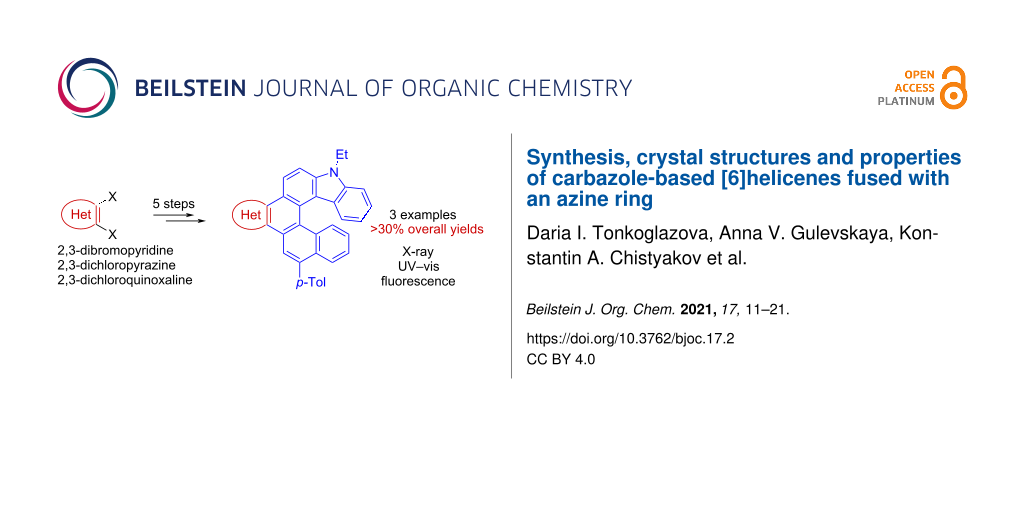
Abstract
Novel carbazole-based [6]helicenes fused with an azine ring (pyridine, pyrazine or quinoxaline) have been prepared through a five-step synthetic sequence in good overall yields. Commercially available 2,3-dihaloazines were used as starting materials. To discern the effect of merging an azine moiety within a helical skeleton, the X-ray structures, UV–vis absorption and fluorescence spectra of the helicenes were investigated and compared to that of the parent carbazole-based [6]helicene (7H-phenanthro[3,4-c]carbazole).
Graphical Abstract
Introduction
[n]Helicenes are polycyclic aromatic molecules with nonplanar screw-shaped helical skeletons formed by n-ortho-fused benzene or other aromatic rings. Their helical structure is a consequence of the steric repulsion of the terminal aromatic nuclei. The steric strain releases by adopting either the P- or M-helix configuration. The helically extended π-conjugated system, axial chirality and associated with these structural peculiarities unique optical and electronic properties of helicenes have attracted scientific interest for decades [1-11]. Compared to other planar π-conjugated systems, helicenes are more thermally stable and soluble in common organic solvents [9]. This fact together with the exceptionally high values of specific optical rotation and strong circular dichroism have led to promising applications of helicenes. The latter have been studied with respect to conductivity [12-15], nonlinear optics [16,17], circularly polarized luminescence [18-24], organocatalysis [25-29], conformational analysis [30], chirality sensing [31], chemical sensors [32], DNA-intercalators [33,34] etc.
Besides typical carbohelicenes, heterohelicenes, incorporating one or more heteroaromatic units in the skeleton, have also gained increasing attention [1-11]. The presence of heteroatoms (S, N, O, P) in the fused polycyclic π-systems additionally contributes to altering electronic structure and helps to fine tune optoelectronic properties [1-11,20,35-37]. The last decades highlighted heterohelicenes, incorporating one or two carbazole fragments, as a very attractive class of molecules [38-51]. This is not surprising, taking into account the excellent thermal stability, the strong electron-donating nature, a good hole-transporting ability of the carbazole unit and, as a consequence, numerous applications of the carbazole-based electroactive materials [52-54]. Carbazole-based [6]helicenes [42] and [7]helicenes [50] showed deep blue electroluminescence and have been investigated in OLED devices. Some carbazole-based [5]- and [6]helicenes have been used as visible light photoinitiators for cationic and radical polymerization [41]. [7]Helicenes of this group demonstrated a relatively high electron affinity and could be good candidates for electron-injection hole-blocking layers [39]. Donor–acceptor hybride [6]helicenes, consisting of carbazole and phenanthridine cores, are interesting as hole-transporting compounds [47]. At last, carbazole-based heterohelicenes were found in nature, for example, purpurone [55] isolated from the marine sponge Iotrochota sp. and having an inhibitory effect on the ATP-citrate lyase, and the marine alkaloid ningalin D produced by Didemnum sp., Dictyodendrins [56] isolated from Dictyodendrilla sp. and displaying inhibitory activities towards telomerases.
The classical synthetic approach for carbohelicenes is the oxidative photocyclization of stilbene derivatives [1-11]. The latter are generally available via the Wittig, Heck-type or McMurry couplings. It is also a useful way to synthesize heterohelicenes, in particular, carbazole-based helicenes [39-42,44,47,48,50,51] (Scheme 1A). However, photocyclization of the stilbene substrates, having two non-equivalent ortho positions, leads to the formation of isomeric polynuclear molecules, which are often difficult to separate. Another drawback of this method is the difficulty of scaling, since the reaction requires strong dilution to prevent the [2π + 2π] dimerization of the starting stilbene. Among other approaches to the carbazole-based helicenes are the Diels–Alder reaction of silyl enol ethers of 3,6-diacetylcarbazole with p-benzoquinone (Scheme 1B) [49], the double Buchwald–Hartwig amination of 4,4'-biphenanthrene derivatives (Scheme 1C) [45] and a enantioselective Fischer indolization–oxidation protocol (Scheme 1D) [43]. Each method is not without drawbacks such as hardly available starting materials, rather expensive catalysts, harsh reaction conditions or low product yields.
Scheme 1: Overview of the synthetic methods for the carbazole-based heterohelicenes. i) Pd2dba3, xantphos, K3PO4, xylene, 100 °C, 123 h, 64–88% ii) SPINOL-derived phosphoric acid, Amberlite CG50, CH2Cl2, −7 °C, 72 h, 74%; iii) chloranil, diphenyl phosphate, CHCl3, 50 °C, 5 h, 76%.
Scheme 1: Overview of the synthetic methods for the carbazole-based heterohelicenes. i) Pd2dba3, xantphos, K3...
Transition-metal-catalyzed, electrophile-induced and oxidative radical cyclizations of ortho-alkynylated biaryls are widely used for the synthesis of polynuclear aromatics [57-69]. Recently, we have described a versatile method for the preparation of aza[4]helicenes [70], diaza[4]helicenes [70,71] and azine-fused [5]helicenes [72] through a five-step synthetic sequence, using commercially available 2,3-dihaloazines as starting materials. Based on this approach, we synthesized carbazole-based [6]helicenes fused with an azine ring (quinoxaline, pyrazine or pyridine ones). Scheme 2 represents the current work. We envisaged that combining the carbazole unit with the readily available azine building block would result in the formation of donor–acceptor hybrid helicenes with interesting electronic properties.
Scheme 2: Synthetic strategy for the carbazole-based [6]helicenes fused with an azine ring.
Scheme 2: Synthetic strategy for the carbazole-based [6]helicenes fused with an azine ring.
Results and Discussion
Synthesis
In accordance with the above strategy, we first synthesized ortho-halogen alkynylazines 1a,b via the Sonogashira reaction of commercially available 2,3-dichloroquinoxaline and 2,3-dichloropyrazine with phenylacetylene using a known procedure [70]. Coupling of compounds 1a,b with 9-ethyl-3-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-9H-carbazole in the Pd(PPh3)4/K2CO3/1,4-dioxane/H2O catalytic system for 24 h at 100 °C (method C) afforded the desired 3-alkynyl-2-carbazolylazines 2a,b in 82–96% yields (Table 1). Other catalytic systems Pd(PPh3)4/K3PO4/THF (method A) and Pd(PPh3)4/K3PO4/1,4-dioxane (method B) were less effective.
Table 1: Suzuki coupling of compounds 1a,b with 9-ethyl-3-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-9H-carbazole.
|
|
|||
| Entry | R, R | Product |
Yield, %
(method) |
| 1 | -(CH=CH)2- | 2a |
36 (A)
44 (B) 96 (C) |
| 2 | H, H | 2b | 82 (C) |
aMethod A: Pd(PPh3)4, K3PO4, THF, reflux, 24 h, argon; method B: Pd(PPh3)4, K3PO4, 1,4-dioxane, 100 °C, 24 h, argon; method C: Pd(PPh3)4, K2CO3, 1,4-dioxane, H2O, 100 °C, 24 h, argon.
3-Alkynyl-2-carbazolylazines can also be prepared using an alternative synthetic sequence, i.e., the Suzuki–Miyaura arylation–Sonogashira reaction. It should be noted that in the case of 2,3-dibromopyridine it was the only way for us to synthesize the target [4]helicenes [70]. Unfortunately, the coupling of 2,3-dichloroquinoxaline (3a) with 9-ethyl-3-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-9H-carbazole in the 5% Pd-C/PPh3/K2CO3/toluene/H2O catalytic system at 100 °C for 24 h (method A) gave the corresponding carbazolyl derivative 4a in 15% yield only (Table 2). The Pd(PPh3)4/K2CO3/1,4-dioxane/H2O catalytic system (method B) was more effective producing 4a in 63% yield. Thus, it was not the way for the synthesis of compounds 2a,b. Arylation of 2,3-dibromopyridine using the Pd(PPh3)4/K2CO3/1,4-dioxane/H2O catalytic system gave a mixture of the corresponding mono- and dicarbazolyl derivatives 4b (80%) and 5b (8%). The products were easily separated by column chromatography. 3-Bromo-2-carbazolylpyridine 4b was then introduced into the Sonogashira reaction with phenylacetylene giving rise to 3-alkynyl-2-carbazolylpyridine 6 in 74% yield (Scheme 3).
