Abstract
The reaction mechanism for the chlorination and bromination of 2-naphthol with PIDA or PIFA and AlX3 (X = Cl, Br), previously reported by our group, was elucidated via quantum chemical calculations using density functional theory. The chlorination mechanism using PIFA and AlCl3 demonstrated a better experimental and theoretical yield compared to using PIDA. Additionally, the lowest-energy chlorinating species was characterized by an equilibrium of Cl–I(Ph)–OTFA–AlCl3 and [Cl–I(Ph)][OTFA–AlCl3], rather than PhICl2 being the active species. On the other hand, bromination using PIDA and AlBr3 was more efficient, wherein the intermediate Br–I(Ph)–OAc–AlBr3 was formed as active brominating species. Similarly, PhIBr2 was higher in energy than our proposed species. The reaction mechanisms are described in detail in this work and were found to be in excellent agreement with the experimental yield. These initial results confirmed that our proposed mechanism was energetically favored and therefore more plausible compared to halogenation via PhIX2.
Graphical Abstract
Introduction
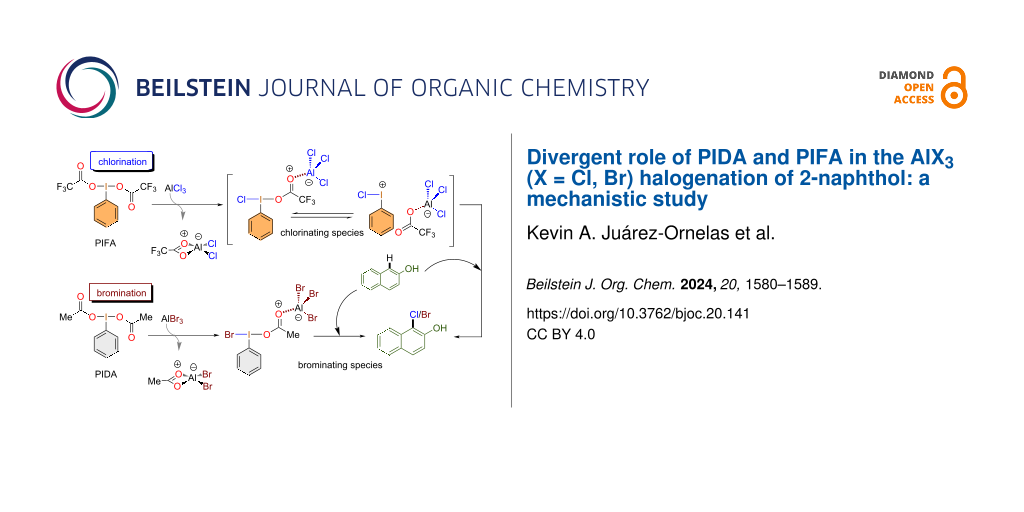
Hypervalent iodine(III) reagents have gained attention as strong oxidants with a low toxicity [1-8] and due to the ability to mimic reactivity [9] usually associated with transition metals [10,11]. Iodine(III) compounds have been used for the formation of different bond types, such as C–C [12,13], C–O [14,15], C–N [16], C–S [17], C–CN [18], C–F [19-21], C–I [22,23], C–NO2 [24,25] and, in the context of this work, C–X (X = Cl, Br) [26-31]. So far, different protocols for the halogenation of arenes using iodine(III) reagents have been described, mainly using (diacetoxyiodo)benzene (PIDA)/TMSCl, PIDA/TMSBr [32], and [bis(trifluoroacetoxy)iodo]benzene (PIFA)/TMSBr [33]. We have recently developed a new protocol for the oxidative chlorination and bromination of naphthols using the PIFA/AlCl3 [26] and PIDA/AlBr3 [28,29] systems. These unprecedented protocols combined iodine(III) reagents and aluminum salts to achieve chlorination and bromination of electron-rich arenes under mild and experimentally straightforward conditions (Scheme 1).
Scheme 1: Representative protocols for the oxidative aromatic chlorination and bromination with iodine(III) reagents.
Scheme 1: Representative protocols for the oxidative aromatic chlorination and bromination with iodine(III) r...
The synthesis of aryl halides is of great academic and industrial importance. Recently, our research group has developed a new procedure for the ortho-selective chlorination of phenols under mild conditions in a short reaction time [26]. The chlorinating species was generated in situ simply by mixing PIFA with a Lewis acid, in this case AlCl3. The importance of this protocol arises from the oxidation of an AlCl3-based chlorine atom, which is an available and cheap reagent. Then it is used as an electrophile source in the chlorination process with an umpolung reactivity. In contrast to the suggested traceroute where the chlorine or bromine atom is attached to the hypervalent iodine center of the plausible reagent PhIX2 (X = Cl, Br), our new protocol opens up a broad path for the reaction through different halogenating species. For a deeper understanding of these reactions, we explored different pathways of the reaction mechanisms for the ortho-halogenation using 2-naphthol as a model substrate (Scheme 2). In such a way, we found a reaction pathway that was energetically favored.
Scheme 2: Chlorination of 2-naphthol using the PIFA/AlCl3, 1:2 system.
Scheme 2: Chlorination of 2-naphthol using the PIFA/AlCl3, 1:2 system.
Based on our successful procedure for chlorination, we also developed an efficient protocol for the electrophilic bromination of arenes, mainly phenols [28,29]. Accordingly, the bromination reaction was initially explored by mixing PIFA and AlBr3, which gave an acceptable yield (84%). However, other iodine(III) reagents were tested as oxidants during the optimization process. Thus, when mixing PIDA with aluminum bromide, the reaction occurred with an unexpectedly higher yield (93%) than with PIFA. Therefore, the bromination reaction proceeded in the presence of PIDA/AlBr3 as a brominating system using MeCN as solvent (Scheme 3).
Scheme 3: Bromination of 2-naphthol using the PIDA/AlBr3, 1:2 system.
Scheme 3: Bromination of 2-naphthol using the PIDA/AlBr3, 1:2 system.
In light of the relevance of this newly discovered reactivity and the scarce mechanistic and theoretical studies available [33], we computationally explored all of the different plausible pathways to elucidate the most feasible route that allowed the reported halogenation under these new reaction conditions. In this work, we systematically investigated the influence of PIDA and PIFA in the chlorination and bromination reactions. Interestingly, we found an excellent agreement between the theoretical predictions and the experimental results.
