Abstract
The gold(I)-catalyzed reaction of water with o-acetylenyl-substituted phenyldiazoacetates provides 1H-isochromene derivatives in good yields. The reaction follows a catalytic sequence of gold carbene formation/water O–H insertion/alcohol-alkyne cyclization. The gold(I) complex is the only catalyst in each of these steps.
Graphical Abstract
Introduction
Transition metal carbene complexes are versatile intermediates and can undergo diverse transformations, including X–H (X = C, O, S, N, etc.) insertions, cyclopropanations, ylide formation, and 1,2-migrations [1-5]. Among the various methods to generate metal carbene complexes, transition metal-catalyzed decomposition of diazo compounds is the most straightforward and is highly reliable. Various transition metals have been found to decompose diazo compounds and then transfer a carbene unit to saturated or unsaturated organic substrates [3]. However, compared to the other group 11 metals, i.e., copper and silver, there are only a few reports on gold-catalyzed carbene transfer reactions of diazo compounds [6-20]. In 2005, Díaz-Requejo, Nolan, Pérez and co-workers reported the first example of carbene transfer from ethyl diazoacetate (EDA) using (IPr)AuCl. The subsequent generation of a gold carbene was followed by insertion into a phenyl C–H bond, an O–H bond, or an N–H bond [6]. Similar reactions were also reported by Dias and co-workers with a gold(I) ethylene complex [7]. Although the scope of those studies was limited to ethyl diazoacetate, the examples therein demonstrated that gold complexes can be used as efficient catalysts in carbene transfer reactions with diazo compounds.
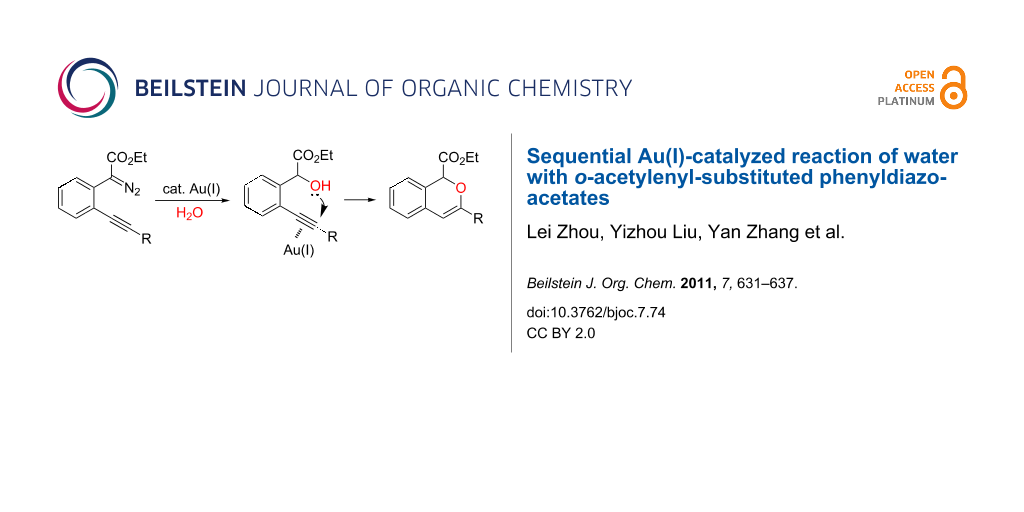
On the other hand, the development of reaction systems in which a single catalyst mediates two or more different reactions in a selective manner has become an emerging area of research [21-28]. This type of sequential or concurrent catalysis is particularly appealing in view of the requirements of green chemical processes in the fine chemical industry. In this context, we have previously reported the copper(I)-catalyzed reaction of amines with o-acetylenyl-substituted phenyldiazoacetates, which leads to a Cu(I)-catalyzed tandem N–H insertion/hydroamination of an alkyne [29]. Subsequently, we have tried to extend this reaction by replacing the amine component with water, and we expected that similar tandem reaction would occur. Copper is a good catalyst for the decomposition of diazo compounds and the subsequent insertion of water. However, we have found that it is not a suitable catalyst for the alcohol–alkyne cyclization. Since gold complexes are well-known for their efficacy in activating alkynes, we reasoned that a concurrent catalysis based on gold-catalyzed reaction of diazo compounds and alkynes might be possible [30]. Herein we report such a catalytic system, namely a gold(I)-catalyzed insertion/cyclization cascade by reacting water with o-acetylenyl-substituted phenyldiazoacetates. The reaction affords 1H-isochromene derivatives in good yields (Scheme 1) [31].
Scheme 1: Gold(I)-catalyzed insertion/cyclization of o-acetylenyl-substituted phenyldiazoacetates providing 1H-isochromene derivatives.
Scheme 1: Gold(I)-catalyzed insertion/cyclization of o-acetylenyl-substituted phenyldiazoacetates providing 1H...