Table 2: Suzuki coupling of compounds 3 with 9-ethyl-3-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-9H-carbazole.
|
|
|||||
| Entry | X | R, R | Hal | Product |
Yield, %
(method) |
| 1 | N | -(CH=CH)2- | Cl | 4a |
15 (A)
63 (B) |
| 2 | CH | H, H | Br |
4b
5b |
80 (B)
8 (B) |
aMethod A: 5% Pd/C, PPh3, K2CO3, H2O, toluene, 100 °C, 24 h, argon; method B: Pd(PPh3)4, K2CO3, 1,4-dioxane, H2O, 100 °C, 24 h, argon.
Scheme 3: Sonogashira coupling of compound 4b with phenylacetylene. i) Pd(PPh3)2Cl2, CuI, iPr2NH, DMSO, 80 °C, 24 h, argon.
Scheme 3: Sonogashira coupling of compound 4b with phenylacetylene. i) Pd(PPh3)2Cl2, CuI, iPr2NH, DMSO, 80 °C...
Electrophilic cyclizations of 3-alkynyl-2-carbazolylazines 2a,b and 6 into azine-fused carbazoles 7a–c were carried out with ICl in dry acetonitrile at room temperature in the dark (Table 3). In the cases of compounds 2a,b, the reaction was found to be very sensitive to the amount of ICl used. In the case of 2a, the use of a 1.5-fold excess of ICl led to a hardly separable mixture of compound 7a and diiodo derivative 8a (Table 3, entry 1, for the NMR spectrum of the mixture see Supporting Information File 1, Figure S13). Iodine chloride, taken in an equimolar amount, made it possible to obtain the required product 7a in 65% yield and to remove minor impurities of diiodo derivative 8a by chromatography (Table 3, entry 2). For the selective synthesis of 7b, iodine chloride was taken in a small deficit (Table 3, entries 3 and 4). Compound 8b was synthesized in 97% yield by the treatment with a 3-fold excess of ICl on monoiodide 7b. The ICl-induced cyclization of the pyridine-based starting compound 6 proceeded smoothly giving rise to product 7c in 62% (Table 3, entry 5). It should be noted that compounds 7a, 7b and 7c are derivatives of the previously unknown heterocyclic systems 1H-carbazolo[3,4-a]phenazine, 7H-quinoxalino[5,6-c]carbazole and 7H-quinolino[8,7-c]carbazole, respectively (SciFinder data).
The fourth step of the synthesis of the target helicenes, namely the Sonogashira coupling of iodides 7a and 7c with p-tolylacetylene, was carried out in the Pd(PPh3)2Cl2/CuI/Et3N/THF catalytic system that proved itself well in the azine-fused [5]helicenes synthesis [72] (Table 4, entries 1 and 3). The corresponding compounds 9a and 9c were obtained in 84 and 88% yields, respectively. In the case of 7b, the reaction was more selective without THF solvent producing alkyne 9b in 82% yield (Table 4, entry 2). The structure of 9c was unambiguously proved by X-ray structural analysis (see Supporting Information File 1, Figure S34).
Table 4: Sonogashira coupling of compounds 7 with p-tolylacetylene.
|
|
|||||
| Entry | X | R, R | Solvent | Product | Yield, % |
| 1 | N | -(CH=CH)2- | THF | 9a | 84 |
| 2 | N | H, H | – | 9b | 82 |
| 3 | CH | H, H | THF | 9c | 88 |
aPd(PPh3)2Cl2, CuI, Et3N, solvent, 85 °C, 24 h, argon.
Earlier, at the final step of a similar synthesis of the azine-fused [5]helicenes, we used trifluoroacetic acid as a cyclizing agent [72]. Unfortunately, only in the case of compound 9c, heating in trifluoroacetic acid led to isomerization into the required carbazole-based [6]helicene 10c (Table 5, entry 3). Under these conditions, alkynes 9a and 9b produced an unseparable mixture of some products. To our delight, treating 9a and 9b with triflic acid in СH2Cl2 solution at room temperature allowed us to obtain helicenes 10a and 10b in high yields (Table 5, entries 1 and 2). Apparently, the presence of the aza group adjacent to the triple bond in compounds 9a and 9b causes the observed difference in the reactivity of compounds 9. Its protonation makes the formation of a key cyclization intermediate 11 difficult.
It is known that the [n]helicenes with at least one five-membered heteroaromatic ring need n ≥ 6 to become intrinsically chiral [9,10]. High-performance liquid chromatography on chiral stationary phases confirmed the presence of configurationally stable (P)- and (M)-enantiomers at room temperature in the samples of synthesized helicenes 10a–c. In all cases, separation of enantiomers of 10 was achieved using Kromasil 5-Cellucoat column (4.6 mm × 250 mm), acetonitrile as a mobile phase and UV-detection (see Supporting Information File 1, Figures S38–S40).
X-ray molecular structures
The structures of the title compounds 10a–c were further explored by a single-crystal X-ray diffraction analysis (Figure 1, see also Supporting Information File 1, Figures S35–S37 and Table S1) and compared with that of the carbazole-based [6]helicene [42] (7-hexyl-7H-phenanthro[3,4-c]carbazole, 12) to see the effect of the azine ring annelation (Table 6).
![[1860-5397-17-2-1]](/bjoc/content/figures/1860-5397-17-2-1.png?scale=2.0&max-width=1024&background=FFFFFF)
Figure 1: Molecular structure of carbazole-based [6]helicenes 10a (a), 10b (b) and 10c (c) (X-ray data).
Figure 1: Molecular structure of carbazole-based [6]helicenes 10a (a), 10b (b) and 10c (c) (X-ray data).
Table 6: Comparison of X-ray data of the carbazole-based [6]helicenes (atomic numbering does not correspond to IUPAC nomenclature).
|
|
|||||||
| Helicene |
Space
group |
Bond length, Å |
Inner helix
angle, o |
Torsion anglea, o | Interplanar angleb, o | ||
| Inner helix | Ring D | ||||||
|
1–a
a–b b–c c–d d–e e–20 |
10–11
11–12 12–13 13–d d–c c–10 |
1–a–b
a–b–c b–c–d c–d–e d–e–20 |
1–a–b–c
a–b–c–d b–c–d–e c–d–e–20 |
9–10–11–f
g–12–13–14 |
A/F | ||
| 12 [42] | P21/c |
1.401
1.434 1.423 1.448 1.459 1.400 (8.565 in total) |
1.391
1.343 1.434 1.407 1.448 1.435 (8.458 in total) |
135.4
135.3 126.2 124.6 122.3 (643.8 in total) |
0.8
17.8 32.2 16.9 (67.7 in total) |
– | 56.7 |
| 10a | P21/c |
1.403
1.457 1.423 1.470 1.441 1.415 (8.609 in total) |
1.457
1.443 1.462 1.396 1.470 1.413 (8.641 in total) |
135.7
135.4 125.0 123.3 121.4 (640.8 in total) |
3.1
10.7 37.8 15.6 (67.2 in total) |
7.6
2.4 |
50.5 |
| 10bc | P21/n |
1.408
1.456 1.430 1.464 1.442 1.417 (8.721 in total) (8.617 in total)* |
1.449
1.412 1.451 1.400 1.464 1.418 (8.594 in total) (8.596 in total)* |
135.8
135.6 125.3 123.7 121.0 (641.4 in total) (640.6 in total)* |
0.8
11.3 32.3 21.6 (65.7 in total) (68.4 in total)* |
5.3
2.4 3.2* 2.0* |
50.6
53.7* |
| 10ca | Pca21 |
1.402
1.466 1.435 1.452 1.450 1.418 (8.623 in total) (8.603 in total)* |
1.461
1.411 1.451 1.401 1.452 1.414 (8.590 in total) (8.605 in total)* |
135.6
134.9 125.0 123.5 121.1 (640.1 in total) (640.8 in total)* |
3.0
14.0 33.9 22.7 (73.6 in total) (72.3 in total)* |
0.9
3.3 0.7* 3.4* |
62.3
61.6* |
aA dihedral angle between the four adjacent inner helix carbon atoms. bAn angle between the two terminal aromatic rings A and F of a helicene. cThere are two independent molecules in the unit cell. Data for the second molecule are marked with an asterisk *.
The values of the torsion and interplanar angles make it possible to describe and compare the structural specificity of the synthesized [6]helicenes. From the data given in Table 6, it can be seen that the interplanar angle between the terminal rings A and F of the pyridine-fused [6]helicene 10c is the largest in the series (average value is 62°). The same value for [6]helicene 12 is equal to 56.7° [42]. The helicity of pyrazine-fused [6]helicene 10b and quinoxaline-fused hybrid 10a is intermediate (52.8° and 50.5°). Comparing the sum of the inner helix torsion angles of the helicenes 10a–c and 12 reveals the same patterns. Evidently, the repulsive interaction of the H(14) and H(g) atoms of 10c is the main reason for the observed extra twisting, whereas relative planarization of 10a and 10b is the result of attractive interactions of the aza groups N(f) and N(g) with the H(9) and H(14) atoms, respectively. The short intramolecular contacts H(9)···N(f) (2.457 Å for 10a, 2.454 Å for 10b) and H(14)···N(g) (2.517 Å for 10a, 2.477 Å for 10b) support this opinion. Apparently, interaction of this type is also realized in solution since the signal of the inner helix proton in the 1H NMR spectra of azine-fused [6]helicenes appeared in the low field at δ 9.3–9.7 ppm. A similar pattern was observed by us in the structures of the azine-fused [5]helicenes [72].
