Abstract
Intramolecular inverse electron demand cycloadditions of isatin-derived 1,2,4-triazines with acetylenic dienophiles tethered by amidations or transesterifications proceed in excellent yields to produce lactam- or lactone-fused α-carbolines. Beginning with various isatins and alkynyl dienophiles, a pilot-scale library of eighty-eight α-carbolines was prepared by using this robust methodology for biological evaluation.
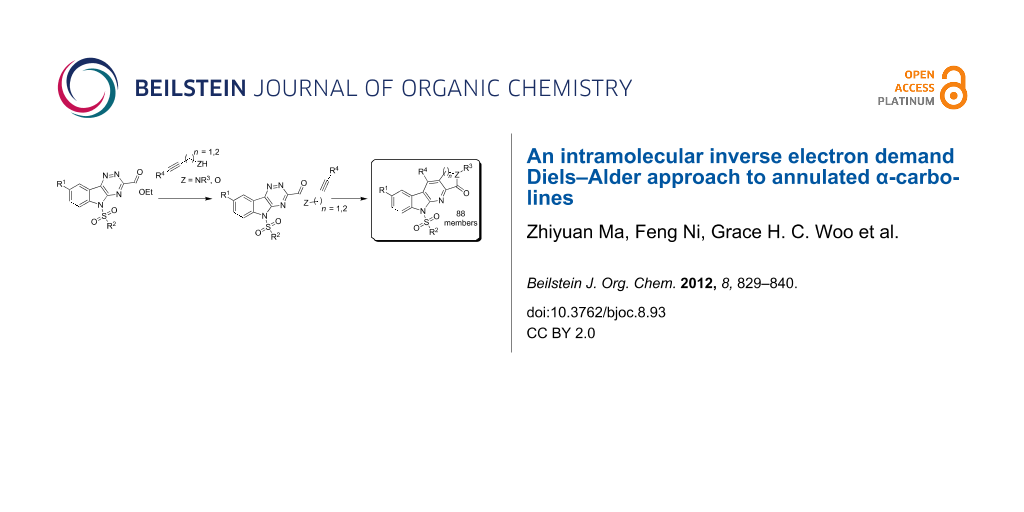
Graphical Abstract
Introduction
In comparison with the well-known β-carbolines, α-carboline alkaloids are quite rare, and only a few natural products isolated to date contain this pyrido[2,3-b]indole (α-carboline, 1, Figure 1) core. The most prominent examples are grossularine-1 (2) and grossularine-2 (3), which are marine cytotoxic agents that were isolated from the tunicate Dendrodoa grossularia (Stylidae) [1,2], and desmethylgrossularine-1 from tunicate Polycarpa aurata [3]. Other natural α-carbolines include mescengricin (3), an inhibitor of L-glutamate excitotoxicity in neutrons, isolated from Streptomyces griseoflavus [4], and cryptotackieine (4) [5], also known as neocryptolepine [6], isolated from roots of the West African plant Cryptolepis sanguinolenta [7]. Cryptotackieine, a member of the indolo[2,3-b]quinoline class of heterocycles [8], has been shown to be a strong inhibitor of Plasmodium falciparum growth [9]. 2-Amino-α-carbolines have also been identified as mutagens produced in the pyrolysis of proteins [10,11] as well as the pyrolysis of tryptophan [12]. Isoeudistomin U, isolated from the ascidian Lissoclinum fragile, was originally reported to have an α-carboline skeleton [13], but this assignment was later shown to be incorrect [14].
Figure 1: Natural products with α-carboline subunits.
Figure 1: Natural products with α-carboline subunits.
Given their isomeric relationship to β-carbolines, α-carbolines have, unsurprisingly, attracted attention from synthetic chemists for a long time [15], and more recently from medicinal chemists [16]. Synthetic members of this class were shown to have a wide range of activities, including topoisomerase II inhibition [17], and 4-amino-α-carbolines have also been shown to possess anxiolytic properties by stabilization of the open chloride channel [18,19]. Relatively few patents have been granted on the medicinal use of α-carbolines, particularly in comparison to β-carbolines, with recent applications including use as antiviral agents [20], inhibitors of ApoB-100-associated lipoprotein production for cholesterol lowering [21], and more recently, as inhibitors of CDK1 kinase as potential anticancer agents [22]. This later filing has triggered investigations into α-carbolines as potential multikinase inhibitors [23].