Results and Discussion
Computational details
The equilibrium geometry of reagents and products, the stationary points, and transition-state structures were optimized by density functional theory (DFT) calculations employing the software Gaussian 16 [34]. Although the B3LYP functional could be suitable for these calculations, e.g., for tracing reaction pathways, nitrations, halogenations, or FC acylations in solution, we found the ω-B97XD functional [35] appropriate for this study because it considered dispersion interactions through a range separation (22% for short range and 100% Hartree–Fock for long range), which properly describes thermochemistry and noncovalent interactions. When searching for the critical points along the potential surface energy of the possible chlorination and bromination pathways studied in this work, Br and I atoms were treated with the revised version of the LANL2DZ basis set and effective core potential, referred to as LANL08(d), providing d-type polarization functions. Meanwhile, the 6-31G(d) basis set was used for the other atoms (i.e., H, C, O, F, Al, etc.).
Geometry optimizations were carried out without any symmetry constraints, and the stationary points were characterized by analytical frequency calculations, i.e., energy minima (reactants, intermediates, and products) must exhibit only positive harmonic frequencies, whereas each energy maximum (transition state) exhibited only one negative frequency. From these last calculations, zero-point energy, thermal, and entropy corrections were obtained, which were added to the electronic energy to express the calculated values as Gibbs free energy at 298 K and 1 atm.
All our calculations were performed in the gas phase. Then, the solvent effects were included according to the polarizable continuum model via the solvent model density (SMD) option considering Truhlar’s model [36-40] and MeCN as the solvent. Single-point calculations were improved using a mixed basis set of triple-ζ quality with a polarization function, 6-311G(d,p) for all atoms except for Br and I, which were treated with the LANL08d relativistic pseudopotential [41-43], i.e., the composite level of theory used is the following: (SMD: MeCN) ω-B97XD/(6-311G(d,p),LANL08d)//ω-B97XD/6-31G(d), LANL08d.
Chlorination mechanism
The reaction mechanism for the chlorination of 2-naphthol using one equivalent of PIFA and two equivalents of aluminum chloride is outlined in Scheme 4.
Scheme 4: Reaction mechanism for the chlorination of 2-naphthol using the PIFA/AlCl3, 1:2 system.
Scheme 4: Reaction mechanism for the chlorination of 2-naphthol using the PIFA/AlCl3, 1:2 system.
The chlorination mechanism starts when PIFA coordinates the first equivalent of aluminum chloride to give the corresponding adduct PIFA–AlCl3. Next, a chlorine atom is transferred to the iodine(III) center to yield I-1–Cl via TS1–Cl with the release of the complex TFAO–AlCl2. Then, the second equivalent of aluminum chloride coordinates the TFAO ligand, giving rise to the chlorinating species I-2–Cl in equilibrium with I-3–Cl. At this point, 2-naphthol reacts, leading to the formation of the ion pair I-4–Cl via chlorine atom transfer, which then yields the adduct I-5–Cl trough transition state TS2–Cl. Then, the release of the second equivalent of the TFAO–AlCl2 complex yields the final product 1-chloro-2-naphthol (P–Cl).
The calculated mechanism for the chlorination reaction starts with coordination of a PIFA oxygen atom to aluminum chloride. This generates a highly exergonic PIFA–AlCl3 adduct. In Figure 1, the Gibbs free energy of this adduct is set as 0 kcal/mol for more clarity. Herein, one chlorine atom is transferred from aluminum to the hypervalent iodine(III) center through six-membered-ring transition state TS1–Cl (ΔG‡ = 9.7 kcal/mol, selected bond lengths 2.76, 1.22, 1.27, 1.78, 2.60, and 2.86 Å for I–O, O–C, C–O, O–Al, Al–Cl, and Cl–I, respectively). Then, the tetracoordinate TFAO–AlCl2 salt is released, giving rise to intermediate I-1–Cl (ΔG = −25.2 kcal/mol), which contains the key Cl–I(III) bond, in a formal TFAO/Cl ligand exchange. The Cl–I bond length is 2.46 Å, with the halogen atom sharing the hypervalent iodine bond in the equatorial position. Next, the second equivalent of aluminum chloride coordinates to the TFAO ligand, forming active chlorinating species I-2–Cl (ΔG = −18.3 kcal/mol). This energetically favored step is in equilibrium with the ion pair I-3–Cl (ΔG = −0.5 kcal/mol). It is worth mentioning that the slight difference in energy between both states indicates the importance of the spontaneous interconversion of both species, which is observed only in the presence of two equivalents of the Lewis acid.
![[1860-5397-20-141-1]](/bjoc/content/figures/1860-5397-20-141-1.png?scale=2.0&max-width=1024&background=FFFFFF)
Figure 1: Energy profile for the chlorination of 2-naphthol in the presence of PIFA and AlCl3.
Figure 1: Energy profile for the chlorination of 2-naphthol in the presence of PIFA and AlCl3.
After the addition of 2-naphthol, the chlorine atom is introduced barrier-free into the phenolic ring, producing the nonaromatic intermediate I-4–Cl (ΔG = −23.1 kcal/mol). Next, aromatization assisted by TFAO–AlCl2 via TS2–Cl (ΔG‡ = 11.1 kcal/mol) and hydrogen transfer from the nonaromatic intermediate to TFAO–AlCl2 are observed. In TS2–Cl, the energy barrier must be overcome to give rise to the 1-chloro-2-naphthol adduct with TFA–OH–AlCl2, I-5–Cl (ΔG = −2.6 kcal/mol), which spontaneously yields the final 1-chloro-2-naphthol (P–Cl) with concomitant release of TFAO–AlCl2 in a highly exothermic process (ΔG = −44.2 kcal/mol, Figure 1).
Other relevant routes for this chlorination process, which involve a different stoichiometry or the formation of PhICl2 as chlorinating species, were also investigated and ruled out. Thus, for the chlorination of 2-naphthol with the PIFA/AlCl3, 1:1 system, we found that in general, once the intermediate I-1–Cl is formed, the following coordination of 2-naphthol with the TFAO ligand via TS2 is energetically less favored (ΔG‡ = 16.2 kcal/mol, see Supporting Information File 1 for details of the explored chlorination and bromination mechanisms). Additionally, for this mechanism, we identified that the formation of TS4 has the highest energy barrier (ΔG‡ = 20.2 kcal/mol), becoming a less probable route. This result also confirms the relevance of using two equivalents of aluminum chloride.
