Abstract
Derivatives of an antifungal agent that targets the β-(1,3)-D-glucan synthase, papulacandin D, were synthesized and tested for activity. The papulacandin D structure contains a challenging benzannulated spiroketal unit, which is introduced in a palladium-catalyzed cross-coupling reaction of a glycal silanolate and an aryl iodide followed by an oxidative spiroketalization. Four different variants were made, differing in the nature of the acyl side chain with respect to the length, and in the number and stereochemistry of the double bonds. Moderate biological activity was observed for the derivatives with a side chain based on palmitic acid and linoleic acid.
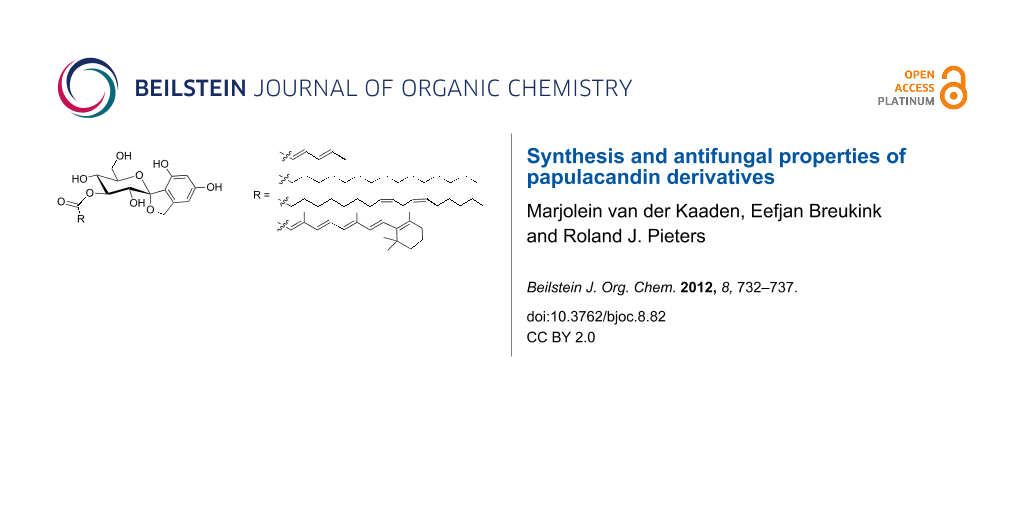
Graphical Abstract
Introduction
In recent years, a steady increase in the incidence of opportunistic fungal infections in immunocompromised patients has been observed [1]. Treatment failure is frequent and mortality is unacceptably high in high-risk patients [2]. Reasons for this include delayed diagnosis, toxicity and low bioavailability of current drugs, and the development of antifungal drug resistance [1]. Clearly, there is a need for new antifungal therapeutics. The papulacandins are a series of naturally occurring antifungal agents whose isolation and characterization were initially reported by Traxler and co-workers in 1977 (Figure 1) [3,4]. They contain a benzannulated spiroketal unit, which has been the signature of a wide series of bioactive natural products [5] and has inspired ample synthetic activity [5-8]. The papulacandins A–E were isolated from the fermentation broths of Papularia sphaerosperma [3]. They block the synthesis of β-(1,3)-D-glucan by inhibition of β-(1,3)-D-glucan synthase [9-12]. The β-(1,3)-D-glucan is an integral and essential component of the fungal cell wall [13] and the dominant glucan in the cell wall of most medically important fungi, and therefore it is a target that is being actively pursued [14]. The papulacandins have demonstrated a very high specific activity against several yeasts, but they are largely inactive against filamentous fungi, bacteria and protozoa [3]. Recently, in a direct comparison, papulacandin B was shown to be superior in some aspects compared to the drug caspofungin and a few drug candidates [15]. Its interesting biological activity has stimulated the search for new members of the papulacandin family, and several new compounds that are structurally related have been isolated, such as chaetiacandein [16,17], L-687,781 [18,19], Mer-WF3010 [20,21], BU-4794F [22], fusacandins [23,24], saricandin and PF-1042 [25], corynecandin [26,27] and the F-10748 series [28]. These structures vary mostly with respect to the two partially unsaturated acyl chains on the sugars. Small changes in these fatty acid tails have only small effects on the activity. However, more drastic changes in these tails or the lack of one of the tails drastically reduce activity, in comparison with the most active member of this family: papulacandin B. For the papulacandins it was shown that the substituted galactose is not essential for activity, since papulacandin D is still active, but it does increase the potency. Apparently, the situation is different for the related disaccharide saricandan carrying different acyl chains, for which the monosaccharide analogue was not active [29]. Removing the galactose acyl chain from papulacandin B results in material that can still inhibit the target (in a spheroplasts glucan synthesis assay) but cannot reach the target site of C. albicans [12]. The same is true for the hydrogenated form of papulacandin D [12]. Clearly some form of unsaturation is needed to maintain biological activity, but it is not known to what extent unsaturation (and alkylation and/or hydroxylation) needs to be present for the compound to reach the target site.