It is well-known that the inner helix and outer helix C–C bonds of helicenes differ in their length [9]. Deviations of some of them from the standard aromatic C–C bond of benzene (1.393 Å) are significant. The length of the inner helix C–C bonds of [6]helicene 12 varies from 1.400 to 1.459 Å. On the contrary, the outer helix bonds of 12 are noticeably shortened (1.330–1.368 Å). The same tendency is observed in cases of azine-fused [6]helicenes 10. Their inner helix bonds are somewhat lengthened: 1.403–1.470 Å (for 10a), 1.408–1.462 Å (for 10b), 1.402–1.466 Å (for 10c). The shortest outer helix bonds of helicenes 10 are 1.369 Å (10a), 1.369 Å (10b) and 1.364 Å (10c). The maximum deviation from the standard value was recorded for the C–C bonds of the central D ring of the [6]helicenes 10a–c. In particular, the C(c)–C(d) length of quinoxaline-fused helicene 10a (1.470 Å) practically does not differ in its length from the standard single C(sp2)–C(sp2) bond (ca. 1.48 Å).
The X-ray analysis of quinoxaline-fused [6]helicene 10a revealed the presence of the face-to-face π–π interaction between the helicene aggregates. The racemic aggregation was composed by (P)- and (M)-enantiomers on the manner of embrace: π-deficient pyrazine ring of one enantiomer of 10a is located over the π-excessive pyrrole ring of another enantiomer (Figure 2a and Figure 2b). An intermolecular distance between the centroids of these rings was found to be equal to 3.74 Å making the π overlapping possible. In the case of helicene 10b, the enantiomeric molecules are aggregated into pairs differently: the pyrazine ring of one enantiomer is located over the E ring of another enantiomer and the distance between the layers is ca. 3.4 Å (Figure 2c and Figure 2d). The crystal packing of helicene 10c (Figure 2e) is peculiar: the alternating enantiomers form a screw along the a axis.
![[1860-5397-17-2-2]](/bjoc/content/figures/1860-5397-17-2-2.png?scale=2.0&max-width=1024&background=FFFFFF)
Figure 2: Crystal packing of carbazole-based [6]helicenes 10a (a, b), 10b (c,d) and 10c (e). Hydrogen atoms are hidden.
Figure 2: Crystal packing of carbazole-based [6]helicenes 10a (a, b), 10b (c,d) and 10c (e). Hydrogen atoms a...
Optical properties
All helicenes 10 are well soluble in dichloromethane and chloroform. Solubility in other common solvents such as acetonitrile, DMSO, tetrahydrofuran, ethanol and hexane is markedly lower. Carbazole-based [6]helicene 12 was described as yellow solid (the longest λmax 414 nm, CH2Cl2) [42]. All azine-fused analogs of 12 are yellow-orange. The annelation of the pyridine or pyrazine ring to the skeleton of 12 only slightly changed the wavelength of the absorption maximum (the longest λmax 411 and 418 nm, respectively), whereas the absorption of quinoxaline-fused [6]helicene 10a was red-shifted by 19 nm (Table 7). Compounds 10 exhibited almost a solvent independence of UV–vis absorption spectra (Supporting Information File 1, Figure S41). The optical band gaps (Egopt), estimated from the onset point of the absorption spectra, for [6]helicene 12 was equal to 2.92 eV [42]. The Egopt values for its π-extended analogs were 2.45 eV (10a), 2.76 eV (10b) and 2.85 eV (10c), suggesting a higher HOMO and lower oxidation potential, which are typically desired characteristics when designing organic materials. Unfortunately, for all azine-fused [6]helicenes 10 only weak fluorescence in the solution under UV irradiation was observed (Table 7, see also Supporting Information File 1, Figures S42–S45). Helicenes 10b and 10c exhibited blue emission with emission peaks at 481 and 440 nm, respectively. Quinoxaline-fused helicene 10a demonstrated a yellow emission with the highest in the series λem = 561 nm and Stokes shift 128 nm.
Table 7: Photophysical properties of carbazole-based [6]helicenes 10.
| Compd. | Absorption (CH2Cl2)a | Egopt, eVb | Absorption and emissionc (CH3CN) | |||
| λmax, nm | λonset, nm | λabs, nmd | λem, nm | Stokes shift, nm | ||
| 12 [42] | 282, 320, sh 347, 393, 414 | 425 | 2.92 | 414 | 426 | 12 |
| 10a | 264, 303, sh 324, 357, 374, 433 | 507 | 2.45 | 433 | 561 | 128 |
| 10b | 294, 323, 359, 397, 418 | 449 | 2.76 | 418 | 481 | 63 |
| 10c | 265, 285, sh 306, 324, 371, sh 388, 411 | 435 | 2.85 | 410 | 440 | 30 |
aAbsorption maxima measured in ≈10−5 M solution, abbreviation “sh” means shoulder. bThe optical gap was estimated from the onset point of the absorption spectra: Egopt = 1240/λonset.. cExcited at the longest absorption maxima. dThe only longest absorption maxima is shown.
Conclusion
In summary, novel carbazole-based [6]helicenes fused with an azine ring (pyridine, pyrazine or quinoxaline) have been prepared from commercially available 2,3-dihaloazines via a five-step synthetic sequence. Two key steps of the method are electrophile-induced 6-endo-dig cyclizations of ortho-alkynylated biaryls. The overall yields of helicenes in five stages of the synthesis exceed 30%.
The single-crystal X-ray diffraction analysis revealed the non-planar crystal structures of the synthesized helicenes responsible for reducing close-packing arrangement, though, in cases of pyrazine- and quinoxaline-fused helicenes, moderate π overlap in pairs of enantiomeric molecules was observed. Carbazole-based [6]helicene, fused with the pyridine ring, is more twisted than the parent carbazole-based [6]helicene (7H-phenanthro[3,4-c]carbazole). The interplanar angle between the two terminal benzene rings of the latter is equal to 56.7°, whereas the same value for the pyridine-fused analog is 62°. The pyrazine-fused [6]helicene demonstrates intermediate helicity (52.8°). In the case of the quinoxaline-fused analog the distortion angle of 50.5° is the smallest in the series.
The photophysical properties of the synthesized [6]helicenes were compared to the parent carbazole-based [6]helicene. A spectrophotometric analysis of the quinoxaline-fused helicene displayed a moderate absorption red-shift (19 nm) and reduced optical band gaps (by ≈0.5 eV). In cases of pyrazine and pyridine-fused analogs, differences are not so noticeable.
Experimental
The synthetic procedures, HPLC, X-ray studies and spectra (1H and 13C NMR) of all new compounds can be found in Supporting Information File 1.
Supporting Information
| Supporting Information File 1: Experimental procedures and analytical data, copies of 1H and 13C NMR spectra of all new compounds, X-ray data for 9c and 10a–c, HPLC spectra of helicenes 10a–c, UV–vis and fluorescence spectra of 10a–c. | ||
| Format: PDF | Size: 5.6 MB | Download |
Acknowledgements
The authors thank Drs. Anna V. Tkachuk and Oleg N. Burov (Scientific and Educational Laboratory of Resonance Spectroscopy, Department of Natural and High Molecular Compounds Chemistry, Southern Federal University) for NMR measurements. We also thank Daria V. Spiridonova and Dr. Alexander D. Misharev for X-ray studies and HRMS measurements (Centre for X-ray Diffraction Studies and Chemical Analysis and Materials Research Centre, Institute of Chemistry, St. Petersburg State University).