The aromatic chlorination with iodine(III) reagents broadly employs PhICl2 [7]. Thus, we explored two alternatives for the chlorination of 2-naphthol to identify or rule out this potential reaction pathway. The first explored mechanism involves PIFA/AlCl3 and the second PIDA/AlCl3 (see Figures S2 and S3, respectively, Supporting Information File 1). In both cases, the route involves the formation of PhICl2 as the chlorinating reagent by considering two equivalents of AlCl3 (PIFA/AlCl3 or PIDA/AlCl3, 1:2). Overall, we characterized four transition states along the reaction coordinates for both pathways. Although the PIFA-assisted mechanism follows a similar route to that described in Figure 1 until the formation of the active chlorinating species, in this case, the formation of TS2 requires 18.1 kcal/mol, which is an energetically more demanding process than the equilibration between I-2–Cl and ion pair I-3–Cl, proposed as active chlorinating species in Figure 1 and requiring less than 1 kcal/mol. It is worth mentioning that once PhICl2 is formed, the energy barrier to TS3 is 21.5 kcal/mol. These energy differences suggest that the traceroute PhICl2 is less viable for the chlorination of 2-naphthol.
On the other hand, in the presence of PIDA (Figure S3, Supporting Information File 1), when the reaction occurs through the chlorinating species PhICl2, we found that TS1, TS2, and TS4 require 17.7, 13.8, and 16.5 kcal/mol, respectively. Considering the high transition-state energy barrier in the proposed mechanism shown in Figure 1 for TS2–Cl (11.1 kcal/mol), this route is less probable. Additionally, we observed that chlorination of naphthol (the formation of I-6) could be the determining step since we found a coupling between the ring of the chlorinating species and naphthol during TS4, i.e., it could disfavor the PIDA-assisted chlorination traceroute via PhICl2. Thus, using PIFA and two equivalents of AlCl3 resulted in the highest yield, which is in agreement with our experiments.
As a consequence of the previous analysis, the chlorination process is energetically favored in the presence of PIFA/AlCl3, 1:2 through the formation of PhICl–TFAO–AlCl3 in equilibrium with [PhICl][TFAO–AlCl3] as chlorinating species.
Bromination mechanism
The reaction mechanism for the bromination of 2-naphthol using one equivalent of PIDA and two equivalents of aluminum bromide is shown in Scheme 5.
Scheme 5: Calculated reaction mechanism for the bromination of 2-naphthol using the PIDA/AlBr3, 1:2 system.
Scheme 5: Calculated reaction mechanism for the bromination of 2-naphthol using the PIDA/AlBr3, 1:2 system.
PIDA coordinates the first equivalent of aluminum bromide to form the adduct PIDA–AlBr3, which spontaneously dissociates, giving ion pair I-1–Br. Next, via TS1–Br, complex AcO–AlBr2 is released with subsequent formation of intermediate I-2–Br. Then, the second equivalent of aluminum bromide coordinates to an acetate ligand, forming adduct I-3–Br, which is the brominating species. At this point, 2-naphthol reacts and forms I-4–Br via TS2–Br. Afterwards, the second equivalent of the AcO–AlBr2 complex and HBr are released with concomitant formation of I-5–Br trough TS2–Br. Then, I-5–Br spontaneously isomerizes to give I-6–Br via the transition state TS4–Br. Finally, I-6–Br tautomerizes, yielding the experimentally observed 1-bromo-2-naphthol (P–Br).
Based on our calculations, the bromination reaction proceeds through a stepwise mechanism. Thus, the reaction starts with the coordination of aluminum bromide to an acetate ligand in PIDA to form the PIDA–AlBr3 adduct in a highly exergonic process. Similar to the previous section, the Gibbs free energy at this point was set as 0 kcal/mol for reference. At this stage, the PIDA–AlBr3 adduct undergoes ionization, giving rise to the corresponding ion pair I-1–Br (ΔG = −31.3 kcal/mol) in a highly exergonic and energetically favorable process. Next, an intramolecular SN2 reaction of the formed aluminum anion transfers a bromine atom to the electrophilic iodine(III) center through TS1–Br, which has a feasible energy barrier of 8.3 kcal/mol. The I–Br and Br–Al bond lengths are 3.15 and 2.78 Å, respectively, and the I–Br–Al angle is 93.1o, which is close to the common T-shape of such hypervalent iodine(III) species. This step releases the tetracoordinate AcO–AlBr2 salt and gives rise to the intermediate I-2–Br (ΔG = −9 kcal/mol), which contains the key Br–I(III) bond with a length of 2.65 Å. Herein, we could identify an energetically favored AcO/Br ligand exchange that releases 35.2 kcal/mol. At this point, the second equivalent of aluminum bromide is coordinated by an acetate ligand to produce the active brominating species Br–I(Ph)–OAc–AlBr3 (I-3–Br). Then, 2-naphthol adds to the iodine(III) species to release the activated Br3Al–OAc ligand through transition state TS2–Br (ΔG‡ = 11.7 kcal/mol), which leads to the protonated intermediate I-4–Br. The next step is a deprotonation assisted by the released Br3Al–OAc species. This allows the formation of the AcO–AlBr2 salt via TS3–Br and the trans intermediate I-5–Br, which contains a Br–I(Ph)–O–naphthyl bond of 2.14 Å length. The last step is the bromination of I-5–Br by isomerization to the cis transition state TS4–Br (ΔG‡ = 16.1 kcal/mol), which yields the brominated nonaromatic intermediate I-6–Br in a highly exothermic step (ΔG = −52.3 kcal/mol). Finally, I-6–Br undergoes spontaneous aromatization, converting it into the experimentally observed 1-bromo-2-naphthol (P–Br), which is more stable than I-6–Br by 2.6 kcal/mol (Figure 2).
![[1860-5397-20-141-2]](/bjoc/content/figures/1860-5397-20-141-2.png?scale=2.0&max-width=1024&background=FFFFFF)
Figure 2: Calculated mechanism for the bromination of 2-naphthol in the presence of PIDA and AlBr3.
Figure 2: Calculated mechanism for the bromination of 2-naphthol in the presence of PIDA and AlBr3.