The papulacandins represent a synthetic challenge and as such the class has stimulated numerous synthetic studies. So far, only the total synthesis of papulacandin D, the simplest member of the family, has been published. This was reported by two different research groups [30-33] and additionally a number of different synthetic routes [34-38] to the spiroketal core structure have been published, including a hetero Diels–Alder reaction [39], a palladium(0)-catalyzed coupling [40-45] and a condensation reaction [46,47]. We herein report our efforts on the synthesis of papulacandin derivatives by total synthesis and the evaluation of their antifungal properties. The synthetic approach is largely based on the Denmark route [31] with a few notable deviations.
Results
The synthesis started with the anomeric aromatic group, i.e., the eventual spiroketal group of the glucose, to be introduced as an aryliodide with an ortho hydroxymethylene as in 12. The commercially available 3,5-dihydroxybenzoic acid (1) was esterified, followed by benzylation of the aromatic hydroxy groups and reduction of the ester to the primary alcohol to give 7 (Scheme 1). This compound was iodinated by using N-iodosuccinimide (NIS), and finally the free hydroxy group was protected with a pivaloyl group to give aryl iodide 12. Derivatives 13 and 14, providing alternative core structures, were prepared similarly.
Scheme 1: (a) H2SO4, MeOH, reflux, 18 h, 3: 98%; (b) BnBr, K2CO3, acetone, reflux, 18 h; (c) LiAlH4, THF, rt, 10–30 min, 7: 92% over three steps, 8: 65% over three steps; (d) NIS, CHCl3, rt, 18 h, 9 and 11 quant. or I2, CF3COOAg, CHCl3, rt, 1 h, 10: 96%; (e) pivaloyl chloride, pyridine, CH2Cl2, rt, 3–18 h, 12: 95% over 2 steps, 13: 96%, 14: 98%.
Scheme 1: (a) H2SO4, MeOH, reflux, 18 h, 3: 98%; (b) BnBr, K2CO3, acetone, reflux, 18 h; (c) LiAlH4, THF, rt,...
The glucose moiety at the core of the structure, as present in papulacandin D, was approached from glycal 15. The aim was to obtain a silanol such as 22 for coupling to an aryl iodide in the palladium-catalyzed cross-coupling reactions of silanolates, as pioneered and applied by the Denmark group (Scheme 2) [48]. The synthesis started with the saponification of 15, followed by the protection of O-4 and O-6 by using di-tert-butylsilyl bis(trifluoromethanesulfonate). Subsequent protection of O-3 with TES-Cl, gave the fully protected glucal 18. According to Denmark et al. [31], lithiation and silylation of 18 should give 20, but in our hands a more complicated mixture resulted. We concluded that under our conditions deprotonation of the protecting group (protons α to silicon) may be competitive with the desired α-lithiation next to oxygen. Use of the more substituted TIPS silyl protecting group in 19 indeed solved this problem and yielded 21 in almost quantitative yield. Additional test reactions confirmed this trend as the corresponding 3,4,6-tri-O-(triisopropylsilyl)-D-glucal (52) yielded a single product after lithiation (Supporting Information File 1) and the 3,4,6-tri-O-(tert-butyldimethylsilyl)-D-glucal gave a mixture of products. Finally, compound 21 was oxidized to give silanol building block 22.
Scheme 2: (a) NaOMe in MeOH, MeOH, rt, 1 h, 96%; (b) (t-Bu)2Si(OTf)2, pyridine, DMF, −40 °C to rt, 2 h, 90%; (c) TESCl, pyridine, CH2Cl2, rt, 18 h, 93%; (d) TIPSCl, imidazole, DMF, 60 °C, 2 d, 74%; (e) t-BuLi, Me2SiHCl, Et2O, −78 °C to rt, 3 h, 20: no yield, 21: 99%; (f) [RuCl2(p-cymene)]2, H2O, benzene/CH3CN, rt, 4 h, 95%.
Scheme 2: (a) NaOMe in MeOH, MeOH, rt, 1 h, 96%; (b) (t-Bu)2Si(OTf)2, pyridine, DMF, −40 °C to rt, 2 h, 90%; ...
With building blocks 13 and 22 in hand, the palladium-catalyzed cross-coupling reaction was performed by using Pd2(dba)3 to give 23 (Scheme 3). Then the pivaloyl ester was selectively cleaved by using DIBAL-H, followed by oxidative spiroketalization under basic conditions to give the α- and β-anomers 29, which were separable using column chromatography. However this was not necessary: The β-anomer could readily be converted into the more stable α-anomer 32 by using a solution of 0.1 M HCl in chloroform. Similarly, the related iodides 14 and 15 performed well in this sequence yielding 33 and 34.