Existing synthetic approaches to α-carbolines are numerous and often independent of a specific target, but are instead methodology oriented. Exceptions include those efforts to target the grossularines [24-30] and the more popular cryptotackieine [31-35].
The first reported synthesis of an α-carboline by Robinson in 1924 [15,36] proceeded through the acid-catalyzed decomposition of 1-(2-pyridyl)benzotriazole, a modification of the Graebe–Ullmann carbazole synthesis, which closes the indole ring. This procedure was improved upon and exploited for decades [37-40]. Later, reversing the roles of the benzene and pyridine rings, with the former as the nucleophile and latter as electrophile through the diazonium salt, led to improved yields in the indole ring closure [41], as did microwave promotion of the original methodology [42]. Nitrene insertion chemistry of appropriately substituted 3-arylpyridines also found application [43,44], and likewise falls into the category of indole ring closure onto an existing substituted pyridine, though in this case with the formation of the C–N bond. More recently, the group of Cuny has reported two strategies that exploit cross-couplings to prepare anilinopyridines, with final indole ring closure occurring by either a second cross-coupling [45] or a photocyclization [46]. The former, sequential cross-coupling strategy closely follows the previous work by Queguiner [47]. A Fischer indole synthesis route through the 2-pyridylhydrazone of cyclohexanone, catalyzed by PPA, followed by dehydrogenation over Pd–C has also been reported for the preparation of the unsubstituted α-carboline [48], but no other successful applications of this strategy have appeared in the literature.
A more common strategy to α-carbolines closes the pyridine ring onto an existing indole framework. In general, applications of this construction sequence fall into two major camps, namely condensations routed through a 2-aminoindole synthon [49-56], and those targeting pyridine closures onto a 2-oxindole [57,58]. In addition, intramolecular Hartwig–Buchwald cross-coupling onto a 2-bromoindole, with formation of the pyridine was reported by Dodd [59], and C–N bond closures in presumed nitrogen-radical processes have also been reported by Narasaka [60,61]. Electrocyclic ring closures forming the pyridine ring from 2-aminoindole-derived intermediates are also known [62-64]. In addition, cycloadditions of 3-vinyl-7-azaindole have also been used to close the pyridine ring [65], as have intramolecular dipolar cycloadditions of 2-azidoindole derivatives [66], and intramolecular cycloadditions of carbodiimides [67,68].
We had envisioned that a library of annulated α-carboline structures 6 could be prepared by the intramolecular inverse electron demand Diels–Alder reaction (IEDDA) of isatin-derived 1,2,4-triazines 7 with tethered electron-rich dienophiles (Scheme 1) [69]. Inverse electron demand Diels–Alder cycloadditions employing electron-deficient heteroaromatic azadienes are well-established for the synthesis of heterocyclic compounds, and have been previously applied to the synthesis of α-carbolines. Intramolecular IEDDA chemistry to prepare α-carbolines employing 2-N-(o-alkynylanilino)pyrimidine systems have been successful in a limited number of examples [70,71]. Hoornaert and co-workers have also employed pyrazinones as dienes in IEDDA reactions to prepare α-carbolines [72].
Scheme 1: Retrosynthetic inverse electron Diels–Alder approach to α-carbolines.
Scheme 1: Retrosynthetic inverse electron Diels–Alder approach to α-carbolines.
Though the existing synthetic routes can readily produce various α-carbolines, the intramolecular IEDDA approach outlined in Scheme 1 has several advantages from the perspective of library synthesis. First, easy access exists to a wide selection of commercially available or easily synthesized starting materials, i.e., isatins, propargylic and homopropargylic amines and alcohols. Second, three easily modifiable diversification sites, R1, R2 and R4, can be built into the core α-carboline structure. Furthermore, construction of the annulated lactam or lactone as the fourth ring would be easily accomplished by the IEDDA reactions, thereby adding two more diversification points in the form of R3 and the lactam/lactone ring size. A particular goal in this work was to establish the reaction chemistry and scope in order to build a library of α-carbolines for biological screening. The targeted α-carbolines 6 (X = O) are similar to those prepared by Dodd by a rather lengthy route, but with a transposed carbonyl group on the lactone ring [59].