Results and Discussion
At the onset of this investigation, o-acetylenyl-substituted phenyldiazoacetate 1a was selected as the model substrate. In a preliminary experiment, 1a was treated with CuI catalyst in a mixture of CH3CN and H2O (v:v = 1:1) (Table 1). As expected, only the water insertion product 4a was obtained as the major product and in high yield (91%) (Table 1, entry 1). Since previous reports have shown that silver [32-34] and gold [35-38] complexes are efficient catalysts for alcohol–alkyne cyclization, we then proceeded to examine other catalysts viz. AgOTf, AgF/PCy3, NaAuCl4 and AuCl (Table 1, entries 2–5). However, these metal complexes were not efficient catalysts for the decomposition of diazo compounds, and a large amount of starting materials remained. However, when 1a was treated with (PPh3)AuCl in a mixture of CH3CN and H2O, product 4a was obtained in 80% yield, along with the minor cyclization product 2a (8%) (Table 1, entry 6). Encouraged by this result, electron-rich ligand coordinated gold complexes (PMe3)AuCl and (IPr)AuCl were examined (Table 1, entries 7 and 8). It was found that (IPr)AuCl was an efficient catalyst for both carbene transfer and cyclization. Product 2a from 6-endo-dig cyclization and product 3a from 5-exo-dig cyclization were both obtained in nearly equal amounts in combined yield of 95%. Next, we examined the effect of the ratio of CH3CN and H2O with (IPr)AuCl as catalyst: A ratio of 1:1 (v:v) afforded the best results (Table 1, entries 9–12). It is worth noting that the reaction also occurred in pure water to afford the cyclization products, 2a and 3a, in moderate yield (Table 1, entry 11). Next, the effect of different co-solvents, such as DMF, NMP (N-methylpyrrolidone) and toluene, was investigated (Table 1, entries 13–15). Interestingly, when a mixture of H2O and DMF (v:v = 1:1) was used as the solvent, the ratio of 2a:3a increased to 4:1, with slightly diminished overall yield. The reaction with NMP or toluene as the co-solvent gave poor yields of desired products (Table 1, entries 14,15). Finally, the effect of temperature was evaluated: The reaction gave diminished yields when carried out at a temperature higher or lower than 80 °C (Table 1, entries 16–18).
Table 1: Optimization of reaction conditions with phenyldiazoacetate 1aa.
|
|
||||||
| Entry | Catalyst | Solvent | T/°C |
Yield
(2a + 3a)b |
2a:3a | 4a, Yield |
|---|---|---|---|---|---|---|
| 1 | CuI | H2O:CH3CN (1:1) | 80 | 0% | — | 91% |
| 2 | AgOTf | H2O:CH3CN (1:1) | 80 | 0% | — | <10% |
| 3 | AgF/PCy3 | H2O:CH3CN (1:1) | 80 | trace | — | trace |
| 4 | NaAuCl4 | H2O:CH3CN (1:1) | 80 | 0% | — | trace |
| 5 | AuCl | H2O:CH3CN (1:1) | 80 | 0% | — | trace |
| 6 | (PPh3)AuCl | H2O:CH3CN (1:1) | 80 | 8% | — | 80% |
| 7 | (PMe3)AuCl | H2O:CH3CN (1:1) | 80 | 10% | — | 15% |
| 8 | (IPr)AuCl | H2O:CH3CN (1:1) | 80 | 95% | 1:1 | 0% |
| 9 | (IPr)AuCl | H2O:CH3CN (1:3) | 80 | 25% | 2:3 | 61% |
| 10 | (IPr)AuCl | H2O:CH3CN (3:1) | 80 | 57% | 1:1.3 | 11% |
| 11 | (IPr)AuCl | H2O | 80 | 54% | 1:2 | 0% |
| 12c | (IPr)AuCl | CH3CN | 80 | 0% | 0 | 41% |
| 13 | (IPr)AuCl | H2O:DMF (1:1) | 80 | 90% | 4:1 | 0% |
| 14 | (IPr)AuCl | H2O:NMP (1:1) | 80 | 0% | — | trace |
| 15 | (IPr)AuCl | H2O:toluene (1:1) | 80 | 19% | 1:1 | 0% |
| 16 | (IPr)AuCl | H2O:DMF (1:1) | 100 | 57% | 3:1 | 0% |
| 17 | (IPr)AuCl | H2O:DMF (1:1) | 60 | 66% | 5:1 | 0% |
| 18 | (IPr)AuCl | H2O:DMF (1:1) | 40 | 48% | 5:1 | 0% |
aAll the reactions were carried out using 0.2 mmol phenyldiazoacetate 1a with 3 mol % of catalyst in 1 mL solvent for 24 h. bYield and ratio of 2a and 3a were measured by 1H NMR. c1 mmol of water was added.
With the optimized reaction conditions in hand, a series of substituted diazo compounds 1a–f were prepared, and their reactions with water in the presence of (IPr)AuCl in aqueous DMF were investigated. As shown in Table 2, all the reactions gave isochromene derivatives as the major products. In the reaction of diazo compounds 1e and 1f with water, only 6-endo-dig cyclization products 2e and 2f were isolated as the sole product in yields of 69% and 75%, respectively. Functional groups such as bromo and hydroxy groups were tolerated under the present catalytic systems. When diazo compound 1g (R’ = H) was employed as the substrate, none of the cyclization product was detected and the water insertion product 4g was obtained in 81% yield.
Table 2: Gold(I)-catalyzed cascade insertion/cyclization of water with various substituted phenyldiazoacetatesa.
|
|
|||
| Entry | Substrate | Yield of 2b | Yield of 3b |
|---|---|---|---|
| 1 |
1a |
2a, 70% |
3a, 15% |
| 2 |
1b |
2b, 64% |
3b, 31% |
| 3 |
1c |
2c, 67% |
3c, 22% |
| 4 |
1d |
2d, 82% |
3d, <10% |
| 5 |
1e |
2e, 69% |
3e, Trace |
| 6 |
1f |
2f, 75% |
3f, Trace |
| 7c |
1g |
2g, 0% |
3g, 0% |
aAll the reactions were carried out using 0.5 mmol phenyldiazoacetate 1 with 3 mol % of (IPr)AuCl catalyst in 2 mL solvent for 24 h. bIsolated yield. cOnly the water insertion product methyl 2-(2-ethynylphenyl)-2-hydroxyacetate (4g) was isolated as the major product in a yield of 81%.