References
-
Shen, Y.; Chen, C.-F. Chem. Rev. 2012, 112, 1463–1535. doi:10.1021/cr200087r
Return to citation in text: [1] [2] [3] [4] -
Gingras, M. Chem. Soc. Rev. 2013, 42, 968–1006. doi:10.1039/c2cs35154d
Return to citation in text: [1] [2] [3] [4] -
Gingras, M.; Félix, G.; Peresutti, R. Chem. Soc. Rev. 2013, 42, 1007–1050. doi:10.1039/c2cs35111k
Return to citation in text: [1] [2] [3] [4] -
Gingras, M. Chem. Soc. Rev. 2013, 42, 1051–1095. doi:10.1039/c2cs35134j
Return to citation in text: [1] [2] [3] [4] -
Dumitrascu, F.; Dumitrescu, D. G.; Aron, I. ARKIVOC 2009, No. 1, 1–32. doi:10.3998/ark.5550190.0011.101
Return to citation in text: [1] [2] [3] [4] -
Saleh, N.; Shen, C.; Crassous, J. Chem. Sci. 2014, 5, 3680–3694. doi:10.1039/c4sc01404a
Return to citation in text: [1] [2] [3] [4] -
Shigeno, M.; Kushida, Y.; Yamaguchi, M. Chem. Commun. 2016, 52, 4955–4970. doi:10.1039/c5cc10379g
Return to citation in text: [1] [2] [3] [4] -
Isla, H.; Crassous, J. C. R. Chim. 2016, 19, 39–49. doi:10.1016/j.crci.2015.06.014
Return to citation in text: [1] [2] [3] [4] -
Chen, C.-F.; Shen, Y. Helicene Chemistry: From Synthesis to Applications; Springer: Berlin, Heidelberg, Germany, 2017. doi:10.1007/978-3-662-53168-6
Return to citation in text: [1] [2] [3] [4] [5] [6] [7] -
Hasan, M.; Borovkov, V. Symmetry 2018, 10, No. 10. doi:10.3390/sym10010010
Return to citation in text: [1] [2] [3] [4] [5] -
Rickhaus, M.; Mayor, M.; Juríček, M. Chem. Soc. Rev. 2016, 45, 1542–1556. doi:10.1039/c5cs00620a
Return to citation in text: [1] [2] [3] [4] -
Rulíšek, L.; Exner, O.; Cwiklik, L.; Jungwirth, P.; Starý, I.; Pospíšil, L.; Havlas, Z. J. Phys. Chem. C 2007, 111, 14948–14955. doi:10.1021/jp075129a
Return to citation in text: [1] -
Hrbac, J.; Storch, J.; Halouzka, V.; Cirkva, V.; Matejka, P.; Vacek, J. RSC Adv. 2014, 4, 46102–46105. doi:10.1039/c4ra06283c
Return to citation in text: [1] -
Vacek, J.; Vacek Chocholoušová, J.; Stará, I. G.; Starý, I.; Dubi, Y. Nanoscale 2015, 7, 8793–8802. doi:10.1039/c5nr01297j
Return to citation in text: [1] -
Fujikawa, T.; Mitoma, N.; Wakamiya, A.; Saeki, A.; Segawa, Y.; Itami, K. Org. Biomol. Chem. 2017, 15, 4697–4703. doi:10.1039/c7ob00987a
Return to citation in text: [1] -
Verbiest, T.; van Elshocht, S.; Kauranen, M.; Hellemans, L.; Snauwaert, J.; Nuckolls, C.; Katz, T. J.; Persoons, A. Science 1998, 282, 913–915. doi:10.1126/science.282.5390.913
Return to citation in text: [1] -
Coe, B. J.; Rusanova, D.; Joshi, V. D.; Sánchez, S.; Vávra, J.; Khobragade, D.; Severa, L.; Císařová, I.; Šaman, D.; Pohl, R.; Clays, K.; Depotter, G.; Brunschwig, B. S.; Teplý, F. J. Org. Chem. 2016, 81, 1912–1920. doi:10.1021/acs.joc.5b02692
Return to citation in text: [1] -
Phillips, K. E. S.; Katz, T. J.; Jockusch, S.; Lovinger, A. J.; Turro, N. J. J. Am. Chem. Soc. 2001, 123, 11899–11907. doi:10.1021/ja011706b
Return to citation in text: [1] -
Field, J. E.; Muller, G.; Riehl, J. P.; Venkataraman, D. J. Am. Chem. Soc. 2003, 125, 11808–11809. doi:10.1021/ja035626e
Return to citation in text: [1] -
Sawada, Y.; Furumi, S.; Takai, A.; Takeuchi, M.; Noguchi, K.; Tanaka, K. J. Am. Chem. Soc. 2012, 134, 4080–4083. doi:10.1021/ja300278e
Return to citation in text: [1] [2] -
Shyam Sundar, M.; Talele, H. R.; Mande, H. M.; Bedekar, A. V.; Tovar, R. C.; Muller, G. Tetrahedron Lett. 2014, 55, 1760–1764. doi:10.1016/j.tetlet.2014.01.108
Return to citation in text: [1] -
Nakamura, K.; Furumi, S.; Takeuchi, M.; Shibuya, T.; Tanaka, K. J. Am. Chem. Soc. 2014, 136, 5555–5558. doi:10.1021/ja500841f
Return to citation in text: [1] -
Yamamoto, Y.; Sakai, H.; Yuasa, J.; Araki, Y.; Wada, T.; Sakanoue, T.; Takenobu, T.; Kawai, T.; Hasobe, T. Chem. – Eur. J. 2016, 22, 4263–4273. doi:10.1002/chem.201504048
Return to citation in text: [1] -
Yamamoto, Y.; Sakai, H.; Yuasa, J.; Araki, Y.; Wada, T.; Sakanoue, T.; Takenobu, T.; Kawai, T.; Hasobe, T. J. Phys. Chem. C 2016, 120, 7421–7427. doi:10.1021/acs.jpcc.6b01123
Return to citation in text: [1] -
Ben Hassine, B.; Gorsane, M.; Pecher, J.; Martin, R. H. Bull. Soc. Chim. Belg. 1986, 95, 557–566. doi:10.1002/bscb.19860950706
Return to citation in text: [1] -
Reetz, M. T.; Beuttenmüller, E. W.; Goddard, R. Tetrahedron Lett. 1997, 38, 3211–3214. doi:10.1016/s0040-4039(97)00562-5
Return to citation in text: [1] -
Takenaka, N.; Sarangthem, R. S.; Captain, B. Angew. Chem., Int. Ed. 2008, 47, 9708–9710. doi:10.1002/anie.200803338
Return to citation in text: [1] -
Aillard, P.; Voituriez, A.; Marinetti, A. Dalton Trans. 2014, 43, 15263–15278. doi:10.1039/c4dt01935k
Return to citation in text: [1] -
Dova, D.; Viglianti, L.; Mussini, P. R.; Prager, S.; Dreuw, A.; Voituriez, A.; Licandro, E.; Cauteruccio, S. Asian J. Org. Chem. 2016, 5, 537–549. doi:10.1002/ajoc.201600025
Return to citation in text: [1] -
Fujikawa, T.; Segawa, Y.; Itami, K. J. Am. Chem. Soc. 2016, 138, 3587–3595. doi:10.1021/jacs.6b01303
Return to citation in text: [1] -
Huang, Q.; Jiang, L.; Liang, W.; Gui, J.; Xu, D.; Wu, W.; Nakai, Y.; Nishijima, M.; Fukuhara, G.; Mori, T.; Inoue, Y.; Yang, C. J. Org. Chem. 2016, 81, 3430–3434. doi:10.1021/acs.joc.6b00130
Return to citation in text: [1] -
Tounsi, M.; Ben Braiek, M.; Baraket, A.; Lee, M.; Zine, N.; Zabala, M.; Bausells, J.; Aloui, F.; Ben Hassine, B.; Maaref, A.; Errachid, A. Electroanalysis 2016, 28, 2892–2899. doi:10.1002/elan.201600104
Return to citation in text: [1] -
Xu, Y.; Zhang, Y. X.; Sugiyama, H.; Umano, T.; Osuga, H.; Tanaka, K. J. Am. Chem. Soc. 2004, 126, 6566–6567. doi:10.1021/ja0499748
Return to citation in text: [1] -
Passeri, R.; Aloisi, G. G.; Elisei, F.; Latterini, L.; Caronna, T.; Fontana, F.; Sora, I. N. Photochem. Photobiol. Sci. 2009, 8, 1574–1582. doi:10.1039/b9pp00015a
Return to citation in text: [1] -
Kelber, J.; Achard, M.-F.; Durola, F.; Bock, H. Angew. Chem., Int. Ed. 2012, 51, 5200–5203. doi:10.1002/anie.201108886
Return to citation in text: [1] -
Xiao, S.; Kang, S. J.; Wu, Y.; Ahn, S.; Kim, J. B.; Loo, Y.-L.; Siegrist, T.; Steigerwald, M. L.; Li, H.; Nuckolls, C. Chem. Sci. 2013, 4, 2018–2023. doi:10.1039/c3sc50374g
Return to citation in text: [1] -
Bédard, A.-C.; Vlassova, A.; Hernandez-Perez, A. C.; Bessette, A.; Hanan, G. S.; Heuft, M. A.; Collins, S. K. Chem. – Eur. J. 2013, 19, 16295–16302. doi:10.1002/chem.201301431
Return to citation in text: [1] -
Meisenheimer, J.; Witte, K. Ber. Dtsch. Chem. Ges. 1903, 36, 4153–4164. doi:10.1002/cber.19030360481