We also explored the plausible mechanisms for the bromination of 2-naphthol mediated by PIFA in a 1:2 ratio (Figure S4, Supporting Information File 1). In this proposal, we observed that the energy barriers to reach TS1 (10.6 kcal/mol) and TS2 (16.7 kcal/mol) are higher than those calculated for the mechanism shown in Figure 2, namely 8.3 kcal/mol for TS1–Br and 16.1 kcal/mol for TS4–Br.
Other possible mechanisms involve the formation of PhIBr2. For these scenarios, the reaction pathway with PIDA and PIFA, respectively, involves two equivalents of AlBr3 (Figures S5 and S6, Supporting Information File 1). Calculations indicated that each of these pathways proceeds along four transition states. Moreover, we found that the coordination of AlBr3 to I-2 to form TS2 has the highest energy barrier (determining step, ΔG‡ for TS2 53.3 kcal/mol) in the presence of PIDA. Meanwhile, formation of TS3 (ΔG‡ = 21.3 kcal/mol) is the limiting step of the mechanism in the presence of PIFA.
Then, in the presence of the PIFA/AlBr3 system, bromination of 2-naphthol is the energetically most favored pathway. Although all these reactions occur through four transition states, significant energy differences exist concerning the PIDA/AlBr3 system. For example, the activation barrier of TS2 was 41.6 kcal/mol higher in energy than that in the mechanism in Figure 2. A similar energy profile was obtained for the bromination of 2-naphthol in the presence of PIFA (and 2 equivalents of AlBr3) compared to Figure 2. The energy difference from I-1 to TS1 (2.3 kcal/mol for the reaction in the presence of PIFA) could be the reason why the experimental yield is higher in the presence of PIDA and two equivalents AlBr3 rather than PIFA and two equivalents AlBr3 when considering the different hypervalent iodine reagents for this reaction.
To find the correlations between experiments and theoretical calculations, chlorination and bromination of 2-naphthol using PIFA/AlCl3 and PIDA/AlBr3 were carried out. Consequently, we found an excellent correlation between the yield and the energy barrier (Table 1).
Conclusion
We elucidated the energetically most viable pathway for the chlorination and bromination of 2-naphthol using the novel systems PIFA/AlCl3 and PIDA/AlBr3 in a 2:1 ratio in both cases. We found that the energetically most favored reaction proceeds through the chlorinating species I-2–Cl and I-3–Cl (rather than PhICl2), which are in an equilibrium. The bromination is more efficient with PIDA/AlBr3 through the formation of the intermediate I-3–Br as active brominating species. Similarly, involvement of PhIBr2 is energetically less favored compared to our proposed pathway. One key step is the coordination of a second equivalent of AlX3 to TFAO or AcO in PIFA or PIDA to promote the formation of the active halogenating species I-2 and I-3 for chlorination and bromination, respectively. Although bromination reactions in the presence of PIDA and PIFA give an excellent experimental yield, slight energy differences in the pathways explained why PIFA/AlCl3 for chlorination and PIDA/AlBr3 for bromination are better choices for these reactions.
Supporting Information
| Supporting Information File 1: Optimized Cartesian coordinates of all structures and alternative mechanisms. | ||
| Format: PDF | Size: 1.2 MB | Download |
Data Availability Statement
Additional research data is not shared.
References
-
Yoshimura, A.; Zhdankin, V. V. Chem. Rev. 2016, 116, 3328–3435. doi:10.1021/acs.chemrev.5b00547
Return to citation in text: [1] -
Zhdankin, V. V. ARKIVOC 2009, No. i, 1–62.
Return to citation in text: [1] -
Zhdankin, V. V. Hypervalent Iodine Chemistry: Preparation, Structure and Synthetic Applications of Polyvalent Iodine Compounds; Wiley-VCH: Weinheim, Germany, 2014. doi:10.1002/9781118341155
Return to citation in text: [1] -
Chávez‐Rivera, R.; Navarro‐Santos, P.; Chacón‐García, L.; Ortiz‐Alvarado, R.; Solorio Alvarado, C. R. ChemistrySelect 2023, 8, e202303425. doi:10.1002/slct.202303425
Return to citation in text: [1] -
Segura-Quezada, L. A.; Torres-Carbajal, K. R.; Juárez-Ornelas, K. A.; Navarro-Santos, P.; Granados-López, A. J.; González-García, G.; Ortiz-Alvarado, R.; de León-Solis, C.; Solorio-Alvarado, C. R. Curr. Org. Chem. 2022, 26, 1954–1968. doi:10.2174/1385272826666220621142211
Return to citation in text: [1] -
Kikushima, K.; Elboray, E. E.; Jiménez-Halla, J. O. C.; Solorio-Alvarado, C. R.; Dohi, T. Org. Biomol. Chem. 2022, 20, 3231–3248. doi:10.1039/d1ob02501e
Return to citation in text: [1] -
Segura-Quezada, L. A.; Torres-Carbajal, K. R.; Juárez-Ornelas, K. A.; Alonso-Castro, A. J.; Ortiz-Alvarado, R.; Dohi, T.; Solorio-Alvarado, C. R. Org. Biomol. Chem. 2022, 20, 5009–5034. doi:10.1039/d2ob00741j
Return to citation in text: [1] [2] -
Segura-Quezada, L. A.; Torres-Carbajal, K. R.; Satkar, Y.; Juárez Ornelas, K. A.; Mali, N.; Patil, D. B.; Gámez-Montaño, R.; Zapata-Morales, J. R.; Lagunas-Rivera, S.; Ortíz-Alvarado, R.; Solorio-Alvarado, C. R. Mini-Rev. Org. Chem. 2021, 18, 159–172. doi:10.2174/1570193x17999200504095803
Return to citation in text: [1] -
Wang, B.; Graskemper, J. W.; Qin, L.; DiMagno, S. G. Angew. Chem., Int. Ed. 2010, 49, 4079–4083. doi:10.1002/anie.201000695
Return to citation in text: [1] -