A tentative mechanism for this gold(I)-catalyzed cascade insertion/cyclization is proposed in Scheme 2. Decomposition of diazo compound 1 by (IPr)AuCl generates gold carbene species A, which inserts into the O–H bond of H2O to form the chelating intermediate B. Subsequently, 6-endo-dig attack of the Au(I)-activated triple bond affords the vinylgold intermediate C, which is protonated to give the final product 2 with regeneration of the catalyst. This mechanism is supported by the fact that when 4a was subjected to the gold(I)-catalyzed reaction under identical conditions 2a and 3a were obtained in similar yields.
Conclusion
In summary, we have developed a cascade insertion/cyclization of water with o-acetylenyl-substituted phenyldiazoacetates catalyzed by a single Au(I) catalyst. This tandem process provides a novel and straightforward method to synthesize isochromene derivatives. Isochromene and its derivatives frequently occur as structural units in natural products and exhibit interesting biological activities such as antibiotic properties [39-51]. Moreover, this study further demonstrates the possibility to incorporate gold-catalyzed reaction of diazo compounds with various other gold-catalyzed transformations. Further studies to broaden the scope of these reactions are currently underway.
Experimental
General. For chromatography, 200–300 mesh silica gel (Qingdao, China) was employed. 1H NMR and 13C NMR spectra were recorded on Varian 300 or Bruker ARX 400 spectrometer in CDCl3 solution and chemical shifts are reported in parts per million (δ) relative to internal standard TMS (0 ppm). Mass spectra were obtained on a VG ZAB-HS mass spectrometer. Diazo compounds were prepared according to our previous reported procedures. Unless otherwise noted, materials obtained from commercial suppliers were used without further purification.
General procedure for the gold(I)-catalyzed cascade insertion/cyclization reaction. Distilled water (0.5 mL) was added to a solution of complex 1 (0.5 mmol) and (IPr)AuCl (3 mol %, 9.3 mg) in DMF (1.5 mL) at room temperature under a N2 atmosphere. The reaction mixture was stirred for 24 h at 80 °C. After the mixture cooled to room temperature, it was diluted with ether and water. The aqueous phase was extracted with Et2O (2 × 10 mL) and the combined organic extracts were washed with brine (1 × 10 mL), dried over Na2SO4, and concentrated. Purification by column chromatography on silica gel afforded products 2 and 3.
Ethyl 3-phenyl-1H-isochromene-1-carboxylate (2a). 1H NMR (400 MHz, CDCl3) δ 7.81 (d, J = 6.8 Hz, 2H), 7.41–7.35 (m, 3H), 7.28–7.20 (m, 3H), 7.09 (d, J = 7.6 Hz, 1H), 6.36 (s, 1H), 5.83 (s, 1H), 4.15 (m, 2H), 1.19 (t, J = 7.2 Hz, 3H); 13C NMR (100 MHz, CDCl3) δ 170.0, 153.0, 134.2, 130.6, 129.18, 129.16, 128.4, 126.7, 126.2, 126.0, 125.8, 124.1, 100.5, 76.6, 61.7, 14.2; MS (70 eV) m/z (%): 280 (29) [M+], 207 (100), 178 (47), 152 (8); HRMS–ESI (m/z): [M + H]+ calcd for C18H17O3, 281.1172; found, 281.1168.
Ethyl 3-benzylidene-1,3-dihydroisobenzofuran-1-carboxylate (3a). 1H NMR (400 MHz, CDCl3) δ 7.81 (d, J = 7.2 Hz, 2H), 7.75–7.55 (m, 1H), 7.53–7.51 (m, 1H), 7.41–7.32 (m, 4H), 7.18–7.17 (m, 1H), 6.03 (s, 1H), 6.00 (s, 1H), 4.33–4.23 (m, 2H), 1.31 (t, J = 7.2 Hz, 3H); 13C NMR (100 MHz, CDCl3) δ 168.8, 154.8, 136.8, 135.8, 134.6, 129.4, 129.1, 128.5, 128.4, 125.9, 122.3, 120.1, 97.9, 82.6, 62.0, 14.3; MS (70 eV) m/z (%): 280 (11) [M+], 264 (8), 207 (100), 191 (13), 178 (43); HRMS–ESI (m/z): [M + H]+ calcd for C18H17O3, 281.1172; found, 281.1166.
Methyl 3-(2-bromophenyl)-1H-isochromene-1-carboxylate (2b). 1H NMR (400 MHz, CDCl3) δ 7.89–7.86 (m, 1H), 7.64–7.62 (m, 1H), 7.39–7.35 (m, 1H), 7.30–7.19 (m, 4H), 7.11–7.09 (m, 1H), 6.27 (s, 1H), 5.87 (s, 1H), 3.76 (s, 3H); 13C NMR (100 MHz, CDCl3) δ 170.1, 151.5, 135.5, 133.5, 131.4, 130.1, 129.9, 129.1, 127.3, 127.2, 125.9, 125.5, 124.2, 106.0, 77.2, 52.5; MS (70 eV) m/z (%): 344 (41, 79Br) [M+], 285 (100), 206 (36), 178 (96), 151 (17); HRMS–ESI (m/z): [M + H]+ calcd for C17H14BrO3, 345.0120; found, 345.0125.