Return to citation in text: [1] -
Ben Braiek, M.; Aloui, F.; Moussa, S.; Tounsi, M.; Marrot, J.; Ben Hassine, B. Tetrahedron Lett. 2013, 54, 5421–5425. doi:10.1016/j.tetlet.2013.07.036
Return to citation in text: [1] [2] [3] -
Upadhyay, G. M.; Bedekar, A. V. Tetrahedron 2015, 71, 5644–5649. doi:10.1016/j.tet.2015.06.040
Return to citation in text: [1] [2] -
Al Mousawi, A.; Dumur, F.; Garra, P.; Toufaily, J.; Hamieh, T.; Goubard, F.; Bui, T.-T.; Graff, B.; Gigmes, D.; Pierre Fouassier, J.; Lalevée, J. J. Polym. Sci., Part A: Polym. Chem. 2017, 55, 1189–1199. doi:10.1002/pola.28476
Return to citation in text: [1] [2] [3] -
Hua, W.; Liu, Z.; Duan, L.; Dong, G.; Qiu, Y.; Zhang, B.; Cui, D.; Tao, X.; Cheng, N.; Liu, Y. RSC Adv. 2015, 5, 75–84. doi:10.1039/c4ra13486a
Return to citation in text: [1] [2] [3] [4] [5] [6] [7] [8] [9] -
Kötzner, L.; Webber, M. J.; Martínez, A.; DeFusco, C.; List, B. Angew. Chem., Int. Ed. 2014, 53, 5202–5205. doi:10.1002/anie.201400474
Return to citation in text: [1] [2] -
Luo, X.-Y.; Liu, Z.; Zhang, B.-J.; Hua, W.-M.; Feng, Y.; Li, L.; Zhang, D.-C.; Cui, D.-L. ChemistrySelect 2018, 3, 3426–3432. doi:10.1002/slct.201800267
Return to citation in text: [1] [2] -
Nakano, K.; Hidehira, Y.; Takahashi, K.; Hiyama, T.; Nozaki, K. Angew. Chem., Int. Ed. 2005, 44, 7136–7138. doi:10.1002/anie.200502855
Return to citation in text: [1] [2] -
Upadhyay, G. M.; Talele, H. R.; Bedekar, A. V. J. Org. Chem. 2016, 81, 7751–7759. doi:10.1021/acs.joc.6b01395
Return to citation in text: [1] -
Bucinskas, A.; Waghray, D.; Bagdziunas, G.; Thomas, J.; Grazulevicius, J. V.; Dehaen, W. J. Org. Chem. 2015, 80, 2521–2528. doi:10.1021/jo5024188
Return to citation in text: [1] [2] [3] -
Ben Braiek, M.; Aloui, F.; Ben Hassine, B. Tetrahedron Lett. 2016, 57, 2763–2766. doi:10.1016/j.tetlet.2016.05.030
Return to citation in text: [1] [2] -
Dreher, S. D.; Weix, D. J.; Katz, T. J. J. Org. Chem. 1999, 64, 3671–3678. doi:10.1021/jo990065o
Return to citation in text: [1] [2] -
Shi, L.; Liu, Z.; Dong, G.; Duan, L.; Qiu, Y.; Jia, J.; Guo, W.; Zhao, D.; Cui, D.; Tao, X. Chem. – Eur. J. 2012, 18, 8092–8099. doi:10.1002/chem.201200068
Return to citation in text: [1] [2] [3] -
Upadhyay, G. M.; Talele, H. R.; Sahoo, S.; Bedekar, A. V. Tetrahedron Lett. 2014, 55, 5394–5399. doi:10.1016/j.tetlet.2014.07.116
Return to citation in text: [1] [2] -
Jiang, H.; Sun, J.; Zhang, J. Curr. Org. Chem. 2012, 16, 2014–2025. doi:10.2174/138527212803251604
Return to citation in text: [1] -
Ziarani, G. M.; Moradi, R.; Lashgari, N.; Kruger, H. G. Carbazole Dyes. Metal-Free Synthetic Organic Dyes; Elsevier: Amsterdam, Netherlands, 2018; pp 109–116. doi:10.1016/b978-0-12-815647-6.00006-6
Return to citation in text: [1] -
D’Ischia, M.; Napolitano, A.; Pezzella, A. Pyrroles and their Benzo Derivatives: Applications. In Comprehensive Heterocyclic Chemistry III; Katritzky, A. R.; Ramsden, C. A.; Scriven, E. F. V.; Taylor, R. J. K., Eds.; Elsevier Science, 2008; Vol. 3, pp 353–388. doi:10.1016/b978-008044992-0.00304-7
Return to citation in text: [1] -
Chan, G. W.; Francis, T.; Thureen, D. R.; Offen, P. H.; Pierce, N. J.; Westley, J. W.; Johnson, R. K.; Faulkner, D. J. J. Org. Chem. 1993, 58, 2544–2546. doi:10.1021/jo00061a031
Return to citation in text: [1] -
Hamasaki, A.; Zimpleman, J. M.; Hwang, I.; Boger, D. L. J. Am. Chem. Soc. 2005, 127, 10767–10770. doi:10.1021/ja0526416
Return to citation in text: [1] -
Baryshnikov, G. V.; Gawrys, P.; Ivaniuk, K.; Witulski, B.; Whitby, R. J.; Al-Muhammad, A.; Minaev, B.; Cherpak, V.; Stakhira, P.; Volyniuk, D.; Wiosna-Salyga, G.; Luszczynska, B.; Lazauskas, A.; Tamulevicius, S.; Grazulevicius, J. V. J. Mater. Chem. C 2016, 4, 5795–5805. doi:10.1039/c6tc01469k
Return to citation in text: [1] -
Weimar, M.; Correa da Costa, R.; Lee, F.-H.; Fuchter, M. J. Org. Lett. 2013, 15, 1706–1709. doi:10.1021/ol400493x
Return to citation in text: [1] -
Carreras, J.; Patil, M.; Thiel, W.; Alcarazo, M. J. Am. Chem. Soc. 2012, 134, 16753–16758. doi:10.1021/ja306947m
Return to citation in text: [1] -
Frigoli, M.; Marrot, J.; Gentili, P. L.; Jacquemin, D.; Vagnini, M.; Pannacci, D.; Ortica, F. ChemPhysChem 2015, 16, 2447–2458. doi:10.1002/cphc.201500251
Return to citation in text: [1] -
Li, Y.; Waser, J. Angew. Chem., Int. Ed. 2015, 54, 5438–5442. doi:10.1002/anie.201412321
Return to citation in text: [1] -
Oyama, H.; Nakano, K.; Harada, T.; Kuroda, R.; Naito, M.; Nobusawa, K.; Nozaki, K. Org. Lett. 2013, 15, 2104–2107. doi:10.1021/ol4005036
Return to citation in text: [1] -
Hirano, K.; Inaba, Y.; Takasu, K.; Oishi, S.; Takemoto, Y.; Fujii, N.; Ohno, H. J. Org. Chem. 2011, 76, 9068–9080. doi:10.1021/jo2018119
Return to citation in text: [1] -
Yao, T.; Campo, M. A.; Larock, R. C. J. Org. Chem. 2005, 70, 3511–3517. doi:10.1021/jo050104y
Return to citation in text: [1] -
Mohamed, R. K.; Mondal, S.; Guerrera, J. V.; Eaton, T. M.; Albrecht-Schmitt, T. E.; Shatruk, M.; Alabugin, I. V. Angew. Chem., Int. Ed. 2016, 55, 12054–12058. doi:10.1002/anie.201606330
Return to citation in text: [1] -
Storch, J.; Čermák, J.; Karban, J.; Císařová, I.; Sýkora, J. J. Org. Chem. 2010, 75, 3137–3140. doi:10.1021/jo100252a
Return to citation in text: [1] -
Mandadapu, A. K.; Dathi, M. D.; Arigela, R. K.; Kundu, B. Tetrahedron 2012, 68, 8207–8215. doi:10.1016/j.tet.2012.07.067
Return to citation in text: [1] -
Kumar, K. S.; Bhaskar, B.; Ramulu, M. S.; Kumar, N. P.; Ashfaq, M. A.; Pal, M. Org. Biomol. Chem. 2017, 15, 82–87. doi:10.1039/c6ob02340a
Return to citation in text: [1] -
Pati, K.; Michas, C.; Allenger, D.; Piskun, I.; Coutros, P. S.; dos Passos Gomes, G.; Alabugin, I. V. J. Org. Chem. 2015, 80, 11706–11717. doi:10.1021/acs.joc.5b01014
Return to citation in text: [1] -
Gulevskaya, A. V.; Shvydkova, E. A.; Tonkoglazova, D. I. Eur. J. Org. Chem. 2018, 5030–5043. doi:10.1002/ejoc.201800613
Return to citation in text: [1] [2] [3] [4] -
Gulevskaya, A. V. Eur. J. Org. Chem. 2016, 4207–4214. doi:10.1002/ejoc.201600660
Return to citation in text: [1] -
Gulevskaya, A. V.; Tonkoglazova, D. I.; Guchunov, A. S.; Misharev, A. D. Eur. J. Org. Chem. 2019, 4879–4890. doi:10.1002/ejoc.201900818
Return to citation in text: [1] [2] [3] [4]
| 70. | Gulevskaya, A. V.; Shvydkova, E. A.; Tonkoglazova, D. I. Eur. J. Org. Chem. 2018, 5030–5043. doi:10.1002/ejoc.201800613 |
| 70. | Gulevskaya, A. V.; Shvydkova, E. A.; Tonkoglazova, D. I. Eur. J. Org. Chem. 2018, 5030–5043. doi:10.1002/ejoc.201800613 |