Segura-Quezada, L. A.; Torres-Carbajal, K. R.; Mali, N.; Patil, D. B.; Luna-Chagolla, M.; Ortiz-Alvarado, R.; Tapia-Juárez, M.; Fraire-Soto, I.; Araujo-Huitrado, J. G.; Granados-López, A. J.; Gutiérrez-Hernández, R.; Reyes-Estrada, C. A.; López-Hernández, Y.; López, J. A.; Chacón-García, L.; Solorio-Alvarado, C. R. ACS Omega 2022, 7, 6944–6955. doi:10.1021/acsomega.1c06637
Return to citation in text: [1] -
Nahide, P. D.; Jiménez-Halla, J. O. C.; Wrobel, K.; Solorio-Alvarado, C. R.; Ortiz Alvarado, R.; Yahuaca-Juárez, B. Org. Biomol. Chem. 2018, 16, 7330–7335. doi:10.1039/c8ob02056f
Return to citation in text: [1] -
Kita, Y.; Dohi, T.; Morimoto, K. J. Synth. Org. Chem., Jpn. 2011, 69, 1241–1250. doi:10.5059/yukigoseikyokaishi.69.1241
Return to citation in text: [1] -
Satkar, Y.; Wrobel, K.; Trujillo-González, D. E.; Ortiz-Alvarado, R.; Jiménez-Halla, J. O. C.; Solorio-Alvarado, C. R. Front. Chem. (Lausanne, Switz.) 2020, 8, 10.3389/fchem.2020.563470. doi:10.3389/fchem.2020.563470
Return to citation in text: [1] -
Dohi, T.; Maruyama, A.; Yoshimura, M.; Morimoto, K.; Tohma, H.; Kita, Y. Angew. Chem., Int. Ed. 2005, 44, 6193–6196. doi:10.1002/anie.200501688
Return to citation in text: [1] -
Yahuaca-Juárez, B.; González, G.; Ramírez-Morales, M. A.; Alba-Betancourt, C.; Deveze-Álvarez, M. A.; Mendoza-Macías, C. L.; Ortiz-Alvarado, R.; Juárez-Ornelas, K. A.; Solorio-Alvarado, C. R.; Maruoka, K. Synth. Commun. 2020, 50, 539–548. doi:10.1080/00397911.2019.1707225
Return to citation in text: [1] -
Dohi, T.; Maruyama, A.; Minamitsuji, Y.; Takenaga, N.; Kita, Y. Chem. Commun. 2007, 1224–1226. doi:10.1039/b616510a
Return to citation in text: [1] -
Kumar, R. K.; Manna, S.; Mahesh, D.; Sar, D.; Punniyamurthy, T. Asian J. Org. Chem. 2013, 2, 843–847. doi:10.1002/ajoc.201300151
Return to citation in text: [1] -
Dohi, T.; Morimoto, K.; Takenaga, N.; Goto, A.; Maruyama, A.; Kiyono, Y.; Tohma, H.; Kita, Y. J. Org. Chem. 2007, 72, 109–116. doi:10.1021/jo061820i
Return to citation in text: [1] -
Karam, O.; Jacquesy, J.-C.; Jouannetaud, M.-P. Tetrahedron Lett. 1994, 35, 2541–2544. doi:10.1016/s0040-4039(00)77165-6
Return to citation in text: [1] -
Zhao, Z.; To, A. J.; Murphy, G. K. Chem. Commun. 2019, 55, 14821–14824. doi:10.1039/c9cc08310c
Return to citation in text: [1] -
Chai, H.; Zhen, X.; Wang, X.; Qi, L.; Qin, Y.; Xue, J.; Xu, Z.; Zhang, H.; Zhu, W. ACS Omega 2022, 7, 19988–19996. doi:10.1021/acsomega.2c01791
Return to citation in text: [1] -
Mali, N.; Ibarra‐Gutiérrez, J. G.; Lugo Fuentes, L. I.; Ortíz‐Alvarado, R.; Chacón‐García, L.; Navarro‐Santos, P.; Jiménez‐Halla, J. O. C.; Solorio‐Alvarado, C. R. Eur. J. Org. Chem. 2022, 10.1002/ejoc.202201067. doi:10.1002/ejoc.202201067
Return to citation in text: [1] -
Satkar, Y.; Yera-Ledesma, L. F.; Mali, N.; Patil, D.; Navarro-Santos, P.; Segura-Quezada, L. A.; Ramírez-Morales, P. I.; Solorio-Alvarado, C. R. J. Org. Chem. 2019, 84, 4149–4164. doi:10.1021/acs.joc.9b00161
Return to citation in text: [1] -
Patil, D. B.; Gámez‐Montaño, R.; Ordoñez, M.; Solis‐Santos, M.; Jiménez‐Halla, J. O. C.; Solorio‐Alvarado, C. R. Eur. J. Org. Chem. 2022, e202201295. doi:10.1002/ejoc.202201295
Return to citation in text: [1] -
Juárez-Ornelas, K. A.; Jiménez-Halla, J. O. C.; Kato, T.; Solorio-Alvarado, C. R.; Maruoka, K. Org. Lett. 2019, 21, 1315–1319. doi:10.1021/acs.orglett.8b04141
Return to citation in text: [1] -
Nahide, P. D.; Ramadoss, V.; Juárez‐Ornelas, K. A.; Satkar, Y.; Ortiz‐Alvarado, R.; Cervera‐Villanueva, J. M. J.; Alonso‐Castro, Á. J.; Zapata‐Morales, J. R.; Ramírez‐Morales, M. A.; Ruiz‐Padilla, A. J.; Deveze‐Álvarez, M. A.; Solorio‐Alvarado, C. R. Eur. J. Org. Chem. 2018, 485–493. doi:10.1002/ejoc.201701399
Return to citation in text: [1] [2] [3] -
Cheng, D. P.; Chen, Z. C.; Zheng, Q. G. J. Chem. Res., Synop. 2002, 624–625. doi:10.3184/030823402103171032
Return to citation in text: [1] -
Satkar, Y.; Ramadoss, V.; Nahide, P. D.; García-Medina, E.; Juárez-Ornelas, K. A.; Alonso-Castro, A. J.; Chávez-Rivera, R.; Jiménez-Halla, J. O. C.; Solorio-Alvarado, C. R. RSC Adv. 2018, 8, 17806–17812. doi:10.1039/c8ra02982b
Return to citation in text: [1] [2] [3] -
Segura-Quezada, A.; Satkar, Y.; Patil, D.; Mali, N.; Wrobel, K.; González, G.; Zárraga, R.; Ortiz-Alvarado, R.; Solorio-Alvarado, C. R. Tetrahedron Lett. 2019, 60, 1551–1555. doi:10.1016/j.tetlet.2019.05.019
Return to citation in text: [1] [2] [3] -
Qin, Y.; Qi, L.; Zhen, X.; Wang, X.; Chai, H.; Ma, X.; Jiang, X.; Cai, X.; Zhu, W. J. Org. Chem. 2023, 88, 4359–4371. doi:10.1021/acs.joc.2c02967
Return to citation in text: [1] -
Juárez-Ornelas, K. A.; Báez, J. E.; Solorio-Alvarado, C. R.; Jiménez-Halla, J. O. C. Starting Computational Study of the Chlorination Mechanism Reaction of 2-Naphthol with PIDA and AlCl3 via PhICl2 Formation as a Chlorinating Reagent. In Proceedings of the 24th International Electronic Conference on Synthetic Organic Chemistry, Nov 15–Dec 15, 2020; MDPI: Basel, Switzerland, 2020. doi:10.3390/ecsoc-24-08358
Return to citation in text: [1] -
Evans, P. A.; Brandt, T. A. Tetrahedron Lett. 1996, 37, 6443–6446. doi:10.1016/0040-4039(96)01427-x
Return to citation in text: [1] -
Granados, A.; Shafir, A.; Arrieta, A.; Cossío, F. P.; Vallribera, A. J. Org. Chem. 2020, 85, 2142–2150. doi:10.1021/acs.joc.9b02784
Return to citation in text: [1] [2] -
Gaussian 16, Revision C.01; Gaussian, Inc.: Wallingford, CT, 2009.