Methyl 3-(2-bromobenzylidene)-1,3-dihydroisobenzofuran-1-carboxylate (3b). 1H NMR (400 MHz, CDCl3) δ 8.36–8.34 (m, 1H), 7.66 (d, J = 8.0 Hz, 1H), 7.58–7.52 (m, 2H), 7.41–7.39 (m, 2H), 7.34–7.29 (m, 1H), 7.03–7.01 (m, 1H), 6.42 (s, 1H), 6.05 (s, 1H), 3.82 (s, 3H); 13C NMR (100 MHz, CDCl3) δ 168.9, 155.9, 136.6, 134.9, 134.2, 132.6, 130.0, 129.5, 129.4, 127.3, 127.0, 123.0, 122.1, 96.1, 82.6, 52.8; MS (70 eV) m/z (%): 344 (13, 79Br) [M+], 285 (100), 206 (32), 178 (67), 151 (14); HRMS–ESI (m/z): [M + H]+ calcd for C17H14BrO3, 345.0120; found, 345.0127.
Methyl 3-(3-bromophenyl)-1H-isochromene-1-carboxylate (2c). 1H NMR (400 MHz, CDCl3) δ 7.96–7.95 (m, 1H), 7.72–7.71 (m, 1H), 7.49–7.47 (m, 1H), 7.32–7.25 (m, 4H), 7.11–7.09 (m, 1H), 6.37 (s, 1H), 5.86 (s, 1H), 3.71 (s, 3H); 13C NMR (100 MHz, CDCl3) δ 170.1, 151.3, 136.1, 131.8, 129.8, 129.1, 128.6, 127.1, 126.2, 125.7, 124.2, 124.1, 122.6, 101.4, 76.2, 52.6; MS (70 eV) m/z (%): 344 (34, 79Br) [M+], 285 (100), 206 (35), 178 (82), 151 (14); HRMS–ESI (m/z): [M + H]+ calcd for C17H14BrO3, 345.0120; found, 345.0117.
Methyl 3-(3-bromobenzylidene)-1,3-dihydroisobenzofuran-1-carboxylate (3c). 1H NMR (400 MHz, CDCl3) δ 7.94–7.93 (m, 1H), 7.68–7.66 (m, 1H), 7.55–7.52 (m, 2H), 7.45–7.38 (m, 2H), 7.30–7.27 (m, 1H), 7.22–7.17 (m, 1H), 6.07 (s, 1H), 5.92 (s, 1H), 3.82 (s, 3H); 13C NMR (100 MHz, CDCl3) δ 168.9, 155.7, 137.8, 136.8, 134.0, 130.8, 129.8, 129.47, 129.44, 128.5, 126.7, 122.5, 122.2, 120.1, 96.4, 82.6, 52.8; MS (70 eV) m/z (%): 344 (28, 79Br) [M+], 285 (100), 206 (34), 178 (50), 151 (19); HRMS–ESI (m/z): [M + H]+ calcd for C17H14BrO3, 345.0120; found, 345.0126.
Methyl 3-p-tolyl-1H-isochromene-1-carboxylate (2d). 1H NMR (400 MHz, CDCl3) δ 7.69 (d, J = 8.4 Hz, 2H), 7.25–7.17 (m, 5H), 7.05 (d, J = 6.8 Hz, 1H), 6.31 (s, 1H), 5.83 (s, 1H), 3.67 (s, 3H), 2.36 (s, 3H); 13C NMR (100 MHz, CDCl3) δ 170.4, 153.1, 139.2, 131.4, 130.7, 129.17, 129.15, 126.5, 126.2, 125.76, 125.71, 123.9, 99.7, 76.5, 52.5, 21.4; MS (70 eV) m/z (%): 280 (17) [M+], 221 (100), 207 (7), 178 (27); HRMS–ESI (m/z): [M + H]+ calcd for C18H17O3, 287.1172; found, 281.1168.
Methyl 3-(2-hydroxyethyl)-1H-isochromene-1-carboxylate (2e). 1H NMR (400 MHz, CDCl3) δ 7.26–7.15 (m, 3H), 6.92 (d, J = 7.2 Hz, 1H), 5.74 (s, 1H), 5.70 (s, 1H), 4.09–4.07 (m, 1H), 3.78–3.64 (m, 2H), 3.69 (s, 3H), 2.51–2.47 (m, 2H); 13C NMR (100 MHz, CDCl3) δ 171.4, 154.0, 130.1, 129.3, 126.5, 126.4, 124.4, 123.2, 102.9, 76.1, 59.7, 52.9, 37.5; MS (70 eV) m/z (%): 234 (2) [M+], 192 (8), 175 (100), 145 (28), 133 (77); HRMS–ESI (m/z): [M + H]+ calcd for C13H15O4, 235.0964; found, 235.0960.