| 71. | Gulevskaya, A. V. Eur. J. Org. Chem. 2016, 4207–4214. doi:10.1002/ejoc.201600660 |
| 72. | Gulevskaya, A. V.; Tonkoglazova, D. I.; Guchunov, A. S.; Misharev, A. D. Eur. J. Org. Chem. 2019, 4879–4890. doi:10.1002/ejoc.201900818 |
| 1. | Shen, Y.; Chen, C.-F. Chem. Rev. 2012, 112, 1463–1535. doi:10.1021/cr200087r |
| 2. | Gingras, M. Chem. Soc. Rev. 2013, 42, 968–1006. doi:10.1039/c2cs35154d |
| 3. | Gingras, M.; Félix, G.; Peresutti, R. Chem. Soc. Rev. 2013, 42, 1007–1050. doi:10.1039/c2cs35111k |
| 4. | Gingras, M. Chem. Soc. Rev. 2013, 42, 1051–1095. doi:10.1039/c2cs35134j |
| 5. | Dumitrascu, F.; Dumitrescu, D. G.; Aron, I. ARKIVOC 2009, No. 1, 1–32. doi:10.3998/ark.5550190.0011.101 |
| 6. | Saleh, N.; Shen, C.; Crassous, J. Chem. Sci. 2014, 5, 3680–3694. doi:10.1039/c4sc01404a |
| 7. | Shigeno, M.; Kushida, Y.; Yamaguchi, M. Chem. Commun. 2016, 52, 4955–4970. doi:10.1039/c5cc10379g |
| 8. | Isla, H.; Crassous, J. C. R. Chim. 2016, 19, 39–49. doi:10.1016/j.crci.2015.06.014 |
| 9. | Chen, C.-F.; Shen, Y. Helicene Chemistry: From Synthesis to Applications; Springer: Berlin, Heidelberg, Germany, 2017. doi:10.1007/978-3-662-53168-6 |
| 10. | Hasan, M.; Borovkov, V. Symmetry 2018, 10, No. 10. doi:10.3390/sym10010010 |
| 11. | Rickhaus, M.; Mayor, M.; Juríček, M. Chem. Soc. Rev. 2016, 45, 1542–1556. doi:10.1039/c5cs00620a |
| 18. | Phillips, K. E. S.; Katz, T. J.; Jockusch, S.; Lovinger, A. J.; Turro, N. J. J. Am. Chem. Soc. 2001, 123, 11899–11907. doi:10.1021/ja011706b |
| 19. | Field, J. E.; Muller, G.; Riehl, J. P.; Venkataraman, D. J. Am. Chem. Soc. 2003, 125, 11808–11809. doi:10.1021/ja035626e |
| 20. | Sawada, Y.; Furumi, S.; Takai, A.; Takeuchi, M.; Noguchi, K.; Tanaka, K. J. Am. Chem. Soc. 2012, 134, 4080–4083. doi:10.1021/ja300278e |
| 21. | Shyam Sundar, M.; Talele, H. R.; Mande, H. M.; Bedekar, A. V.; Tovar, R. C.; Muller, G. Tetrahedron Lett. 2014, 55, 1760–1764. doi:10.1016/j.tetlet.2014.01.108 |
| 22. | Nakamura, K.; Furumi, S.; Takeuchi, M.; Shibuya, T.; Tanaka, K. J. Am. Chem. Soc. 2014, 136, 5555–5558. doi:10.1021/ja500841f |
| 23. | Yamamoto, Y.; Sakai, H.; Yuasa, J.; Araki, Y.; Wada, T.; Sakanoue, T.; Takenobu, T.; Kawai, T.; Hasobe, T. Chem. – Eur. J. 2016, 22, 4263–4273. doi:10.1002/chem.201504048 |
| 24. | Yamamoto, Y.; Sakai, H.; Yuasa, J.; Araki, Y.; Wada, T.; Sakanoue, T.; Takenobu, T.; Kawai, T.; Hasobe, T. J. Phys. Chem. C 2016, 120, 7421–7427. doi:10.1021/acs.jpcc.6b01123 |
| 42. | Hua, W.; Liu, Z.; Duan, L.; Dong, G.; Qiu, Y.; Zhang, B.; Cui, D.; Tao, X.; Cheng, N.; Liu, Y. RSC Adv. 2015, 5, 75–84. doi:10.1039/c4ra13486a |
| 42. | Hua, W.; Liu, Z.; Duan, L.; Dong, G.; Qiu, Y.; Zhang, B.; Cui, D.; Tao, X.; Cheng, N.; Liu, Y. RSC Adv. 2015, 5, 75–84. doi:10.1039/c4ra13486a |
| 16. | Verbiest, T.; van Elshocht, S.; Kauranen, M.; Hellemans, L.; Snauwaert, J.; Nuckolls, C.; Katz, T. J.; Persoons, A. Science 1998, 282, 913–915. doi:10.1126/science.282.5390.913 |
| 17. | Coe, B. J.; Rusanova, D.; Joshi, V. D.; Sánchez, S.; Vávra, J.; Khobragade, D.; Severa, L.; Císařová, I.; Šaman, D.; Pohl, R.; Clays, K.; Depotter, G.; Brunschwig, B. S.; Teplý, F. J. Org. Chem. 2016, 81, 1912–1920. doi:10.1021/acs.joc.5b02692 |
| 50. | Shi, L.; Liu, Z.; Dong, G.; Duan, L.; Qiu, Y.; Jia, J.; Guo, W.; Zhao, D.; Cui, D.; Tao, X. Chem. – Eur. J. 2012, 18, 8092–8099. doi:10.1002/chem.201200068 |
| 42. | Hua, W.; Liu, Z.; Duan, L.; Dong, G.; Qiu, Y.; Zhang, B.; Cui, D.; Tao, X.; Cheng, N.; Liu, Y. RSC Adv. 2015, 5, 75–84. doi:10.1039/c4ra13486a |
| 12. | Rulíšek, L.; Exner, O.; Cwiklik, L.; Jungwirth, P.; Starý, I.; Pospíšil, L.; Havlas, Z. J. Phys. Chem. C 2007, 111, 14948–14955. doi:10.1021/jp075129a |
| 13. | Hrbac, J.; Storch, J.; Halouzka, V.; Cirkva, V.; Matejka, P.; Vacek, J. RSC Adv. 2014, 4, 46102–46105. doi:10.1039/c4ra06283c |
| 14. | Vacek, J.; Vacek Chocholoušová, J.; Stará, I. G.; Starý, I.; Dubi, Y. Nanoscale 2015, 7, 8793–8802. doi:10.1039/c5nr01297j |
| 15. | Fujikawa, T.; Mitoma, N.; Wakamiya, A.; Saeki, A.; Segawa, Y.; Itami, K. Org. Biomol. Chem. 2017, 15, 4697–4703. doi:10.1039/c7ob00987a |
| 38. | Meisenheimer, J.; Witte, K. Ber. Dtsch. Chem. Ges. 1903, 36, 4153–4164. doi:10.1002/cber.19030360481 |
| 39. | Ben Braiek, M.; Aloui, F.; Moussa, S.; Tounsi, M.; Marrot, J.; Ben Hassine, B. Tetrahedron Lett. 2013, 54, 5421–5425. doi:10.1016/j.tetlet.2013.07.036 |
| 40. | Upadhyay, G. M.; Bedekar, A. V. Tetrahedron 2015, 71, 5644–5649. doi:10.1016/j.tet.2015.06.040 |
| 41. | Al Mousawi, A.; Dumur, F.; Garra, P.; Toufaily, J.; Hamieh, T.; Goubard, F.; Bui, T.-T.; Graff, B.; Gigmes, D.; Pierre Fouassier, J.; Lalevée, J. J. Polym. Sci., Part A: Polym. Chem. 2017, 55, 1189–1199. doi:10.1002/pola.28476 |
| 42. | Hua, W.; Liu, Z.; Duan, L.; Dong, G.; Qiu, Y.; Zhang, B.; Cui, D.; Tao, X.; Cheng, N.; Liu, Y. RSC Adv. 2015, 5, 75–84. doi:10.1039/c4ra13486a |
| 43. | Kötzner, L.; Webber, M. J.; Martínez, A.; DeFusco, C.; List, B. Angew. Chem., Int. Ed. 2014, 53, 5202–5205. doi:10.1002/anie.201400474 |
| 44. | Luo, X.-Y.; Liu, Z.; Zhang, B.-J.; Hua, W.-M.; Feng, Y.; Li, L.; Zhang, D.-C.; Cui, D.-L. ChemistrySelect 2018, 3, 3426–3432. doi:10.1002/slct.201800267 |
| 45. | Nakano, K.; Hidehira, Y.; Takahashi, K.; Hiyama, T.; Nozaki, K. Angew. Chem., Int. Ed. 2005, 44, 7136–7138. doi:10.1002/anie.200502855 |
| 46. | Upadhyay, G. M.; Talele, H. R.; Bedekar, A. V. J. Org. Chem. 2016, 81, 7751–7759. doi:10.1021/acs.joc.6b01395 |
| 47. | Bucinskas, A.; Waghray, D.; Bagdziunas, G.; Thomas, J.; Grazulevicius, J. V.; Dehaen, W. J. Org. Chem. 2015, 80, 2521–2528. doi:10.1021/jo5024188 |
| 48. | Ben Braiek, M.; Aloui, F.; Ben Hassine, B. Tetrahedron Lett. 2016, 57, 2763–2766. doi:10.1016/j.tetlet.2016.05.030 |
| 49. | Dreher, S. D.; Weix, D. J.; Katz, T. J. J. Org. Chem. 1999, 64, 3671–3678. doi:10.1021/jo990065o |
| 50. | Shi, L.; Liu, Z.; Dong, G.; Duan, L.; Qiu, Y.; Jia, J.; Guo, W.; Zhao, D.; Cui, D.; Tao, X. Chem. – Eur. J. 2012, 18, 8092–8099. doi:10.1002/chem.201200068 |
| 51. | Upadhyay, G. M.; Talele, H. R.; Sahoo, S.; Bedekar, A. V. Tetrahedron Lett. 2014, 55, 5394–5399. doi:10.1016/j.tetlet.2014.07.116 |
| 9. | Chen, C.-F.; Shen, Y. Helicene Chemistry: From Synthesis to Applications; Springer: Berlin, Heidelberg, Germany, 2017. doi:10.1007/978-3-662-53168-6 |
| 10. | Hasan, M.; Borovkov, V. Symmetry 2018, 10, No. 10. doi:10.3390/sym10010010 |