One consideration in this design was the electron donation from the indole nitrogen into the isatin-derived triazine ring of 9, which would result in an elevated LUMO of the triazinyl azadiene, and thereby inhibit the desired cycloaddition. Thus, it was anticipated that an electron-withdrawing group R2, which could also serve as a diversification point, would be needed on the indole nitrogen.
Results and Discussion
Feasibility studies began with isatin-derived 1,2,4-triazine 9a (9, R1 = H) [73,74], which was easily prepared by the condensation of isatin (10, R1= H) with ethyl oxaloamidrazonate [75-77] (11) in quantitative yield (Scheme 2). The first step in the cyclocondensation was accomplished by stirring in ethanol at rt for 12 h and heating under reflux for 20 min, after which the solvent was removed and the cyclocondensation completed by heating under reflux in bromobenzene (bp 156 °C) for 24 h. The two-step condensation with different solvents was needed to optimize the triazine formation. Sulfonylation of the indole nitrogen also proceeded routinely to give triazine 8a (R1 = H, R2 = p-Tol), and served two purposes. As noted, the reduction of electron donation from this nitrogen into the triazine ring was thought to be important for the subsequent cycloaddition to proceed, as was shown to be correct in later studies. Furthermore, sulfonylation greatly improved the solubility of the triazines 8 in organic solvents in comparison to 9, which showed only limited solubility in dichloromethane, chloroform, THF, toluene, methanol and acetone. Starting with other 5-substituted isatin derivatives, analogous triazines were similarly prepared in good yields (84–92%, Table 1). 5-Nitro- and 5-carboxamidoisatins also readily participated in the cyclocondensation with the oxaloamidrazonate 11 to form the corresponding triazines (9, R1 = NO2, CONH2), but due to the electron withdrawing nature of the isatin substituents, subsequent sulfonylations were not successful (not shown).
Scheme 2: Condensation of isatins with ethyl oxaloamidrazonate to form triazines.
Scheme 2: Condensation of isatins with ethyl oxaloamidrazonate to form triazines.
Table 1: Preparation of N-protected isatin-derived 1,2,4-triazines 8.a
|
|
||||
| R2 | R1 = H | R1 = Me | R1 = MeO | R1 = F |
|---|---|---|---|---|
| p-MePh | 8a, 88% | 8b, 92% | 8c, 89% | 8d, 85% |
| p-MeOPh | 8e, 90% | 8f, 87% | 8g, 84% | 8h, 88% |
| Ph | 8i, 90% | 8j, 88% | 8k, 87% | 8l, 91% |
aIsolated yield over two steps (Scheme 2).
The intermolecular IEDDA reactions of 8a with various electron-rich dienophiles, including ethyl ethynyl ether, the enamines (E)-1-N-propenylpyrrolidine, 1-pyrrolidino-1-cyclohexene, and 2-(dimethylamino)imidazole [78-80], all of which have been shown to react quite well with triazines or tetrazines in intermolecular IEDDA cycloaditions, were investigated. However, 8a showed no reactivity with any of these dienophiles under any conditions, further confirming the importance of the proposed intramolecular strategy for preparing α-carbolines.
Lewis acid catalyzed amidation of 8a with methyl propargyl amine (12) gave the cycloaddition precursor 13a in excellent yield when Al(Me)3 was used as catalyst (Scheme 3, Table 2) [81-84]. However, the most convenient procedure for library protocols employed 1.2 equiv of Zr(Ot-Bu)4 [85]. Weinreb amidation with Al(Me)3 also gave excellent yields, but required the extra step of first mixing Al(Me)3 and the amine, then cannulating this amine–AlMe3 complex into the triazine solution in order to avoid ketonization of the ester [86]. Other Lewis acids, MgCl2, Mg(OTf)2, Zn(OTf)2, Yb(OTf)3 and Sc(OTf)3, were not successful in catalyzing the amidation. Stoichiometric amounts of catalyst were required for the amidation, presumably due to the product itself sequestering the catalyst and preventing efficient turnover.
Scheme 3: Amidation of triazine ester 8a.
Scheme 3: Amidation of triazine ester 8a.