Methyl 3-butyl-1H-isochromene-1-carboxylate (2f). 1H NMR (400 MHz, CDCl3) δ 7.21–7.11 (m, 3H), 6.89 (d, J = 7.6 Hz, 1H), 5.67 (s, 1H), 5.58 (s, 1H), 3.68 (s, 3H), 2.77 (t , J = 7.6 Hz, 2H), 1.65–1.58 (m , 2H), 1.42–1.36 (m , 2H), 0.94 (t , J = 7.2 Hz, 2H); 13C NMR (100 MHz, CDCl3) δ 170.5, 157.3, 130.7, 129.0, 126.0, 124.7, 123.0, 99.9, 76.3, 52.4, 33.5, 28.6, 22.4, 14.0; MS (70 eV) m/z (%): 246 (12) [M+], 187 (100), 115 (16); HRMS–ESI (m/z): [M + H]+ calcd for C15H19O3, 247.1328; found, 247.1322.
Methyl 2-(2-ethynylphenyl)-2-hydroxyacetate (4g). 1H NMR (400 MHz, CDCl3) δ 7.53 (d, J = 7.2 Hz, 1H), 7.37–7.36 (m, 2H), 7.32–7.29 (m, 1H), 5.63 (d, J = 5.6 Hz, 1H), 3.75 (s, 3H), 3.59 (d, J = 5.2 Hz, 1H), 3.31 (s , 1H); 13C NMR (100 MHz, CDCl3) δ 174.0, 140.5, 133.4, 129.4, 128.5, 127.1, 121.5, 82.2, 81.3, 71.4, 53.2; MS (70 eV) m/z (%): 190 (100) [M+], 159 (69), 132 (57), 103 (53); HRMS–ESI (m/z): [M + H]+ calcd for C11H11O3, 191.0702; found, 191.0699.
Supporting Information
| Supporting Information File 1: 1H and 13C NMR spectra. | ||
| Format: PDF | Size: 959.0 KB | Download |
References
-
Davies, H. M. L.; Beckwith, R. E. J. Chem. Rev. 2003, 103, 2861–2904. doi:10.1021/cr0200217
Return to citation in text: [1] -
Ye, T.; McKervey, M. A. Chem. Rev. 1994, 94, 1091–1160. doi:10.1021/cr00028a010
Return to citation in text: [1] -
Doyle, M. P.; McKervey, M. A.; Ye, T. Modern Catalytic Methods for Organic Synthesis with Diazo Compounds; Wiley-Interscience: New York, 1998.
Return to citation in text: [1] [2] -
Zhang, Z.; Wang, J. Tetrahedron 2008, 64, 6577–6605. doi:10.1016/j.tet.2008.04.074
Return to citation in text: [1] -
Barluenga, J.; Rodríguez, F.; Fañanás, F. J.; Flórez, J. In Metal Carbenes in Organic Synthesis; Dötz, K. H., Ed.; Topics in Organometallic Chemistry, Vol. 13; Springer: Berlin, Germany, 2004; pp 59–122.
Return to citation in text: [1] -
Fructos, M. R.; Belderrain, T. R.; de Frémont, P.; Scott, N. M.; Nolan, S. P.; Díaz-Requejo, M. M.; Pérez, P. J. Angew. Chem., Int. Ed. 2005, 44, 5284–5288. doi:10.1002/anie.200501056
Return to citation in text: [1] [2] -
Flores, J. A.; Dias, H. V. R. Inorg. Chem. 2008, 47, 4448–4450. doi:10.1021/ic800373u
Return to citation in text: [1] [2] -
Ricard, L.; Gagosz, F. Organometallics 2007, 26, 4704–4707. doi:10.1021/om7006002
Return to citation in text: [1] -
Li, Z.; Ding, X.; He, C. J. Org. Chem. 2006, 71, 5876–5880. doi:10.1021/jo060016t
Return to citation in text: [1] -
Fañanás-Mastral, M.; Aznar, F. Organometallics 2009, 28, 666–668. doi:10.1021/om801146z
Return to citation in text: [1] -
Hashmi, A. S. K. Chem. Rev. 2007, 107, 3180–3211. doi:10.1021/cr000436x
Return to citation in text: [1] -
Li, Z.; Brouwer, C.; He, C. Chem. Rev. 2008, 108, 3239–3265. doi:10.1021/cr068434l
Return to citation in text: [1] -
Nolan, S. P. Acc. Chem. Res. 2011, 44, 91–100. doi:10.1021/ar1000764
Return to citation in text: [1] -
Arcadi, A. Chem. Rev. 2008, 108, 3266–3325. doi:10.1021/cr068435d
Return to citation in text: [1] -
Gorin, D. J.; Sherry, B. D.; Toste, F. D. Chem. Rev. 2008, 108, 3351–3378. doi:10.1021/cr068430g
Return to citation in text: [1] -
Díaz-Requejo, M. M.; Pérez, P. J. Chem. Rev. 2008, 108, 3379–3394. doi:10.1021/cr078364y
Return to citation in text: [1] -