Scheme 3: (a) Pd2(dba)3∙CHCl3, NaOt-Bu, toluene, 50 °C, 6–20 h, 23: 72%, 24: 79%, 25: 62%; (b) DiBAL-H, CH2Cl2, −78 °C to rt, 0.5–1 h, 26: 86%, 27: 70%, 28: 75%; (c) m-CPBA, NaHCO3, CH2Cl2, 0 °C to rt, 2–2.5 h; (d) HCl, CHCl3, rt, 1 h, 32: 91% over two steps, 33: 83% over two steps, 34: 86% over two steps.
Scheme 3: (a) Pd2(dba)3∙CHCl3, NaOt-Bu, toluene, 50 °C, 6–20 h, 23: 72%, 24: 79%, 25: 62%; (b) DiBAL-H, CH2Cl2...
The first step in the synthesis of the mimics of papulacandin D was debenzylation of 32, since these groups cannot be removed in the presence of olefines in the final product side chain (Scheme 4). The liberated hydroxy groups were reprotected with MOM groups by using MOM-Cl to give 36. Then the glucose O(3) needed to be selectively deprotected for the incorporation of the fatty acid tail. Unfortunately, this was not possible from 36 because of the use of the more stable TIPS group in this synthesis. Therefore, the cyclic silyl protecting group was first removed by using TBAHF in THF, followed by the deprotection of the TIPS group with TBAF and reintroduction of the cyclic silyl protecting group, giving 39. To circumvent this problem a silanol building block analogue of 22 bearing three TIPS protecting groups was also used (Supporting Information File 1).
Scheme 4: (a) Pd/C, NaHCO3, H2, THF, rt, 1.5 h, 98%; (b) MOMCl, DiPEA, DMAP, CH2Cl2, rt, 4 d, 78%; (c) TBAHF, THF, rt, 2 d, 84%; (d) TBAF∙3H2O, THF, rt, 3 d; (e) t-Bu2Si(OTf)2, pyridine, DMF, −40°C to rt, 2 h, 84% over two steps; (f) RCOOH, NEt3, 2,4,6-trichlorobenzoyl chloride, toluene, rt, 1 h, 39, DMAP, toluene, rt, 3 h, 40: 89%, 41: quant., 42: 92%, 43: 90%; (g) TBAHF, THF, rt, 2–3 d, 44: 25%, 45: 85%, 46: 86%, 47: 39%; (h) Dowex 50 × 8, MeOH, 50 °C, 20 h 48: 13%, 50: 43%, 51: 7% or Sc(OTf)3, 1,3-propanediol, CH3CN, 50 °C, 3 h, 49: 9%, isolated yields after preparative HPLC and lyophilization.
Scheme 4: (a) Pd/C, NaHCO3, H2, THF, rt, 1.5 h, 98%; (b) MOMCl, DiPEA, DMAP, CH2Cl2, rt, 4 d, 78%; (c) TBAHF,...
At this stage, four commercially available side-chain acids, comprising variations in their degree of unsaturation, length and branching, were coupled: sorbic acid (sor), palmitic acid (pal), linoleic acid (lin) and trans-retinoic (ret) acid. Their coupling by 2,4,6-trichlorobenzoyl chloride to 39 gave the desired compounds 40–43 in good yields. Following this the cyclic disilyl protecting group was removed by TBAHF yielding 44–47. Finally, removal of the MOM group proved to be a challenge. The use of Dowex (50 × 8) acidic resin in dry MeOH gave the most consistent results, but also the use of Sc(OTf)3 in dry acetonitrile in the presence of 1,3-propanediol [49] yielded the deprotected product. The final compounds were purified by preparative HPLC. Due to some degradation during deprotection, the final yields were low. For 51 no clear NMR spectra could be obtained, possibly caused by aggregation. This was not a problem for the other three compounds.
The final products were tested for inhibition of Candida albicans. No inhibition was observed for the all-trans-retinoic acid derivative 51 and the sorbic acid derivative 48. The MIC for the linoleic acid derivative 50 was measured as 100 μg/mL. The saturated palmitic acid derivative 49 was slightly more potent with an MIC of 88 μg/mL.
Discussion
Starting from glycal the synthesis of papulacandin D derivatives was undertaken with an initial focus on the acyl side chain, a clearly sensitive area for activity. The synthesis followed the general approach of Denmark et al. [31] with the palladium-catalyzed cross-coupling reactions of silanolates as its main feature. In our hands the reaction between glycal 18 and t-BuLi gave a mixture of products, possibly due to competing deprotonation of the hydrogens α to the Si in the TES group. Moving to the more substituted TIPS group at this position gave a clean reaction and allowed us to proceed, albeit with a more complicated endgame. It should be pointed out that the unplanned synthetic route eventually taken here due to the troublesome lithiation of glycal 18, led us to compound 37. This compound opens the way to a total synthesis of disaccharides such as the papulacandins A–C. Selective protection of the hydroxy group of C(6) followed by glycosylation of the hydroxy group at C(4), should accomplish this.