Return to citation in text: [1] -
Chai, J.-D.; Head-Gordon, M. Phys. Chem. Chem. Phys. 2008, 10, 6615–6620. doi:10.1039/b810189b
Return to citation in text: [1] -
Marenich, A. V.; Cramer, C. J.; Truhlar, D. G. J. Phys. Chem. B 2009, 113, 6378–6396. doi:10.1021/jp810292n
Return to citation in text: [1] -
Barone, V.; Cossi, M. J. Phys. Chem. A 1998, 102, 1995–2001. doi:10.1021/jp9716997
Return to citation in text: [1] -
Cossi, M.; Barone, V.; Mennucci, B.; Tomasi, J. Chem. Phys. Lett. 1998, 286, 253–260. doi:10.1016/s0009-2614(98)00106-7
Return to citation in text: [1] -
Barone, V.; Cossi, M.; Tomasi, J. J. Comput. Chem. 1998, 19, 404. doi:10.1002/(sici)1096-987x(199803)19:4<404::aid-jcc3>3.3.co;2-l
Return to citation in text: [1] -
Tomasi, J.; Mennucci, B.; Cammi, R. Chem. Rev. 2005, 105, 2999–3094. doi:10.1021/cr9904009
Return to citation in text: [1] -
Wadt, W. R.; Hay, P. J. J. Chem. Phys. 1985, 82, 284–298. doi:10.1063/1.448800
Return to citation in text: [1] -
Hay, P. J.; Wadt, W. R. J. Chem. Phys. 1985, 82, 299–310. doi:10.1063/1.448975
Return to citation in text: [1] -
Roy, L. E.; Hay, P. J.; Martin, R. L. J. Chem. Theory Comput. 2008, 4, 1029–1031. doi:10.1021/ct8000409
Return to citation in text: [1]
| 33. | Granados, A.; Shafir, A.; Arrieta, A.; Cossío, F. P.; Vallribera, A. J. Org. Chem. 2020, 85, 2142–2150. doi:10.1021/acs.joc.9b02784 |
| 26. | Nahide, P. D.; Ramadoss, V.; Juárez‐Ornelas, K. A.; Satkar, Y.; Ortiz‐Alvarado, R.; Cervera‐Villanueva, J. M. J.; Alonso‐Castro, Á. J.; Zapata‐Morales, J. R.; Ramírez‐Morales, M. A.; Ruiz‐Padilla, A. J.; Deveze‐Álvarez, M. A.; Solorio‐Alvarado, C. R. Eur. J. Org. Chem. 2018, 485–493. doi:10.1002/ejoc.201701399 |
| 28. | Satkar, Y.; Ramadoss, V.; Nahide, P. D.; García-Medina, E.; Juárez-Ornelas, K. A.; Alonso-Castro, A. J.; Chávez-Rivera, R.; Jiménez-Halla, J. O. C.; Solorio-Alvarado, C. R. RSC Adv. 2018, 8, 17806–17812. doi:10.1039/c8ra02982b |
| 29. | Segura-Quezada, A.; Satkar, Y.; Patil, D.; Mali, N.; Wrobel, K.; González, G.; Zárraga, R.; Ortiz-Alvarado, R.; Solorio-Alvarado, C. R. Tetrahedron Lett. 2019, 60, 1551–1555. doi:10.1016/j.tetlet.2019.05.019 |
| 1. | Yoshimura, A.; Zhdankin, V. V. Chem. Rev. 2016, 116, 3328–3435. doi:10.1021/acs.chemrev.5b00547 |
| 2. | Zhdankin, V. V. ARKIVOC 2009, No. i, 1–62. |
| 3. | Zhdankin, V. V. Hypervalent Iodine Chemistry: Preparation, Structure and Synthetic Applications of Polyvalent Iodine Compounds; Wiley-VCH: Weinheim, Germany, 2014. doi:10.1002/9781118341155 |
| 4. | Chávez‐Rivera, R.; Navarro‐Santos, P.; Chacón‐García, L.; Ortiz‐Alvarado, R.; Solorio Alvarado, C. R. ChemistrySelect 2023, 8, e202303425. doi:10.1002/slct.202303425 |
| 5. | Segura-Quezada, L. A.; Torres-Carbajal, K. R.; Juárez-Ornelas, K. A.; Navarro-Santos, P.; Granados-López, A. J.; González-García, G.; Ortiz-Alvarado, R.; de León-Solis, C.; Solorio-Alvarado, C. R. Curr. Org. Chem. 2022, 26, 1954–1968. doi:10.2174/1385272826666220621142211 |
| 6. | Kikushima, K.; Elboray, E. E.; Jiménez-Halla, J. O. C.; Solorio-Alvarado, C. R.; Dohi, T. Org. Biomol. Chem. 2022, 20, 3231–3248. doi:10.1039/d1ob02501e |
| 7. | Segura-Quezada, L. A.; Torres-Carbajal, K. R.; Juárez-Ornelas, K. A.; Alonso-Castro, A. J.; Ortiz-Alvarado, R.; Dohi, T.; Solorio-Alvarado, C. R. Org. Biomol. Chem. 2022, 20, 5009–5034. doi:10.1039/d2ob00741j |
| 8. | Segura-Quezada, L. A.; Torres-Carbajal, K. R.; Satkar, Y.; Juárez Ornelas, K. A.; Mali, N.; Patil, D. B.; Gámez-Montaño, R.; Zapata-Morales, J. R.; Lagunas-Rivera, S.; Ortíz-Alvarado, R.; Solorio-Alvarado, C. R. Mini-Rev. Org. Chem. 2021, 18, 159–172. doi:10.2174/1570193x17999200504095803 |