Marion, N.; Nolan, S. P. Chem. Soc. Rev. 2008, 37, 1776–1782. doi:10.1039/B711132K
Return to citation in text: [1] -
Jiménez-Núñez, E.; Echavarren, A. M. Chem. Rev. 2008, 108, 3326–3350. doi:10.1021/cr0684319
Return to citation in text: [1] -
Soriano, E.; Marco-Contelles, J. Acc. Chem. Res. 2009, 42, 1026–1036. doi:10.1021/ar800200m
Return to citation in text: [1] -
Echavarren, A. M.; Nevado, C. Chem. Soc. Rev. 2004, 33, 431–436. doi:10.1039/B308768A
Return to citation in text: [1] -
Bunce, R. A. Tetrahedron 1995, 51, 13103–13159. doi:10.1016/0040-4020(95)00649-S
Return to citation in text: [1] -
Parsons, P. J.; Penkett, C. S.; Shell, A. J. Chem. Rev. 1996, 96, 195–206. doi:10.1021/cr950023+
Return to citation in text: [1] -
Fogg, D. E.; dos Santos, E. N. Coord. Chem. Rev. 2004, 248, 2365–2379. doi:10.1016/j.ccr.2004.05.012
Return to citation in text: [1] -
Wasilke, J.-C.; Obrey, S. J.; Baker, R. T.; Bazan, G. C. Chem. Rev. 2005, 105, 1001–1020. doi:10.1021/cr020018n
Return to citation in text: [1] -
Ajamian, A.; Gleason, J. L. Angew. Chem., Int. Ed. 2004, 43, 3754–3760. doi:10.1002/anie.200301727
Return to citation in text: [1] -
Louie, J.; Bielawski, C. W.; Grubbs, R. H. J. Am. Chem. Soc. 2001, 123, 11312–11313. doi:10.1021/ja016431e
Return to citation in text: [1] -
Evans, P. A.; Robinson, J. E. J. Am. Chem. Soc. 2001, 123, 4609–4610. doi:10.1021/ja015531h
Return to citation in text: [1] -
Mariampillai, B.; Alberico, D.; Bidau, V.; Lautens, M. J. Am. Chem. Soc. 2006, 128, 14436–14437. doi:10.1021/ja064742p
Return to citation in text: [1] -
Peng, C.; Chen, J.; Wang, J. Adv. Synth. Catal. 2008, 350, 2359–2364. doi:10.1002/adsc.200800249
Return to citation in text: [1] -
Dubé, P.; Toste, F. D. J. Am. Chem. Soc. 2006, 128, 12062–12063. doi:10.1021/ja064209+
Return to citation in text: [1] -
Kotera, A.; Uenishi, J.; Uemura, M. Tetrahedron Lett. 2010, 51, 1166–1169. doi:10.1016/j.tetlet.2009.12.052
Return to citation in text: [1] -
Bianchi, G.; Chiarini, M.; Marinelli, F.; Rossi, L.; Arcadi, A. Adv. Synth. Catal. 2010, 352, 136–142. doi:10.1002/adsc.200900668
Return to citation in text: [1] -
Godet, T.; Vaxelaire, C.; Michel, C.; Milet, A.; Belmont, P. Chem.–Eur. J. 2007, 13, 5632–5641. doi:10.1002/chem.200700202
Return to citation in text: [1] -
Yu, M.; Skouta, R.; Zhou, L.; Jiang, H.-f.; Yao, X.; Li, C.-J. J. Org. Chem. 2009, 74, 3378–3383. doi:10.1021/jo900079u
Return to citation in text: [1] -
Harkat, H.; Blanc, A.; Weibel, J.-M.; Pale, P. J. Org. Chem. 2008, 73, 1620–1623. doi:10.1021/jo702197b
Return to citation in text: [1] -
Hashmi, A. S. K.; Ramamurthi, T. D.; Rominger, F. Adv. Synth. Catal. 2010, 352, 971–975. doi:10.1002/adsc.201000011
Return to citation in text: [1] -
Belting, V.; Krause, N. Org. Lett. 2006, 8, 4489–4492. doi:10.1021/ol061751u
Return to citation in text: [1] -
Yao, X.; Li, C.-J. Org. Lett. 2006, 8, 1953–1955. doi:10.1021/ol060645p
Return to citation in text: [1] -
Bieber, B.; Nüske, J.; Ritzau, M.; Gräfe, U. J. Antibiot. 1998, 51, 381–382.
Return to citation in text: [1] -
Naruse, N.; Goto, M.; Watanabe, Y.; Terasawa, T.; Dobashi, K. J. Antibiot. 1998, 51, 545–552.
Return to citation in text: [1] -
Wang, W.; Breining, T.; Li, T.; Milbum, R.; Attardo, G. Tetrahedron Lett. 1998, 39, 2459–2462. doi:10.1016/S0040-4039(98)00287-1
and the references therein.