In our biological assay we did not see any activity for the sorbic acid derivative 48, suggesting that this tail may be too short to be effective. The same was true for the retinoic acid derivative 51, but its identity was also uncertain due to instability. Some activity was observed for linoleic acid derivative 50, which contains two cis double bonds in contrast to the four trans double bonds of papulacandin D. The highest activity was observed for the saturated palmitic acid derivative 49, whose tail was of the same length as the one from papulacandin D. The fact that we observed some activity with 49 is encouraging, since this was not the case for the hydrogenated form of papulacandin D [12], although that compound was able to block the target in vitro. This could be the case for the compounds described here as well, but this was not investigated. In summary, we synthesized a number of papulacandin D derivatives with differing acyl side chains. Even though the compounds were moderately active at best it is clear that the side chains are of crucial importance. The synthetic route taken will allow future variation of the aromatic core and will extend the synthesis to the more potent disaccharide papulacandins.
Supporting Information
| Supporting Information File 1: Synthetic procedures, the biological assay procedure and spectral data. | ||
| Format: PDF | Size: 2.8 MB | Download |
References
-
Mathew, B. P.; Nath, M. ChemMedChem 2009, 4, 310–323. doi:10.1002/cmdc.200800353
Return to citation in text: [1] [2] -
Pound, M. W.; Townsend, M. L.; Dimondi, V.; Wilson, D.; Drew, R. H. Med. Mycol. 2011, 49, 561–580. doi:10.3109/13693786.2011.560197
Return to citation in text: [1] -
Traxler, P.; Gruner, J.; Auden, J. A. L. J. Antibiot. 1977, 30, 289–296. doi:10.7164/antibiotics.30.289
Return to citation in text: [1] [2] [3] -
Traxler, P.; Fritz, H.; Fuhrer, H.; Richter, W. J. J. Antibiot. 1980, 33, 967–978. doi:10.7164/antibiotics.33.967
Return to citation in text: [1] -
Sperry, J.; Wilson, Z. E.; Rathwell, D. C. K.; Brimble, M. A. Nat. Prod. Rep. 2010, 27, 1117–1137. doi:10.1039/b911514p
Return to citation in text: [1] [2] -
Liu, G.; Wurst, J. M.; Tan, D. S. Org. Lett. 2009, 11, 3670–3673. doi:10.1021/ol901437f
Return to citation in text: [1] -
Yamamoto, Y.; Yamashita, K.; Hotta, T.; Hashimoto, T.; Kikuchi, M.; Nishiyama, H. Chem.–Asian J. 2007, 2, 1388–1399. doi:10.1002/asia.200700077
Return to citation in text: [1] -
Subrahmanyam, A. V.; Palanichamy, K.; Kaliappan, K. P. Chem.–Eur. J. 2010, 16, 8545–8556. doi:10.1002/chem.201000482
Return to citation in text: [1] -
Baguley, B. C.; Römmele, G.; Gruner, J.; Wehrli, W. Eur. J. Biochem. 1979, 97, 345–351. doi:10.1111/j.1432-1033.1979.tb13120.x
Return to citation in text: [1] -
Pérez, P.; Varona, R.; Garcia-Acha, I.; Duran, A. FEBS Lett. 1981, 129, 249–252. doi:10.1016/0014-5793(81)80176-7
Return to citation in text: [1] -
Varona, R.; Pérez, P.; Durán, A. FEMS Microbiol. Lett. 1983, 20, 243–247. doi:10.1111/j.1574-6968.1983.tb00125.x
Return to citation in text: [1] -
Römmele, G.; Traxler, P.; Wehrli, W. J. Antibiot. 1983, 36, 1539–1542. doi:10.7164/antibiotics.36.1539
Return to citation in text: [1] [2] [3] [4] -
Debono, M.; Gordee, R. S. Annu. Rev. Microbiol. 1994, 48, 471–497. doi:10.1146/annurev.mi.48.100194.002351
Return to citation in text: [1] -
Hector, R. F.; Bierer, D. E. Exp. Opin. Ther. Pat. 2011, 21, 1597–1610. doi:10.1517/13543776.2011.603899