| 9. | Chen, C.-F.; Shen, Y. Helicene Chemistry: From Synthesis to Applications; Springer: Berlin, Heidelberg, Germany, 2017. doi:10.1007/978-3-662-53168-6 |
| 52. | Jiang, H.; Sun, J.; Zhang, J. Curr. Org. Chem. 2012, 16, 2014–2025. doi:10.2174/138527212803251604 |
| 53. | Ziarani, G. M.; Moradi, R.; Lashgari, N.; Kruger, H. G. Carbazole Dyes. Metal-Free Synthetic Organic Dyes; Elsevier: Amsterdam, Netherlands, 2018; pp 109–116. doi:10.1016/b978-0-12-815647-6.00006-6 |
| 54. | D’Ischia, M.; Napolitano, A.; Pezzella, A. Pyrroles and their Benzo Derivatives: Applications. In Comprehensive Heterocyclic Chemistry III; Katritzky, A. R.; Ramsden, C. A.; Scriven, E. F. V.; Taylor, R. J. K., Eds.; Elsevier Science, 2008; Vol. 3, pp 353–388. doi:10.1016/b978-008044992-0.00304-7 |
| 42. | Hua, W.; Liu, Z.; Duan, L.; Dong, G.; Qiu, Y.; Zhang, B.; Cui, D.; Tao, X.; Cheng, N.; Liu, Y. RSC Adv. 2015, 5, 75–84. doi:10.1039/c4ra13486a |
| 32. | Tounsi, M.; Ben Braiek, M.; Baraket, A.; Lee, M.; Zine, N.; Zabala, M.; Bausells, J.; Aloui, F.; Ben Hassine, B.; Maaref, A.; Errachid, A. Electroanalysis 2016, 28, 2892–2899. doi:10.1002/elan.201600104 |
| 1. | Shen, Y.; Chen, C.-F. Chem. Rev. 2012, 112, 1463–1535. doi:10.1021/cr200087r |
| 2. | Gingras, M. Chem. Soc. Rev. 2013, 42, 968–1006. doi:10.1039/c2cs35154d |
| 3. | Gingras, M.; Félix, G.; Peresutti, R. Chem. Soc. Rev. 2013, 42, 1007–1050. doi:10.1039/c2cs35111k |
| 4. | Gingras, M. Chem. Soc. Rev. 2013, 42, 1051–1095. doi:10.1039/c2cs35134j |
| 5. | Dumitrascu, F.; Dumitrescu, D. G.; Aron, I. ARKIVOC 2009, No. 1, 1–32. doi:10.3998/ark.5550190.0011.101 |
| 6. | Saleh, N.; Shen, C.; Crassous, J. Chem. Sci. 2014, 5, 3680–3694. doi:10.1039/c4sc01404a |
| 7. | Shigeno, M.; Kushida, Y.; Yamaguchi, M. Chem. Commun. 2016, 52, 4955–4970. doi:10.1039/c5cc10379g |
| 8. | Isla, H.; Crassous, J. C. R. Chim. 2016, 19, 39–49. doi:10.1016/j.crci.2015.06.014 |
| 9. | Chen, C.-F.; Shen, Y. Helicene Chemistry: From Synthesis to Applications; Springer: Berlin, Heidelberg, Germany, 2017. doi:10.1007/978-3-662-53168-6 |
| 10. | Hasan, M.; Borovkov, V. Symmetry 2018, 10, No. 10. doi:10.3390/sym10010010 |
| 11. | Rickhaus, M.; Mayor, M.; Juríček, M. Chem. Soc. Rev. 2016, 45, 1542–1556. doi:10.1039/c5cs00620a |
| 72. | Gulevskaya, A. V.; Tonkoglazova, D. I.; Guchunov, A. S.; Misharev, A. D. Eur. J. Org. Chem. 2019, 4879–4890. doi:10.1002/ejoc.201900818 |
| 31. | Huang, Q.; Jiang, L.; Liang, W.; Gui, J.; Xu, D.; Wu, W.; Nakai, Y.; Nishijima, M.; Fukuhara, G.; Mori, T.; Inoue, Y.; Yang, C. J. Org. Chem. 2016, 81, 3430–3434. doi:10.1021/acs.joc.6b00130 |
| 1. | Shen, Y.; Chen, C.-F. Chem. Rev. 2012, 112, 1463–1535. doi:10.1021/cr200087r |
| 2. | Gingras, M. Chem. Soc. Rev. 2013, 42, 968–1006. doi:10.1039/c2cs35154d |
| 3. | Gingras, M.; Félix, G.; Peresutti, R. Chem. Soc. Rev. 2013, 42, 1007–1050. doi:10.1039/c2cs35111k |
| 4. | Gingras, M. Chem. Soc. Rev. 2013, 42, 1051–1095. doi:10.1039/c2cs35134j |
| 5. | Dumitrascu, F.; Dumitrescu, D. G.; Aron, I. ARKIVOC 2009, No. 1, 1–32. doi:10.3998/ark.5550190.0011.101 |
| 6. | Saleh, N.; Shen, C.; Crassous, J. Chem. Sci. 2014, 5, 3680–3694. doi:10.1039/c4sc01404a |
| 7. | Shigeno, M.; Kushida, Y.; Yamaguchi, M. Chem. Commun. 2016, 52, 4955–4970. doi:10.1039/c5cc10379g |
| 8. | Isla, H.; Crassous, J. C. R. Chim. 2016, 19, 39–49. doi:10.1016/j.crci.2015.06.014 |
| 9. | Chen, C.-F.; Shen, Y. Helicene Chemistry: From Synthesis to Applications; Springer: Berlin, Heidelberg, Germany, 2017. doi:10.1007/978-3-662-53168-6 |
| 10. | Hasan, M.; Borovkov, V. Symmetry 2018, 10, No. 10. doi:10.3390/sym10010010 |
| 11. | Rickhaus, M.; Mayor, M.; Juríček, M. Chem. Soc. Rev. 2016, 45, 1542–1556. doi:10.1039/c5cs00620a |
| 20. | Sawada, Y.; Furumi, S.; Takai, A.; Takeuchi, M.; Noguchi, K.; Tanaka, K. J. Am. Chem. Soc. 2012, 134, 4080–4083. doi:10.1021/ja300278e |
| 35. | Kelber, J.; Achard, M.-F.; Durola, F.; Bock, H. Angew. Chem., Int. Ed. 2012, 51, 5200–5203. doi:10.1002/anie.201108886 |
| 36. | Xiao, S.; Kang, S. J.; Wu, Y.; Ahn, S.; Kim, J. B.; Loo, Y.-L.; Siegrist, T.; Steigerwald, M. L.; Li, H.; Nuckolls, C. Chem. Sci. 2013, 4, 2018–2023. doi:10.1039/c3sc50374g |
| 37. | Bédard, A.-C.; Vlassova, A.; Hernandez-Perez, A. C.; Bessette, A.; Hanan, G. S.; Heuft, M. A.; Collins, S. K. Chem. – Eur. J. 2013, 19, 16295–16302. doi:10.1002/chem.201301431 |
| 72. | Gulevskaya, A. V.; Tonkoglazova, D. I.; Guchunov, A. S.; Misharev, A. D. Eur. J. Org. Chem. 2019, 4879–4890. doi:10.1002/ejoc.201900818 |
| 30. | Fujikawa, T.; Segawa, Y.; Itami, K. J. Am. Chem. Soc. 2016, 138, 3587–3595. doi:10.1021/jacs.6b01303 |
| 70. | Gulevskaya, A. V.; Shvydkova, E. A.; Tonkoglazova, D. I. Eur. J. Org. Chem. 2018, 5030–5043. doi:10.1002/ejoc.201800613 |
| 25. | Ben Hassine, B.; Gorsane, M.; Pecher, J.; Martin, R. H. Bull. Soc. Chim. Belg. 1986, 95, 557–566. doi:10.1002/bscb.19860950706 |
| 26. | Reetz, M. T.; Beuttenmüller, E. W.; Goddard, R. Tetrahedron Lett. 1997, 38, 3211–3214. doi:10.1016/s0040-4039(97)00562-5 |
| 27. | Takenaka, N.; Sarangthem, R. S.; Captain, B. Angew. Chem., Int. Ed. 2008, 47, 9708–9710. doi:10.1002/anie.200803338 |
| 28. | Aillard, P.; Voituriez, A.; Marinetti, A. Dalton Trans. 2014, 43, 15263–15278. doi:10.1039/c4dt01935k |
| 29. | Dova, D.; Viglianti, L.; Mussini, P. R.; Prager, S.; Dreuw, A.; Voituriez, A.; Licandro, E.; Cauteruccio, S. Asian J. Org. Chem. 2016, 5, 537–549. doi:10.1002/ajoc.201600025 |
| 33. | Xu, Y.; Zhang, Y. X.; Sugiyama, H.; Umano, T.; Osuga, H.; Tanaka, K. J. Am. Chem. Soc. 2004, 126, 6566–6567. doi:10.1021/ja0499748 |
| 34. | Passeri, R.; Aloisi, G. G.; Elisei, F.; Latterini, L.; Caronna, T.; Fontana, F.; Sora, I. N. Photochem. Photobiol. Sci. 2009, 8, 1574–1582. doi:10.1039/b9pp00015a |
| 70. | Gulevskaya, A. V.; Shvydkova, E. A.; Tonkoglazova, D. I. Eur. J. Org. Chem. 2018, 5030–5043. doi:10.1002/ejoc.201800613 |
| 47. | Bucinskas, A.; Waghray, D.; Bagdziunas, G.; Thomas, J.; Grazulevicius, J. V.; Dehaen, W. J. Org. Chem. 2015, 80, 2521–2528. doi:10.1021/jo5024188 |
| 41. | Al Mousawi, A.; Dumur, F.; Garra, P.; Toufaily, J.; Hamieh, T.; Goubard, F.; Bui, T.-T.; Graff, B.; Gigmes, D.; Pierre Fouassier, J.; Lalevée, J. J. Polym. Sci., Part A: Polym. Chem. 2017, 55, 1189–1199. doi:10.1002/pola.28476 |
| 72. | Gulevskaya, A. V.; Tonkoglazova, D. I.; Guchunov, A. S.; Misharev, A. D. Eur. J. Org. Chem. 2019, 4879–4890. doi:10.1002/ejoc.201900818 |