Table 2: Lewis acid catalysis of amidation of triazine 8a.a
| Entry | Catalyst | Loading (equiv) | Solvent | Yield (%) |
|---|---|---|---|---|
| 1b | Al(Me)3 | 1.5 | DCM | >95 |
| 2 | Zr(Ot-Bu)4 | 0.5 | toluene | 60 |
| 3 | Zr(Ot-Bu)4 | 1.2 | toluene | 90 |
| 4c | Zr(Ot-Bu)4 | 1.2 | DCM | >95 |
aAll reactions were carried out at rt for 20 h unless otherwise noted; isolated yield. bMix catalyst and amine, then transfer the mixture of the amine–aluminum complex into a triazine solution by cannula. cReaction time was 16 h.
The intramolecular cycloaddition of 13a, the alkyne-tethered triazine, was studied under various conditions (Scheme 4, Table 3). Ultimately, it was found that the IEDDA reaction proceeded smoothly under microwave irradiation, in diglyme (120 °C, 20 min; Table 3, entry 3) to give the γ-lactam annulated α-carboline 14a in quantitative yield. The microwave reaction conditions were preferred over the more traditional heating (Table 3, entry 1) due to the shorter reaction time. Attempts to lower the temperature and/or shorten the reaction time led to lower yields (Table 3, entries 2, 4 and 5). Little reaction occurred in toluene under microwave irradiation (Table 3, entry 6) unless silicon carbide chips were added as a microwave facilitator (Table 3, entry 7) [87].
Scheme 4: Microwave-promoted IEDDA reaction of isatin derived triazines.
Scheme 4: Microwave-promoted IEDDA reaction of isatin derived triazines.
Table 3: IEDDA reaction conditions of 13a to give cycloadduct 14a.a
| Entry | Solvent | Temperature (°C) | Time | Yield |
|---|---|---|---|---|
| 1b | diglyme | 164 | 7 h | >95% |
| 2 | diglyme | 100 | 20 min | 40% |
| 3 | diglyme | 120 | 20 min | >95% |
| 4 | diglyme | 120 | 10 min | 70% |
| 5 | diglyme | 160 | 10 min | 85% |
| 6 | toluene | 160 | 30 min | trace |
| 7c | toluene/Si–C | 160 | 30 min | 60% |
aIEDDA reactions were carried out under microwave irradiation unless otherwise noted. All yields were determined by UPLC unless otherwise noted. bHeating in oil bath. cSi–C chip was added to the reaction under microwave irradiation. Isolated yield.
Upon further exploration of the reaction conditions, it was discovered that the amidation/cycloaddition sequence could be accomplished in one pot (Scheme 5). The Zr(IV)-catalyzed amidation was thus accomplished in tetrahydrofuran (16 h, rt), then the reaction mixture was heated under reflux at 65 °C for another 24 h to yield cycloadduct 14a. This one-pot procedure gave a yield (89%) that was comparable to the overall yield (94%) of the corresponding two-step sequence, with isolation of the intermediate amide. Control experiments were carried out to determine the possible catalytic role of Zr(Ot-Bu)4 in the cycloaddition. In two side-by-side reactions, 13a, with and without Zr(Ot-Bu)4 catalyst, was heated to 65 °C. The two reactions required the same amount of time (24 h) to achieve complete conversions, indicating that the presence of amidation catalyst Zr(Ot-Bu)4 in the one-pot two-step sequence had no effect on the cycloaddition of 13a. For the library synthesis, the two-step sequence was adopted in order to isolate the triazine intermediates 13, which can also serve as library members.
Scheme 5: One-pot amidation/cycloaddition of triazine ester 8a.
Scheme 5: One-pot amidation/cycloaddition of triazine ester 8a.
With the two-step sequence optimized for the preparation of 14a, the scope of this chemistry was then probed with other alkynyl amines. The amidations all proceeded in high yields under the optimized conditions with Zr(Ot-Bu)4 as catalyst. The cycloaddition precursors were then subjected to the optimized microwave-promoted cycloadditions to give the final cycloadducts 14 (Scheme 6 and Table 4).
Scheme 6: Amidation/cycloaddition forming α-carbolines 14.
Scheme 6: Amidation/cycloaddition forming α-carbolines 14.