Return to citation in text: [1] -
Li, J.; Chin, E.; Lui, A. S.; Chen, L. Tetrahedron Lett. 2010, 51, 5937–5939. doi:10.1016/j.tetlet.2010.09.023
Return to citation in text: [1] -
Enomoto, T.; Girard, A.-L.; Yasui, Y.; Takemoto, Y. J. Org. Chem. 2009, 74, 9158–9164. doi:10.1021/jo901906b
Return to citation in text: [1] -
Wang, F.; Miao, Z.; Chen, R. Org. Biomol. Chem. 2009, 7, 2848–2850. doi:10.1039/b908313h
Return to citation in text: [1] -
Obika, S.; Kono, H.; Yasui, Y.; Yanada, R.; Takemoto, Y. J. Org. Chem. 2007, 72, 4462–4468. doi:10.1021/jo070615f
Return to citation in text: [1] -
Asao, N.; Nogami, T.; Takahashi, K.; Yamamoto, Y. J. Am. Chem. Soc. 2002, 124, 764–765. doi:10.1021/ja017366b
Return to citation in text: [1] -
Patil, N. T.; Yamamoto, Y. J. Org. Chem. 2004, 69, 5139–5142. doi:10.1021/jo049416b
Return to citation in text: [1] -
Nakamura, H.; Ohtaka, M.; Yamamoto, Y. Tetrahedron Lett. 2002, 43, 7631–7633. doi:10.1016/S0040-4039(02)01604-0
Return to citation in text: [1] -
Asao, N.; Chan, C. S.; Takahashi, K.; Yamamoto, Y. Tetrahedron 2005, 61, 11322–11326. doi:10.1016/j.tet.2005.09.012
Return to citation in text: [1] -
Patil, N. T.; Pahadi, N. K.; Yamamoto, Y. J. Org. Chem. 2005, 70, 10096–10098. doi:10.1021/jo051524q
Return to citation in text: [1] -
Yue, D.; Cà, N. D.; Larock, R. C. Org. Lett. 2004, 6, 1581–1584. doi:10.1021/ol049690s
Return to citation in text: [1]
| 1. | Davies, H. M. L.; Beckwith, R. E. J. Chem. Rev. 2003, 103, 2861–2904. doi:10.1021/cr0200217 |
| 2. | Ye, T.; McKervey, M. A. Chem. Rev. 1994, 94, 1091–1160. doi:10.1021/cr00028a010 |
| 3. | Doyle, M. P.; McKervey, M. A.; Ye, T. Modern Catalytic Methods for Organic Synthesis with Diazo Compounds; Wiley-Interscience: New York, 1998. |
| 4. | Zhang, Z.; Wang, J. Tetrahedron 2008, 64, 6577–6605. doi:10.1016/j.tet.2008.04.074 |
| 5. | Barluenga, J.; Rodríguez, F.; Fañanás, F. J.; Flórez, J. In Metal Carbenes in Organic Synthesis; Dötz, K. H., Ed.; Topics in Organometallic Chemistry, Vol. 13; Springer: Berlin, Germany, 2004; pp 59–122. |
| 7. | Flores, J. A.; Dias, H. V. R. Inorg. Chem. 2008, 47, 4448–4450. doi:10.1021/ic800373u |
| 6. | Fructos, M. R.; Belderrain, T. R.; de Frémont, P.; Scott, N. M.; Nolan, S. P.; Díaz-Requejo, M. M.; Pérez, P. J. Angew. Chem., Int. Ed. 2005, 44, 5284–5288. doi:10.1002/anie.200501056 |
| 6. | Fructos, M. R.; Belderrain, T. R.; de Frémont, P.; Scott, N. M.; Nolan, S. P.; Díaz-Requejo, M. M.; Pérez, P. J. Angew. Chem., Int. Ed. 2005, 44, 5284–5288. doi:10.1002/anie.200501056 |
| 7. | Flores, J. A.; Dias, H. V. R. Inorg. Chem. 2008, 47, 4448–4450. doi:10.1021/ic800373u |
| 8. | Ricard, L.; Gagosz, F. Organometallics 2007, 26, 4704–4707. doi:10.1021/om7006002 |
| 9. | Li, Z.; Ding, X.; He, C. J. Org. Chem. 2006, 71, 5876–5880. doi:10.1021/jo060016t |
| 10. | Fañanás-Mastral, M.; Aznar, F. Organometallics 2009, 28, 666–668. doi:10.1021/om801146z |
| 11. | Hashmi, A. S. K. Chem. Rev. 2007, 107, 3180–3211. doi:10.1021/cr000436x |
| 12. | Li, Z.; Brouwer, C.; He, C. Chem. Rev. 2008, 108, 3239–3265. doi:10.1021/cr068434l |
| 13. | Nolan, S. P. Acc. Chem. Res. 2011, 44, 91–100. doi:10.1021/ar1000764 |
| 14. | Arcadi, A. Chem. Rev. 2008, 108, 3266–3325. doi:10.1021/cr068435d |
| 15. | Gorin, D. J.; Sherry, B. D.; Toste, F. D. Chem. Rev. 2008, 108, 3351–3378. doi:10.1021/cr068430g |
| 16. | Díaz-Requejo, M. M.; Pérez, P. J. Chem. Rev. 2008, 108, 3379–3394. doi:10.1021/cr078364y |
| 17. | Marion, N.; Nolan, S. P. Chem. Soc. Rev. 2008, 37, 1776–1782. doi:10.1039/B711132K |
| 18. | Jiménez-Núñez, E.; Echavarren, A. M. Chem. Rev. 2008, 108, 3326–3350. doi:10.1021/cr0684319 |
| 19. | Soriano, E.; Marco-Contelles, J. Acc. Chem. Res. 2009, 42, 1026–1036. doi:10.1021/ar800200m |
| 20. | Echavarren, A. M.; Nevado, C. Chem. Soc. Rev. 2004, 33, 431–436. doi:10.1039/B308768A |
| 3. | Doyle, M. P.; McKervey, M. A.; Ye, T. Modern Catalytic Methods for Organic Synthesis with Diazo Compounds; Wiley-Interscience: New York, 1998. |