| 14. | Dohi, T.; Maruyama, A.; Yoshimura, M.; Morimoto, K.; Tohma, H.; Kita, Y. Angew. Chem., Int. Ed. 2005, 44, 6193–6196. doi:10.1002/anie.200501688 |
| 15. | Yahuaca-Juárez, B.; González, G.; Ramírez-Morales, M. A.; Alba-Betancourt, C.; Deveze-Álvarez, M. A.; Mendoza-Macías, C. L.; Ortiz-Alvarado, R.; Juárez-Ornelas, K. A.; Solorio-Alvarado, C. R.; Maruoka, K. Synth. Commun. 2020, 50, 539–548. doi:10.1080/00397911.2019.1707225 |
| 26. | Nahide, P. D.; Ramadoss, V.; Juárez‐Ornelas, K. A.; Satkar, Y.; Ortiz‐Alvarado, R.; Cervera‐Villanueva, J. M. J.; Alonso‐Castro, Á. J.; Zapata‐Morales, J. R.; Ramírez‐Morales, M. A.; Ruiz‐Padilla, A. J.; Deveze‐Álvarez, M. A.; Solorio‐Alvarado, C. R. Eur. J. Org. Chem. 2018, 485–493. doi:10.1002/ejoc.201701399 |
| 12. | Kita, Y.; Dohi, T.; Morimoto, K. J. Synth. Org. Chem., Jpn. 2011, 69, 1241–1250. doi:10.5059/yukigoseikyokaishi.69.1241 |
| 13. | Satkar, Y.; Wrobel, K.; Trujillo-González, D. E.; Ortiz-Alvarado, R.; Jiménez-Halla, J. O. C.; Solorio-Alvarado, C. R. Front. Chem. (Lausanne, Switz.) 2020, 8, 10.3389/fchem.2020.563470. doi:10.3389/fchem.2020.563470 |
| 28. | Satkar, Y.; Ramadoss, V.; Nahide, P. D.; García-Medina, E.; Juárez-Ornelas, K. A.; Alonso-Castro, A. J.; Chávez-Rivera, R.; Jiménez-Halla, J. O. C.; Solorio-Alvarado, C. R. RSC Adv. 2018, 8, 17806–17812. doi:10.1039/c8ra02982b |
| 29. | Segura-Quezada, A.; Satkar, Y.; Patil, D.; Mali, N.; Wrobel, K.; González, G.; Zárraga, R.; Ortiz-Alvarado, R.; Solorio-Alvarado, C. R. Tetrahedron Lett. 2019, 60, 1551–1555. doi:10.1016/j.tetlet.2019.05.019 |
| 10. | Segura-Quezada, L. A.; Torres-Carbajal, K. R.; Mali, N.; Patil, D. B.; Luna-Chagolla, M.; Ortiz-Alvarado, R.; Tapia-Juárez, M.; Fraire-Soto, I.; Araujo-Huitrado, J. G.; Granados-López, A. J.; Gutiérrez-Hernández, R.; Reyes-Estrada, C. A.; López-Hernández, Y.; López, J. A.; Chacón-García, L.; Solorio-Alvarado, C. R. ACS Omega 2022, 7, 6944–6955. doi:10.1021/acsomega.1c06637 |
| 11. | Nahide, P. D.; Jiménez-Halla, J. O. C.; Wrobel, K.; Solorio-Alvarado, C. R.; Ortiz Alvarado, R.; Yahuaca-Juárez, B. Org. Biomol. Chem. 2018, 16, 7330–7335. doi:10.1039/c8ob02056f |
| 32. | Evans, P. A.; Brandt, T. A. Tetrahedron Lett. 1996, 37, 6443–6446. doi:10.1016/0040-4039(96)01427-x |
| 7. | Segura-Quezada, L. A.; Torres-Carbajal, K. R.; Juárez-Ornelas, K. A.; Alonso-Castro, A. J.; Ortiz-Alvarado, R.; Dohi, T.; Solorio-Alvarado, C. R. Org. Biomol. Chem. 2022, 20, 5009–5034. doi:10.1039/d2ob00741j |
| 9. | Wang, B.; Graskemper, J. W.; Qin, L.; DiMagno, S. G. Angew. Chem., Int. Ed. 2010, 49, 4079–4083. doi:10.1002/anie.201000695 |
| 33. | Granados, A.; Shafir, A.; Arrieta, A.; Cossío, F. P.; Vallribera, A. J. Org. Chem. 2020, 85, 2142–2150. doi:10.1021/acs.joc.9b02784 |
| 19. | Karam, O.; Jacquesy, J.-C.; Jouannetaud, M.-P. Tetrahedron Lett. 1994, 35, 2541–2544. doi:10.1016/s0040-4039(00)77165-6 |
| 20. | Zhao, Z.; To, A. J.; Murphy, G. K. Chem. Commun. 2019, 55, 14821–14824. doi:10.1039/c9cc08310c |
| 21. | Chai, H.; Zhen, X.; Wang, X.; Qi, L.; Qin, Y.; Xue, J.; Xu, Z.; Zhang, H.; Zhu, W. ACS Omega 2022, 7, 19988–19996. doi:10.1021/acsomega.2c01791 |
| 24. | Patil, D. B.; Gámez‐Montaño, R.; Ordoñez, M.; Solis‐Santos, M.; Jiménez‐Halla, J. O. C.; Solorio‐Alvarado, C. R. Eur. J. Org. Chem. 2022, e202201295. doi:10.1002/ejoc.202201295 |
| 25. | Juárez-Ornelas, K. A.; Jiménez-Halla, J. O. C.; Kato, T.; Solorio-Alvarado, C. R.; Maruoka, K. Org. Lett. 2019, 21, 1315–1319. doi:10.1021/acs.orglett.8b04141 |
| 36. | Marenich, A. V.; Cramer, C. J.; Truhlar, D. G. J. Phys. Chem. B 2009, 113, 6378–6396. doi:10.1021/jp810292n |
| 37. | Barone, V.; Cossi, M. J. Phys. Chem. A 1998, 102, 1995–2001. doi:10.1021/jp9716997 |