Table 4: Amidation-IEDDA sequence to α-carbolines 14 from triazine 8a.a
| Entry | Amine | Cycloaddition precursor 13 (yield) | Cycloadduct 14 (yield)b |
|---|---|---|---|
| 1 |
|
13a (98%) |
14a (96%) |
| 2c |
|
13b (90%) |
14b (83%) |
| 3d |
|
13c (89%) |
14c (91%) |
| 4 |
|
13d (96%) |
14d (98%) |
| 5 |
|
13e (96%) |
14e (97%) |
| 6e |
|
13f (95%) |
14f (93%) |
| 7f |
|
13g (93%) |
14g (95%) |
| 8 |
|
13h (95%) | no reaction |
aAmidation reactions were carried out in DCM, with 1.2 equiv of amine and 1.2 equiv of Zr(Ot-Bu)4 at rt for 16 h, 0.25 M. IEDDA reactions were carried out in diglyme under microwave irradiation at 120 °C for 20 min, unless otherwise noted. All IEDDA reaction concentrations were 0.1 M. bIsolated yields. cIEDDA in DMF at 160 °C for 30 min. dIEDDA in diglyme at 160 °C for 30 min. eIEDDA in diglyme at 120 °C for 40 min, 0.1 M. fIEDDA in diglyme at 160 °C for 30 min.
Tertiary propargyl amides 13a, 13d, and 13e with terminal alkyne dienophiles all showed comparable reactivity (Table 4, entry 1, 4, and 5), under the optimized cycloaddition conditions (diglyme, microwave irradiation, 120 °C, 20 min), producing the desired α-carbolines in excellent yields (96–98%). However, propargyl amide 13f with the internal alkyne (Table 4, entry 6) required a longer reaction time (40 min) for the cycloaddition to be completed to good yield (93%). Increasing the tether length between the alkyne group and the amide linkage also decreased the reactivity (Table 4, entry 7). The cycloaddition of tertiary propargyl amide 13g to produce the δ-lactam 14g required higher temperatures (160 °C versus 120 °C) to proceed in high yield, still in 30 min.
In contrast to the tertiary amides, the secondary amides 13b and 13c (Table 4, entry 2 and 3) showed no or very little reaction under the standard microwave conditions. The reason for this lack of reactivity was thought to be strong intramolecular hydrogen bonding, which inhibits the rotation of the alkyne group to the proper position for the cycloaddition to occur (Scheme 7).
Scheme 7: Intramolecular hydrogen bonding prevents IEDDA cycloaddition of 14b.
Scheme 7: Intramolecular hydrogen bonding prevents IEDDA cycloaddition of 14b.
The NMR spectra also supported this hypothesis: While all tertiary amides showed the presence of two rotamers in a 1:1 ratio in CDCl3, the secondary amides 13b and 13c showed only a single rotamer in the NMR spectra. In the 1H NMR spectrum for 13b, a very slowly exchanging proton (48 h for complete exchange) at δ 8.254 supported the presence of a single conformation with strong intramolecular hydrogen bonding. When the cycloaddition of 13b was run in DMF instead of diglyme (160 °C, 20 min), the cycloadditions proceeded in good yield to give the desired cycloadduct 14b (83%; Table 4, entry 3). Presumably the significantly greater capabilities of DMF to accept hydrogen bonds in comparison to diglyme help to disrupt the intramolecular hydrogen bonding and enable the cycloaddition to proceed. When 13c, with the gem dimethyl substituted propargylic carbon, was run in diglyme at a higher temperature (160 °C, 30 min), the cycloaddition proceeded in good yield to give the desired product 14c (91%; Table 4, entry 3), presumably aided by the Thorpe–Ingold effect [88-90].
To confirm the importance of the electron-withdrawing group on the original indole nitrogen position, the reactivity of amide 9a (R1 = H) was also examined (Scheme 8). As expected, no cycloaddition was observed under any conditions, presumably due to the greater electron donation from the indole nitrogen, which elevates the LUMO of the triazine ring, thereby preventing the desired cycloaddition.
Scheme 8: Preparation of unprotected triazine 15, and its lack of reactivity in cycloadditions.
Scheme 8: Preparation of unprotected triazine 15, and its lack of reactivity in cycloadditions.