| 31. | Kotera, A.; Uenishi, J.; Uemura, M. Tetrahedron Lett. 2010, 51, 1166–1169. doi:10.1016/j.tetlet.2009.12.052 |
| 35. | Harkat, H.; Blanc, A.; Weibel, J.-M.; Pale, P. J. Org. Chem. 2008, 73, 1620–1623. doi:10.1021/jo702197b |
| 36. | Hashmi, A. S. K.; Ramamurthi, T. D.; Rominger, F. Adv. Synth. Catal. 2010, 352, 971–975. doi:10.1002/adsc.201000011 |
| 37. | Belting, V.; Krause, N. Org. Lett. 2006, 8, 4489–4492. doi:10.1021/ol061751u |
| 38. | Yao, X.; Li, C.-J. Org. Lett. 2006, 8, 1953–1955. doi:10.1021/ol060645p |
| 30. | Dubé, P.; Toste, F. D. J. Am. Chem. Soc. 2006, 128, 12062–12063. doi:10.1021/ja064209+ |
| 39. | Bieber, B.; Nüske, J.; Ritzau, M.; Gräfe, U. J. Antibiot. 1998, 51, 381–382. |
| 40. | Naruse, N.; Goto, M.; Watanabe, Y.; Terasawa, T.; Dobashi, K. J. Antibiot. 1998, 51, 545–552. |
| 41. |
Wang, W.; Breining, T.; Li, T.; Milbum, R.; Attardo, G. Tetrahedron Lett. 1998, 39, 2459–2462. doi:10.1016/S0040-4039(98)00287-1
and the references therein. |
| 42. | Li, J.; Chin, E.; Lui, A. S.; Chen, L. Tetrahedron Lett. 2010, 51, 5937–5939. doi:10.1016/j.tetlet.2010.09.023 |
| 43. | Enomoto, T.; Girard, A.-L.; Yasui, Y.; Takemoto, Y. J. Org. Chem. 2009, 74, 9158–9164. doi:10.1021/jo901906b |
| 44. | Wang, F.; Miao, Z.; Chen, R. Org. Biomol. Chem. 2009, 7, 2848–2850. doi:10.1039/b908313h |
| 45. | Obika, S.; Kono, H.; Yasui, Y.; Yanada, R.; Takemoto, Y. J. Org. Chem. 2007, 72, 4462–4468. doi:10.1021/jo070615f |
| 46. | Asao, N.; Nogami, T.; Takahashi, K.; Yamamoto, Y. J. Am. Chem. Soc. 2002, 124, 764–765. doi:10.1021/ja017366b |
| 47. | Patil, N. T.; Yamamoto, Y. J. Org. Chem. 2004, 69, 5139–5142. doi:10.1021/jo049416b |
| 48. | Nakamura, H.; Ohtaka, M.; Yamamoto, Y. Tetrahedron Lett. 2002, 43, 7631–7633. doi:10.1016/S0040-4039(02)01604-0 |
| 49. | Asao, N.; Chan, C. S.; Takahashi, K.; Yamamoto, Y. Tetrahedron 2005, 61, 11322–11326. doi:10.1016/j.tet.2005.09.012 |
| 50. | Patil, N. T.; Pahadi, N. K.; Yamamoto, Y. J. Org. Chem. 2005, 70, 10096–10098. doi:10.1021/jo051524q |
| 51. | Yue, D.; Cà, N. D.; Larock, R. C. Org. Lett. 2004, 6, 1581–1584. doi:10.1021/ol049690s |
| 29. | Peng, C.; Chen, J.; Wang, J. Adv. Synth. Catal. 2008, 350, 2359–2364. doi:10.1002/adsc.200800249 |
| 21. | Bunce, R. A. Tetrahedron 1995, 51, 13103–13159. doi:10.1016/0040-4020(95)00649-S |
| 22. | Parsons, P. J.; Penkett, C. S.; Shell, A. J. Chem. Rev. 1996, 96, 195–206. doi:10.1021/cr950023+ |
| 23. | Fogg, D. E.; dos Santos, E. N. Coord. Chem. Rev. 2004, 248, 2365–2379. doi:10.1016/j.ccr.2004.05.012 |
| 24. | Wasilke, J.-C.; Obrey, S. J.; Baker, R. T.; Bazan, G. C. Chem. Rev. 2005, 105, 1001–1020. doi:10.1021/cr020018n |
| 25. | Ajamian, A.; Gleason, J. L. Angew. Chem., Int. Ed. 2004, 43, 3754–3760. doi:10.1002/anie.200301727 |
| 26. | Louie, J.; Bielawski, C. W.; Grubbs, R. H. J. Am. Chem. Soc. 2001, 123, 11312–11313. doi:10.1021/ja016431e |
| 27. | Evans, P. A.; Robinson, J. E. J. Am. Chem. Soc. 2001, 123, 4609–4610. doi:10.1021/ja015531h |
| 28. | Mariampillai, B.; Alberico, D.; Bidau, V.; Lautens, M. J. Am. Chem. Soc. 2006, 128, 14436–14437. doi:10.1021/ja064742p |
| 32. | Bianchi, G.; Chiarini, M.; Marinelli, F.; Rossi, L.; Arcadi, A. Adv. Synth. Catal. 2010, 352, 136–142. doi:10.1002/adsc.200900668 |
| 33. | Godet, T.; Vaxelaire, C.; Michel, C.; Milet, A.; Belmont, P. Chem.–Eur. J. 2007, 13, 5632–5641. doi:10.1002/chem.200700202 |
| 34. | Yu, M.; Skouta, R.; Zhou, L.; Jiang, H.-f.; Yao, X.; Li, C.-J. J. Org. Chem. 2009, 74, 3378–3383. doi:10.1021/jo900079u |
© 2011 Zhou et al; licensee Beilstein-Institut.
This is an Open Access article under the terms of the Creative Commons Attribution License (http://creativecommons.org/licenses/by/2.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
The license is subject to the Beilstein Journal of Organic Chemistry terms and conditions: (http://www.beilstein-journals.org/bjoc)