Return to citation in text: [1] -
Martins, I. M.; Cortés, J. C. G.; Muñoz, J.; Belén Moreno, M.; Ramos, M.; Clemente-Ramos, J. A.; Durán, A.; Ribas, J. C. J. Biol. Chem. 2011, 286, 3484–3496. doi:10.1074/jbc.M110.174300
Return to citation in text: [1] -
Komori, T.; Yamashita, M.; Tsurumi, Y.; Kohsaka, M. J. Antibiot. 1985, 38, 455–459. doi:10.7164/antibiotics.38.455
Return to citation in text: [1] -
Komori, T.; Itoh, Y. J. Antibiot. 1985, 38, 544–546. doi:10.7164/antibiotics.38.544
Return to citation in text: [1] -
VanMiddlesworth, F.; Omstead, M. N.; Schmatz, D.; Bartizal, K.; Fromtling, R.; Bills, G.; Nollstadt, K.; Honeycutt, S.; Zweerink, M.; Garrity, G.; Wilson, K. E. J. Antibiot. 1991, 44, 45–51. doi:10.7164/antibiotics.44.45
Return to citation in text: [1] -
VanMiddlesworth, F.; Dufresne, C.; Smith, J.; Wilson, K. E. Tetrahedron 1991, 47, 7563–7568. doi:10.1016/S0040-4020(01)88280-6
Return to citation in text: [1] -
Kaneto, R.; Chiba, H.; Agematu, H.; Shibamoto, N.; Yoshioka, T.; Nishida, H.; Okamoto, R. J. Antibiot. 1993, 46, 247–250. doi:10.7164/antibiotics.46.247
Return to citation in text: [1] -
Chiba, H.; Kaneto, R.; Agematu, H.; Shibamoto, N.; Yoshioka, T.; Nishida, H.; Okamoto, R. J. Antibiot. 1993, 46, 356–358. doi:10.7164/antibiotics.46.356
Return to citation in text: [1] -
Aoki, M.; Andoh, T.; Ueki, T.; Masuyoshi, S.; Sugawara, K.; Oki, T. J. Antibiot. 1993, 46, 952–960. doi:10.7164/antibiotics.46.952
Return to citation in text: [1] -
Jackson, M.; Frost, D. J.; Karwowski, J. P.; Humphrey, P. E.; Dahod, S. K.; Choi, W. S.; Brandt, K.; Malmberg, L.-H.; Rasmussen, R. R.; Scherr, M. H.; Flamm, R. K.; Kadam, S.; McAlpine, J. B. J. Antibiot. 1995, 48, 608–613. doi:10.7164/antibiotics.48.608
Return to citation in text: [1] -
Hochlowski, J. E.; Whittern, D. N.; Buko, A.; Alder, L.; McAlpine, J. B. J. Antibiot. 1995, 48, 614–618. doi:10.7164/antibiotics.48.614
Return to citation in text: [1] -
Chen, R. H.; Tennant, S.; Frost, D.; O’Beirne, M. J.; Karwowski, J. P.; Humphrey, P. E.; Malmberg, L.-H.; Choi, W.; Brandt, K. D.; West, P.; Kadam, S. K.; Clement, J. J.; McAlpine, J. B. J. Antibiot. 1996, 49, 596–598. doi:10.7164/antibiotics.49.596
Return to citation in text: [1] -
Gunawardana, G.; Rasmussen, R. R.; Scherr, M.; Frost, D.; Brandt, K. D.; Choi, W.; Jackson, M.; Karwowski, J. P.; Sunga, G.; Malmberg, L.-H.; West, P.; Chen, R. H.; Kadam, S.; Clement, J. J.; McAlpine, J. B. J. Antibiot. 1997, 50, 884–886. doi:10.7164/antibiotics.50.884
Return to citation in text: [1] -
Gunawardana, G. P.; Frost, D.; Jackson, M.; Karwowski, J. P.; Rasmussen, R. R. Antifungal corynecandin. U.S. Patent 5,863,773, Jan 26, 1999.
Return to citation in text: [1] -
Ohyama, T.; Iwadate-Kurihara, Y.; Hosoya, T.; Ishikawa, T.; Miyakoshi, S.; Hamano, K.; Inukai, M. J. Antibiot. 2002, 55, 758–763. doi:10.7164/antibiotics.55.758
Return to citation in text: [1] -
Hamdouchi, C.; Sanchez-Martinez, C. Synthesis 2001, 833–840. doi:10.1055/s-2001-13399
Return to citation in text: [1] -
Barrett, A. G. M.; Peña, M.; Willardsen, J. A. J. Org. Chem. 1996, 61, 1082–1100. doi:10.1021/jo951895e
Return to citation in text: [1] -
Denmark, S. E.; Regens, C. S.; Kobayashi, T. J. Am. Chem. Soc. 2007, 129, 2774–2776. doi:10.1021/ja070071z
Return to citation in text: [1] [2] [3] [4] -
Kobayashi, T.; Regens, C. S.; Denmark, S. E. J. Synth. Org. Chem., Jpn. 2008, 66, 616–628. doi:10.5059/yukigoseikyokaishi.66.616