In addition to the amide linkage of the alkyne dienophiles, transesterification of 8a with alkynyl alcohols led to tethered alkynyl esters as cycloaddition precursors 17a and 17b in good yields (Scheme 9). Various catalysts for the transesterification reaction were screened. Boronic acid [91] and indium(III) iodide [92] yielded no transesterification product with propargyl alcohol, while Ti(OiPr)4 [93] gave only a trace of the desired product, with mostly detosylation resulting. Otera’s catalyst [94] proved to be optimal, giving cycloaddition precursors 17a and 17b in excellent yields (84% and 87%, respectively).
Scheme 9: Transesterification and subsequent cycloaddition of 17a.
Scheme 9: Transesterification and subsequent cycloaddition of 17a.
Cycloaddition precursors 17a and 17b showed a much lower reactivity in the cycloadditions in comparison to the amides 13. For ester 17a, after microwave irradiation at 160 °C for 2 h, the desired cycloadduct 17a with the annulated γ-lactone was produced in good yield (80%). In contrast, 17b, with the longer tether (n = 2) failed to produce the desired δ-lactone under any conditions. A sharp decrease in reactivity in intramolecular IEDDA reactions of alkynes tethered to 1,2,4-triazines upon progression from 5- to 6-membered ring annulations was previously noted by Taylor [95-98], and has been ascribed to the greater entropy loss for the larger rings [99]. Indeed, it has been estimated that the effective molarity for 5-membered ring closures can be as high as 1,000-fold greater in comparison to 6-membered ring cyclizations [100].
Using this optimized two-step amidation-IEDDA reaction-sequence methodology, an 88-membered pilot-scale library of α-carbolines was prepared by using triazine analogues 8 and alkynyl amines (Table 5). The crude library was analyzed by UPLC. The average yield of the library was 94%, with 90% of the library members produced in yields greater than 85%. Purification by LC–MS produced the final library.
Table 5: Library synthesis matrix.a
|
|
||||||||||||
| R3 ↓ | R2 (p-) → | R1 = H | R1 = Me | R1 = OMe | R1 = F | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| MeOPh | MePh | Ph | MePh | MeOPh | Ph | MeOPh | Ph | MeOPh | MePh | Ph | ||
|
|
100 | 100 | 100 | 50 | 100 | 50 | 90 | 72 | 100 | 100 | 85 | |
|
|
100 | 94b | 100 | 100 | 100 | 66 | 100 | 100 | 99 | 100 | 100 | |
|
|
97 | 94b | 100 | 77 | 98 | 90 | 100 | 100 | 100 | 98 | 100 | |
|
|
40 | 100 | 97 | 98 | 90 | 85 | 95 | 97 | 80 | 93 | 96 | |
|
|
100 | 100 | 100 | 100 | 100 | 95 | 100 | 100 | 100 | 100 | 100 | |
|
|
100 | 100 | 100 | 100 | 100 | 100 | 100 | 100 | 100 | 100 | 100 | |
|
|
65 | 100 | 95 | 100 | 100 | 92 | 100 | 100 | 100 | 100 | 100 | |
|
|
–c | 100 | 100 | 100 | –c | 56 | –c | –c | 94 | 100 | –c | |
aUPLC yields against starting materials. bIsolated yields. cMembers were not prepared due to the limited amount of cyclohexyl propargylamine.
Conclusion
Isatin-derived 1,2,4-triazines have proven to be excellent heteroaromatic azadienes for intramolecular inverse electron demand Diels–Alder reactions with tethered alkynyl dienophiles. These cycloadditions led to lactam-annulated α-carbolines in excellent yields under microwave assistance. The scope of the chemistry was probed by using various alkynyl amines and alkynyl alcohols, and by variation of the tether length between the aminoalkyne and the triazine. The triazines with ester linkages showed significantly less reactivity in cycloadditions compared to those with amide linkages. The longer tether length also led to a decrease in cycloaddition reactivity. This chemistry was subsequently applied to a library synthesis, producing a focused library of eighty-eight members. Diversity was introduced by using a combination of various substituted isatin-derived triazines, with various sulfonylations of the indole nitrogen, and propargyl amine derivatives as dienophiles.