| 38. | Cossi, M.; Barone, V.; Mennucci, B.; Tomasi, J. Chem. Phys. Lett. 1998, 286, 253–260. doi:10.1016/s0009-2614(98)00106-7 |
| 39. | Barone, V.; Cossi, M.; Tomasi, J. J. Comput. Chem. 1998, 19, 404. doi:10.1002/(sici)1096-987x(199803)19:4<404::aid-jcc3>3.3.co;2-l |
| 40. | Tomasi, J.; Mennucci, B.; Cammi, R. Chem. Rev. 2005, 105, 2999–3094. doi:10.1021/cr9904009 |
| 18. | Dohi, T.; Morimoto, K.; Takenaga, N.; Goto, A.; Maruyama, A.; Kiyono, Y.; Tohma, H.; Kita, Y. J. Org. Chem. 2007, 72, 109–116. doi:10.1021/jo061820i |
| 26. | Nahide, P. D.; Ramadoss, V.; Juárez‐Ornelas, K. A.; Satkar, Y.; Ortiz‐Alvarado, R.; Cervera‐Villanueva, J. M. J.; Alonso‐Castro, Á. J.; Zapata‐Morales, J. R.; Ramírez‐Morales, M. A.; Ruiz‐Padilla, A. J.; Deveze‐Álvarez, M. A.; Solorio‐Alvarado, C. R. Eur. J. Org. Chem. 2018, 485–493. doi:10.1002/ejoc.201701399 |
| 27. | Cheng, D. P.; Chen, Z. C.; Zheng, Q. G. J. Chem. Res., Synop. 2002, 624–625. doi:10.3184/030823402103171032 |
| 28. | Satkar, Y.; Ramadoss, V.; Nahide, P. D.; García-Medina, E.; Juárez-Ornelas, K. A.; Alonso-Castro, A. J.; Chávez-Rivera, R.; Jiménez-Halla, J. O. C.; Solorio-Alvarado, C. R. RSC Adv. 2018, 8, 17806–17812. doi:10.1039/c8ra02982b |
| 29. | Segura-Quezada, A.; Satkar, Y.; Patil, D.; Mali, N.; Wrobel, K.; González, G.; Zárraga, R.; Ortiz-Alvarado, R.; Solorio-Alvarado, C. R. Tetrahedron Lett. 2019, 60, 1551–1555. doi:10.1016/j.tetlet.2019.05.019 |
| 30. | Qin, Y.; Qi, L.; Zhen, X.; Wang, X.; Chai, H.; Ma, X.; Jiang, X.; Cai, X.; Zhu, W. J. Org. Chem. 2023, 88, 4359–4371. doi:10.1021/acs.joc.2c02967 |
| 31. | Juárez-Ornelas, K. A.; Báez, J. E.; Solorio-Alvarado, C. R.; Jiménez-Halla, J. O. C. Starting Computational Study of the Chlorination Mechanism Reaction of 2-Naphthol with PIDA and AlCl3 via PhICl2 Formation as a Chlorinating Reagent. In Proceedings of the 24th International Electronic Conference on Synthetic Organic Chemistry, Nov 15–Dec 15, 2020; MDPI: Basel, Switzerland, 2020. doi:10.3390/ecsoc-24-08358 |
| 41. | Wadt, W. R.; Hay, P. J. J. Chem. Phys. 1985, 82, 284–298. doi:10.1063/1.448800 |
| 42. | Hay, P. J.; Wadt, W. R. J. Chem. Phys. 1985, 82, 299–310. doi:10.1063/1.448975 |
| 43. | Roy, L. E.; Hay, P. J.; Martin, R. L. J. Chem. Theory Comput. 2008, 4, 1029–1031. doi:10.1021/ct8000409 |
| 17. | Kumar, R. K.; Manna, S.; Mahesh, D.; Sar, D.; Punniyamurthy, T. Asian J. Org. Chem. 2013, 2, 843–847. doi:10.1002/ajoc.201300151 |
| 16. | Dohi, T.; Maruyama, A.; Minamitsuji, Y.; Takenaga, N.; Kita, Y. Chem. Commun. 2007, 1224–1226. doi:10.1039/b616510a |
| 22. | Mali, N.; Ibarra‐Gutiérrez, J. G.; Lugo Fuentes, L. I.; Ortíz‐Alvarado, R.; Chacón‐García, L.; Navarro‐Santos, P.; Jiménez‐Halla, J. O. C.; Solorio‐Alvarado, C. R. Eur. J. Org. Chem. 2022, 10.1002/ejoc.202201067. doi:10.1002/ejoc.202201067 |
| 23. | Satkar, Y.; Yera-Ledesma, L. F.; Mali, N.; Patil, D.; Navarro-Santos, P.; Segura-Quezada, L. A.; Ramírez-Morales, P. I.; Solorio-Alvarado, C. R. J. Org. Chem. 2019, 84, 4149–4164. doi:10.1021/acs.joc.9b00161 |
| 35. | Chai, J.-D.; Head-Gordon, M. Phys. Chem. Chem. Phys. 2008, 10, 6615–6620. doi:10.1039/b810189b |
© 2024 Juárez-Ornelas et al.; licensee Beilstein-Institut.
This is an open access article licensed under the terms of the Beilstein-Institut Open Access License Agreement (https://www.beilstein-journals.org/bjoc/terms), which is identical to the Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0). The reuse of material under this license requires that the author(s), source and license are credited. Third-party material in this article could be subject to other licenses (typically indicated in the credit line), and in this case, users are required to obtain permission from the license holder to reuse the material.