Return to citation in text: [1] -
Denmark, S. E.; Kobayashi, T.; Regens, C. S. Tetrahedron 2010, 66, 4745–4759. doi:10.1016/j.tet.2010.03.093
Return to citation in text: [1] -
McDonald, F. E.; Zhu, H. Y. H.; Holmquist, C. R. J. Am. Chem. Soc. 1995, 117, 6605–6606. doi:10.1021/ja00129a030
Return to citation in text: [1] -
Parker, K. A.; Georges, A. T. Org. Lett. 2000, 2, 497–499. doi:10.1021/ol991346l
Return to citation in text: [1] -
Balachari, D.; O’Doherty, G. A. Org. Lett. 2000, 2, 863–866. doi:10.1021/ol0000253
Return to citation in text: [1] -
Balachari, D.; O’Doherty, G. A. Org. Lett. 2000, 2, 4033–4036. doi:10.1021/ol006662a
Return to citation in text: [1] -
Ahmed, M. M.; O’Doherty, G. A. Tetrahedron Lett. 2005, 46, 4151–4155. doi:10.1016/j.tetlet.2005.04.073
Return to citation in text: [1] -
Danishefski, S.; Phillips, G.; Ciufolini, M. Carbohydr. Res. 1987, 171, 317–327. doi:10.1016/S0008-6215(00)90895-4
Return to citation in text: [1] -
Rosenblum, S. B.; Bihovsky, R. J. Am. Chem. Soc. 1990, 112, 2746–2748. doi:10.1021/ja00163a042
Return to citation in text: [1] -
Friesen, R. W.; Sturino, C. F. J. Org. Chem. 1990, 55, 2572–2574. doi:10.1021/jo00296a005
Return to citation in text: [1] -
Friesen, R. W.; Sturino, C. F. J. Org. Chem. 1990, 55, 5808–5810. doi:10.1021/jo00310a004
Return to citation in text: [1] -
Dubois, E.; Beau, J.-M. J. Chem. Soc., Chem. Commun. 1990, 1191–1192. doi:10.1039/C39900001191
Return to citation in text: [1] -
Dubois, E.; Beau, J.-M. Tetrahedron Lett. 1990, 31, 5165–5168. doi:10.1016/S0040-4039(00)97832-8
Return to citation in text: [1] -
Dubois, E.; Beau, J.-M. Carbohydr. Res. 1992, 223, 157–167. doi:10.1016/0008-6215(92)80014-R
Return to citation in text: [1] -
Schmidt, R. R.; Frick, W. Tetrahedron 1988, 44, 7163–7169. doi:10.1016/S0040-4020(01)86084-1
Return to citation in text: [1] -
Czernecki, S.; Perlat, M. C. J. Org. Chem. 1991, 56, 6289–6292. doi:10.1021/jo00022a014
Return to citation in text: [1] -
Denmark, S. E.; Baird, J. D. Chem.–Eur. J. 2006, 12, 4954–4963. doi:10.1002/chem.200600034
Return to citation in text: [1] -
Oriyama, T.; Watahiki, T.; Kobayashi, Y.; Hirano, H.; Suzuki, T. Synth. Commun. 2001, 31, 2305–2311. doi:10.1081/SCC-100104830
Return to citation in text: [1]
| 46. | Schmidt, R. R.; Frick, W. Tetrahedron 1988, 44, 7163–7169. doi:10.1016/S0040-4020(01)86084-1 |
| 47. | Czernecki, S.; Perlat, M. C. J. Org. Chem. 1991, 56, 6289–6292. doi:10.1021/jo00022a014 |
| 31. | Denmark, S. E.; Regens, C. S.; Kobayashi, T. J. Am. Chem. Soc. 2007, 129, 2774–2776. doi:10.1021/ja070071z |
| 48. | Denmark, S. E.; Baird, J. D. Chem.–Eur. J. 2006, 12, 4954–4963. doi:10.1002/chem.200600034 |
| 1. | Mathew, B. P.; Nath, M. ChemMedChem 2009, 4, 310–323. doi:10.1002/cmdc.200800353 |
| 5. | Sperry, J.; Wilson, Z. E.; Rathwell, D. C. K.; Brimble, M. A. Nat. Prod. Rep. 2010, 27, 1117–1137. doi:10.1039/b911514p |
| 20. | Kaneto, R.; Chiba, H.; Agematu, H.; Shibamoto, N.; Yoshioka, T.; Nishida, H.; Okamoto, R. J. Antibiot. 1993, 46, 247–250. doi:10.7164/antibiotics.46.247 |
| 21. | Chiba, H.; Kaneto, R.; Agematu, H.; Shibamoto, N.; Yoshioka, T.; Nishida, H.; Okamoto, R. J. Antibiot. 1993, 46, 356–358. doi:10.7164/antibiotics.46.356 |
| 3. | Traxler, P.; Gruner, J.; Auden, J. A. L. J. Antibiot. 1977, 30, 289–296. doi:10.7164/antibiotics.30.289 |
| 4. | Traxler, P.; Fritz, H.; Fuhrer, H.; Richter, W. J. J. Antibiot. 1980, 33, 967–978. doi:10.7164/antibiotics.33.967 |