| 39. | Ben Braiek, M.; Aloui, F.; Moussa, S.; Tounsi, M.; Marrot, J.; Ben Hassine, B. Tetrahedron Lett. 2013, 54, 5421–5425. doi:10.1016/j.tetlet.2013.07.036 |
| 9. | Chen, C.-F.; Shen, Y. Helicene Chemistry: From Synthesis to Applications; Springer: Berlin, Heidelberg, Germany, 2017. doi:10.1007/978-3-662-53168-6 |
| 42. | Hua, W.; Liu, Z.; Duan, L.; Dong, G.; Qiu, Y.; Zhang, B.; Cui, D.; Tao, X.; Cheng, N.; Liu, Y. RSC Adv. 2015, 5, 75–84. doi:10.1039/c4ra13486a |
| 43. | Kötzner, L.; Webber, M. J.; Martínez, A.; DeFusco, C.; List, B. Angew. Chem., Int. Ed. 2014, 53, 5202–5205. doi:10.1002/anie.201400474 |
| 57. | Baryshnikov, G. V.; Gawrys, P.; Ivaniuk, K.; Witulski, B.; Whitby, R. J.; Al-Muhammad, A.; Minaev, B.; Cherpak, V.; Stakhira, P.; Volyniuk, D.; Wiosna-Salyga, G.; Luszczynska, B.; Lazauskas, A.; Tamulevicius, S.; Grazulevicius, J. V. J. Mater. Chem. C 2016, 4, 5795–5805. doi:10.1039/c6tc01469k |
| 58. | Weimar, M.; Correa da Costa, R.; Lee, F.-H.; Fuchter, M. J. Org. Lett. 2013, 15, 1706–1709. doi:10.1021/ol400493x |
| 59. | Carreras, J.; Patil, M.; Thiel, W.; Alcarazo, M. J. Am. Chem. Soc. 2012, 134, 16753–16758. doi:10.1021/ja306947m |
| 60. | Frigoli, M.; Marrot, J.; Gentili, P. L.; Jacquemin, D.; Vagnini, M.; Pannacci, D.; Ortica, F. ChemPhysChem 2015, 16, 2447–2458. doi:10.1002/cphc.201500251 |
| 61. | Li, Y.; Waser, J. Angew. Chem., Int. Ed. 2015, 54, 5438–5442. doi:10.1002/anie.201412321 |
| 62. | Oyama, H.; Nakano, K.; Harada, T.; Kuroda, R.; Naito, M.; Nobusawa, K.; Nozaki, K. Org. Lett. 2013, 15, 2104–2107. doi:10.1021/ol4005036 |
| 63. | Hirano, K.; Inaba, Y.; Takasu, K.; Oishi, S.; Takemoto, Y.; Fujii, N.; Ohno, H. J. Org. Chem. 2011, 76, 9068–9080. doi:10.1021/jo2018119 |
| 64. | Yao, T.; Campo, M. A.; Larock, R. C. J. Org. Chem. 2005, 70, 3511–3517. doi:10.1021/jo050104y |
| 65. | Mohamed, R. K.; Mondal, S.; Guerrera, J. V.; Eaton, T. M.; Albrecht-Schmitt, T. E.; Shatruk, M.; Alabugin, I. V. Angew. Chem., Int. Ed. 2016, 55, 12054–12058. doi:10.1002/anie.201606330 |
| 66. | Storch, J.; Čermák, J.; Karban, J.; Císařová, I.; Sýkora, J. J. Org. Chem. 2010, 75, 3137–3140. doi:10.1021/jo100252a |
| 67. | Mandadapu, A. K.; Dathi, M. D.; Arigela, R. K.; Kundu, B. Tetrahedron 2012, 68, 8207–8215. doi:10.1016/j.tet.2012.07.067 |
| 68. | Kumar, K. S.; Bhaskar, B.; Ramulu, M. S.; Kumar, N. P.; Ashfaq, M. A.; Pal, M. Org. Biomol. Chem. 2017, 15, 82–87. doi:10.1039/c6ob02340a |
| 69. | Pati, K.; Michas, C.; Allenger, D.; Piskun, I.; Coutros, P. S.; dos Passos Gomes, G.; Alabugin, I. V. J. Org. Chem. 2015, 80, 11706–11717. doi:10.1021/acs.joc.5b01014 |
| 49. | Dreher, S. D.; Weix, D. J.; Katz, T. J. J. Org. Chem. 1999, 64, 3671–3678. doi:10.1021/jo990065o |
| 45. | Nakano, K.; Hidehira, Y.; Takahashi, K.; Hiyama, T.; Nozaki, K. Angew. Chem., Int. Ed. 2005, 44, 7136–7138. doi:10.1002/anie.200502855 |
| 1. | Shen, Y.; Chen, C.-F. Chem. Rev. 2012, 112, 1463–1535. doi:10.1021/cr200087r |
| 2. | Gingras, M. Chem. Soc. Rev. 2013, 42, 968–1006. doi:10.1039/c2cs35154d |
| 3. | Gingras, M.; Félix, G.; Peresutti, R. Chem. Soc. Rev. 2013, 42, 1007–1050. doi:10.1039/c2cs35111k |
| 4. | Gingras, M. Chem. Soc. Rev. 2013, 42, 1051–1095. doi:10.1039/c2cs35134j |
| 5. | Dumitrascu, F.; Dumitrescu, D. G.; Aron, I. ARKIVOC 2009, No. 1, 1–32. doi:10.3998/ark.5550190.0011.101 |
| 6. | Saleh, N.; Shen, C.; Crassous, J. Chem. Sci. 2014, 5, 3680–3694. doi:10.1039/c4sc01404a |
| 7. | Shigeno, M.; Kushida, Y.; Yamaguchi, M. Chem. Commun. 2016, 52, 4955–4970. doi:10.1039/c5cc10379g |
| 8. | Isla, H.; Crassous, J. C. R. Chim. 2016, 19, 39–49. doi:10.1016/j.crci.2015.06.014 |
| 9. | Chen, C.-F.; Shen, Y. Helicene Chemistry: From Synthesis to Applications; Springer: Berlin, Heidelberg, Germany, 2017. doi:10.1007/978-3-662-53168-6 |
| 10. | Hasan, M.; Borovkov, V. Symmetry 2018, 10, No. 10. doi:10.3390/sym10010010 |
| 11. | Rickhaus, M.; Mayor, M.; Juríček, M. Chem. Soc. Rev. 2016, 45, 1542–1556. doi:10.1039/c5cs00620a |
| 39. | Ben Braiek, M.; Aloui, F.; Moussa, S.; Tounsi, M.; Marrot, J.; Ben Hassine, B. Tetrahedron Lett. 2013, 54, 5421–5425. doi:10.1016/j.tetlet.2013.07.036 |
| 40. | Upadhyay, G. M.; Bedekar, A. V. Tetrahedron 2015, 71, 5644–5649. doi:10.1016/j.tet.2015.06.040 |
| 41. | Al Mousawi, A.; Dumur, F.; Garra, P.; Toufaily, J.; Hamieh, T.; Goubard, F.; Bui, T.-T.; Graff, B.; Gigmes, D.; Pierre Fouassier, J.; Lalevée, J. J. Polym. Sci., Part A: Polym. Chem. 2017, 55, 1189–1199. doi:10.1002/pola.28476 |
| 42. | Hua, W.; Liu, Z.; Duan, L.; Dong, G.; Qiu, Y.; Zhang, B.; Cui, D.; Tao, X.; Cheng, N.; Liu, Y. RSC Adv. 2015, 5, 75–84. doi:10.1039/c4ra13486a |
| 44. | Luo, X.-Y.; Liu, Z.; Zhang, B.-J.; Hua, W.-M.; Feng, Y.; Li, L.; Zhang, D.-C.; Cui, D.-L. ChemistrySelect 2018, 3, 3426–3432. doi:10.1002/slct.201800267 |
| 47. | Bucinskas, A.; Waghray, D.; Bagdziunas, G.; Thomas, J.; Grazulevicius, J. V.; Dehaen, W. J. Org. Chem. 2015, 80, 2521–2528. doi:10.1021/jo5024188 |
| 48. | Ben Braiek, M.; Aloui, F.; Ben Hassine, B. Tetrahedron Lett. 2016, 57, 2763–2766. doi:10.1016/j.tetlet.2016.05.030 |
| 50. | Shi, L.; Liu, Z.; Dong, G.; Duan, L.; Qiu, Y.; Jia, J.; Guo, W.; Zhao, D.; Cui, D.; Tao, X. Chem. – Eur. J. 2012, 18, 8092–8099. doi:10.1002/chem.201200068 |
| 51. | Upadhyay, G. M.; Talele, H. R.; Sahoo, S.; Bedekar, A. V. Tetrahedron Lett. 2014, 55, 5394–5399. doi:10.1016/j.tetlet.2014.07.116 |
| 55. | Chan, G. W.; Francis, T.; Thureen, D. R.; Offen, P. H.; Pierce, N. J.; Westley, J. W.; Johnson, R. K.; Faulkner, D. J. J. Org. Chem. 1993, 58, 2544–2546. doi:10.1021/jo00061a031 |
| 42. | Hua, W.; Liu, Z.; Duan, L.; Dong, G.; Qiu, Y.; Zhang, B.; Cui, D.; Tao, X.; Cheng, N.; Liu, Y. RSC Adv. 2015, 5, 75–84. doi:10.1039/c4ra13486a |
| 56. | Hamasaki, A.; Zimpleman, J. M.; Hwang, I.; Boger, D. L. J. Am. Chem. Soc. 2005, 127, 10767–10770. doi:10.1021/ja0526416 |
| 42. | Hua, W.; Liu, Z.; Duan, L.; Dong, G.; Qiu, Y.; Zhang, B.; Cui, D.; Tao, X.; Cheng, N.; Liu, Y. RSC Adv. 2015, 5, 75–84. doi:10.1039/c4ra13486a |
© 2021 Tonkoglazova et al.; licensee Beilstein-Institut.
This is an Open Access article under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0). Please note that the reuse, redistribution and reproduction in particular requires that the author(s) and source are credited and that individual graphics may be subject to special legal provisions.
The license is subject to the Beilstein Journal of Organic Chemistry terms and conditions: (https://www.beilstein-journals.org/bjoc/terms)