 137, 54–57. doi:10.1021/ja5109358 |
| 52. | Gandi, V. R.; Lu, Y. Chem. Commun. 2015, 51, 16188–16190. doi:10.1039/c5cc06197k |
| 53. | Han, X.; Yao, W.; Wang, T.; Tan, Y. R.; Yan, Z.; Kwiatkowski, J.; Lu, Y. Angew. Chem., Int. Ed. 2014, 53, 5643–5647. doi:10.1002/anie.201311214 |
| 54. | Wang, T.; Yao, W.; Zhong, F.; Pang, G. H.; Lu, Y. Angew. Chem., Int. Ed. 2014, 53, 2964–2968. doi:10.1002/anie.201307757 |
| 55. | Zhong, F.; Dou, X.; Han, X.; Yao, W.; Zhu, Q.; Meng, Y.; Lu, Y. Angew. Chem., Int. Ed. 2013, 52, 943–947. doi:10.1002/anie.201208285 |
| 56. | Lee, R.; Zhong, F.; Zheng, B.; Meng, Y.; Lu, Y.; Huang, K.-W. Org. Biomol. Chem. 2013, 11, 4818–4824. doi:10.1039/C3OB40144H |
| 57. | Zhong, F.; Luo, J.; Chen, G.-Y.; Dou, X.; Lu, Y. J. Am. Chem. Soc. 2012, 134, 10222–10227. doi:10.1021/ja303115m |
| 58. | Zhong, F.; Chen, G.-Y.; Han, X.; Yao, W.; Lu, Y. Org. Lett. 2012, 14, 3764–3767. doi:10.1021/ol301647g |
| 59. | Han, X.; Zhong, F.; Wang, Y.; Lu, Y. Angew. Chem., Int. Ed. 2012, 51, 767–770. doi:10.1002/anie.201106672 |
| 60. | Zhong, F.; Wang, Y.; Han, X.; Huang, K.-W.; Lu, Y. Org. Lett. 2011, 13, 1310–1313. doi:10.1021/ol103145g |
| 61. | Zhong, F.; Han, X.; Wang, Y.; Lu, Y. Angew. Chem., Int. Ed. 2011, 123, 7983–7987. doi:10.1002/ange.201102094 |
| 62. | Han, X.; Wang, Y.; Zhong, F.; Lu, Y. Org. Biomol. Chem. 2011, 9, 6734–6740. doi:10.1039/c1ob05881a |
| 63. | Han, X.; Wang, S.-X.; Zhong, F.; Lu, Y. Synthesis 2011, 1859–1864. doi:10.1055/s-0030-1260460 |
© 2016 Ni et al; licensee Beilstein-Institut.
This is an Open Access article under the terms of the Creative Commons Attribution License (http://creativecommons.org/licenses/by/2.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
The license is subject to the Beilstein Journal of Organic Chemistry terms and conditions: (http://www.beilstein-journals.org/bjoc)