| 22. | Aoki, M.; Andoh, T.; Ueki, T.; Masuyoshi, S.; Sugawara, K.; Oki, T. J. Antibiot. 1993, 46, 952–960. doi:10.7164/antibiotics.46.952 |
| 1. | Mathew, B. P.; Nath, M. ChemMedChem 2009, 4, 310–323. doi:10.1002/cmdc.200800353 |
| 16. | Komori, T.; Yamashita, M.; Tsurumi, Y.; Kohsaka, M. J. Antibiot. 1985, 38, 455–459. doi:10.7164/antibiotics.38.455 |
| 17. | Komori, T.; Itoh, Y. J. Antibiot. 1985, 38, 544–546. doi:10.7164/antibiotics.38.544 |
| 2. | Pound, M. W.; Townsend, M. L.; Dimondi, V.; Wilson, D.; Drew, R. H. Med. Mycol. 2011, 49, 561–580. doi:10.3109/13693786.2011.560197 |
| 18. | VanMiddlesworth, F.; Omstead, M. N.; Schmatz, D.; Bartizal, K.; Fromtling, R.; Bills, G.; Nollstadt, K.; Honeycutt, S.; Zweerink, M.; Garrity, G.; Wilson, K. E. J. Antibiot. 1991, 44, 45–51. doi:10.7164/antibiotics.44.45 |
| 19. | VanMiddlesworth, F.; Dufresne, C.; Smith, J.; Wilson, K. E. Tetrahedron 1991, 47, 7563–7568. doi:10.1016/S0040-4020(01)88280-6 |
| 13. | Debono, M.; Gordee, R. S. Annu. Rev. Microbiol. 1994, 48, 471–497. doi:10.1146/annurev.mi.48.100194.002351 |
| 3. | Traxler, P.; Gruner, J.; Auden, J. A. L. J. Antibiot. 1977, 30, 289–296. doi:10.7164/antibiotics.30.289 |
| 31. | Denmark, S. E.; Regens, C. S.; Kobayashi, T. J. Am. Chem. Soc. 2007, 129, 2774–2776. doi:10.1021/ja070071z |
| 9. | Baguley, B. C.; Römmele, G.; Gruner, J.; Wehrli, W. Eur. J. Biochem. 1979, 97, 345–351. doi:10.1111/j.1432-1033.1979.tb13120.x |
| 10. | Pérez, P.; Varona, R.; Garcia-Acha, I.; Duran, A. FEBS Lett. 1981, 129, 249–252. doi:10.1016/0014-5793(81)80176-7 |
| 11. | Varona, R.; Pérez, P.; Durán, A. FEMS Microbiol. Lett. 1983, 20, 243–247. doi:10.1111/j.1574-6968.1983.tb00125.x |
| 12. | Römmele, G.; Traxler, P.; Wehrli, W. J. Antibiot. 1983, 36, 1539–1542. doi:10.7164/antibiotics.36.1539 |
| 15. | Martins, I. M.; Cortés, J. C. G.; Muñoz, J.; Belén Moreno, M.; Ramos, M.; Clemente-Ramos, J. A.; Durán, A.; Ribas, J. C. J. Biol. Chem. 2011, 286, 3484–3496. doi:10.1074/jbc.M110.174300 |
| 12. | Römmele, G.; Traxler, P.; Wehrli, W. J. Antibiot. 1983, 36, 1539–1542. doi:10.7164/antibiotics.36.1539 |
| 3. | Traxler, P.; Gruner, J.; Auden, J. A. L. J. Antibiot. 1977, 30, 289–296. doi:10.7164/antibiotics.30.289 |
| 31. | Denmark, S. E.; Regens, C. S.; Kobayashi, T. J. Am. Chem. Soc. 2007, 129, 2774–2776. doi:10.1021/ja070071z |
| 5. | Sperry, J.; Wilson, Z. E.; Rathwell, D. C. K.; Brimble, M. A. Nat. Prod. Rep. 2010, 27, 1117–1137. doi:10.1039/b911514p |
| 6. | Liu, G.; Wurst, J. M.; Tan, D. S. Org. Lett. 2009, 11, 3670–3673. doi:10.1021/ol901437f |
| 7. | Yamamoto, Y.; Yamashita, K.; Hotta, T.; Hashimoto, T.; Kikuchi, M.; Nishiyama, H. Chem.–Asian J. 2007, 2, 1388–1399. doi:10.1002/asia.200700077 |
| 8. | Subrahmanyam, A. V.; Palanichamy, K.; Kaliappan, K. P. Chem.–Eur. J. 2010, 16, 8545–8556. doi:10.1002/chem.201000482 |
| 14. | Hector, R. F.; Bierer, D. E. Exp. Opin. Ther. Pat. 2011, 21, 1597–1610. doi:10.1517/13543776.2011.603899 |
| 49. | Oriyama, T.; Watahiki, T.; Kobayashi, Y.; Hirano, H.; Suzuki, T. Synth. Commun. 2001, 31, 2305–2311. doi:10.1081/SCC-100104830 |
| 26. | Gunawardana, G.; Rasmussen, R. R.; Scherr, M.; Frost, D.; Brandt, K. D.; Choi, W.; Jackson, M.; Karwowski, J. P.; Sunga, G.; Malmberg, L.-H.; West, P.; Chen, R. H.; Kadam, S.; Clement, J. J.; McAlpine, J. B. J. Antibiot. 1997, 50, 884–886. doi:10.7164/antibiotics.50.884 |
| 27. | Gunawardana, G. P.; Frost, D.; Jackson, M.; Karwowski, J. P.; Rasmussen, R. R. Antifungal corynecandin. U.S. Patent 5,863,773, Jan 26, 1999. |
| 23. | Jackson, M.; Frost, D. J.; Karwowski, J. P.; Humphrey, P. E.; Dahod, S. K.; Choi, W. S.; Brandt, K.; Malmberg, L.-H.; Rasmussen, R. R.; Scherr, M. H.; Flamm, R. K.; Kadam, S.; McAlpine, J. B. J. Antibiot. 1995, 48, 608–613. doi:10.7164/antibiotics.48.608 |
| 24. | Hochlowski, J. E.; Whittern, D. N.; Buko, A.; Alder, L.; McAlpine, J. B. J. Antibiot. 1995, 48, 614–618. doi:10.7164/antibiotics.48.614 |
| 25. | Chen, R. H.; Tennant, S.; Frost, D.; O’Beirne, M. J.; Karwowski, J. P.; Humphrey, P. E.; Malmberg, L.-H.; Choi, W.; Brandt, K. D.; West, P.; Kadam, S. K.; Clement, J. J.; McAlpine, J. B. J. Antibiot. 1996, 49, 596–598. doi:10.7164/antibiotics.49.596 |
| 39. | Danishefski, S.; Phillips, G.; Ciufolini, M. Carbohydr. Res. 1987, 171, 317–327. doi:10.1016/S0008-6215(00)90895-4 |
| 40. | Rosenblum, S. B.; Bihovsky, R. J. Am. Chem. Soc. 1990, 112, 2746–2748. doi:10.1021/ja00163a042 |
| 41. | Friesen, R. W.; Sturino, C. F. J. Org. Chem. 1990, 55, 2572–2574. doi:10.1021/jo00296a005 |
| 42. | Friesen, R. W.; Sturino, C. F. J. Org. Chem. 1990, 55, 5808–5810. doi:10.1021/jo00310a004 |
| 43. | Dubois, E.; Beau, J.-M. J. Chem. Soc., Chem. Commun. 1990, 1191–1192. doi:10.1039/C39900001191 |
| 44. | Dubois, E.; Beau, J.-M. Tetrahedron Lett. 1990, 31, 5165–5168. doi:10.1016/S0040-4039(00)97832-8 |
| 45. | Dubois, E.; Beau, J.-M. Carbohydr. Res. 1992, 223, 157–167. doi:10.1016/0008-6215(92)80014-R |
| 30. | Barrett, A. G. M.; Peña, M.; Willardsen, J. A. J. Org. Chem. 1996, 61, 1082–1100. doi:10.1021/jo951895e |
| 31. | Denmark, S. E.; Regens, C. S.; Kobayashi, T. J. Am. Chem. Soc. 2007, 129, 2774–2776. doi:10.1021/ja070071z |
| 32. | Kobayashi, T.; Regens, C. S.; Denmark, S. E. J. Synth. Org. Chem., Jpn. 2008, 66, 616–628. doi:10.5059/yukigoseikyokaishi.66.616 |
| 33. | Denmark, S. E.; Kobayashi, T.; Regens, C. S. Tetrahedron 2010, 66, 4745–4759. doi:10.1016/j.tet.2010.03.093 |
| 34. | McDonald, F. E.; Zhu, H. Y. H.; Holmquist, C. R. J. Am. Chem. Soc. 1995, 117, 6605–6606. doi:10.1021/ja00129a030 |
| 35. | Parker, K. A.; Georges, A. T. Org. Lett. 2000, 2, 497–499. doi:10.1021/ol991346l |
| 36. | Balachari, D.; O’Doherty, G. A. Org. Lett. 2000, 2, 863–866. doi:10.1021/ol0000253 |
| 37. | Balachari, D.; O’Doherty, G. A. Org. Lett. 2000, 2, 4033–4036. doi:10.1021/ol006662a |
| 38. | Ahmed, M. M.; O’Doherty, G. A. Tetrahedron Lett. 2005, 46, 4151–4155. doi:10.1016/j.tetlet.2005.04.073 |
| 12. | Römmele, G.; Traxler, P.; Wehrli, W. J. Antibiot. 1983, 36, 1539–1542. doi:10.7164/antibiotics.36.1539 |
| 12. | Römmele, G.; Traxler, P.; Wehrli, W. J. Antibiot. 1983, 36, 1539–1542. doi:10.7164/antibiotics.36.1539 |
| 28. | Ohyama, T.; Iwadate-Kurihara, Y.; Hosoya, T.; Ishikawa, T.; Miyakoshi, S.; Hamano, K.; Inukai, M. J. Antibiot. 2002, 55, 758–763. doi:10.7164/antibiotics.55.758 |
| 29. | Hamdouchi, C.; Sanchez-Martinez, C. Synthesis 2001, 833–840. doi:10.1055/s-2001-13399 |
© 2012 van der Kaaden et al; licensee Beilstein-Institut.
This is an Open Access article under the terms of the Creative Commons Attribution License (http://creativecommons.org/licenses/by/2.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
The license is subject to the Beilstein Journal of Organic Chemistry terms and conditions: (http://www.beilstein-journals.org/bjoc)