Experimental
General procedure A, preparation of isatin-derived triazines 9: Freshly prepared ethyl oxalamidrazonate (11) [76] was dissolved in anhydrous EtOH (0.1 M) and the isatin (1.0 equiv) was added at rt under stirring. The reaction mixture was stirred at rt for 12 h, and then heated under reflux for 20 min. After removal of the EtOH in vacuo, the residue was dissolved in anhydrous bromobenzene (0.2 M) and refluxed for 24 h. After removal of the solvent in vacuo, the residue was dried by the addition and evaporation of toluene three times, and used directly in the next step without any further purification.
General procedure B, preparation of sulfonamides 8: The isatin-derived triazine 9 was suspended in THF (0.25 M) and triethylamine (2.0 equiv) was added to the solution at rt. The reaction mixture was stirred at rt for 30 min until dissolution was completed, then the sulfonyl chloride (2.0 equiv) was added at rt, and the reaction mixture was stirred at rt for 7 h. After removal of the solvent in vacuo, the residue was purified by flash chromatography to yield the desired sulfonylated triazine 8.
Representative example: Ethyl 5-[(4-methylbenzene)sulfonyl]-5H-[1,2,4]triazino[5,6-b]indole-3-carboxylate (8a). According to general procedures A and B, beginning with isatin (350 mg, 2.38 mmol, 1.0 equiv) and p-toluenesulfonyl chloride (907 mg, 4.76 mmol, 2.0 equiv). Purification by flash chromatography, Rf 0.68 (hexanes/EtOAc, 1:1) gave 8a (827 mg, 2.09 mmol, 88% yield) as a yellow solid: mp 145–146 °C; IR (neat): 2982, 1742, 1379, 1191, 1180, 671, 579 cm−1; 1H NMR (400 MHz, CDCl3) δ 1.54 (t, J = 7.0 Hz, 3H), 2.36 (s, 3H), 4.60 (q, J = 7.0 Hz, 2H), 7.29 (d, J = 8.4 Hz, 2H), 7.58 (ddd, J = 8.0, 7.4, 0.8 Hz, 1H), 7.83 (ddd, J = 8.6, 7.4, 1.0 Hz, 1H), 8.20 (d, J = 8.4 Hz, 2H), 8.49 (ddd, J = 8.0, 0.8, 0.8 Hz, 1H), 8.52 (ddd, J = 8.6, 1.0, 0.8 Hz, 1H); 13C NMR (100 MHz, CDCl3) δ 14.4, 21.9, 63.1, 115.1, 118.9, 123.4, 125.8, 128.8 (2C), 130.1 (2C), 133.7, 134.4, 140.5, 145.0, 146.8, 146.9, 152.6, 162.7; HRMS–ESI (m/z): [M + Na]+ calcd for C19H16N4O4SNa, 419.0790; found, 419.0770 (100%).
General procedure C, amidation of 8 to amides 13: To a solution of triazine 8 (1.0 equiv) and propargyl amine (1.2 equiv) in CH2Cl2 (0.25 M) was added Zr(Ot-Bu)4 (1.2 equiv) at rt. The reaction mixture was stirred at rt for 16 h, then the mixture was diluted with CH2Cl2 (2 mL) and passed through an HM/HCl plug (hydro-matrix plug saturated with 0.1 M HCl aqueous solution) eluting with CH2Cl2. The filtrate was dried over sodium sulfate, and then the solvent was removed in vacuo. The residue was purified by flash chromatography to yield the desired amide 13.
Representative example: N-Methyl-5-[(4-methylbenzene)sulfonyl]-N-(prop-2-yn-1-yl)-5H-[1,2,4]triazino[5,6-b]indole-3-carboxamide (13a). According to general procedure C, beginning with 8a (80 mg, 0.20 mmol, 1.0 equiv) and N-methyl-propargylamine (16.7 mg, 0.24 mmol, 1.2 equiv). Purification by flash chromatography, Rf 0.54 (DCM/EtOAc, 10:1, 83 mg, 0.19 mmol, 98% yield) gave 13a as a brown-orange oil: IR (neat): 2933, 1655, 1370, 1192, 1178, 732, 666 cm−1; 1H NMR (400 MHz, CDCl3) δ 2.31 (t, J = 2.4 Hz, 0.37H, minor rotamer), 2.34–2.36 (overlap, 3.63H), 3.12 (s, 1.89H, major rotamer), 3.31 (s, 1.11H, minor rotamer), 4.22 (d, J = 2.4 Hz, 0.74H, minor rotamer), 4.52 (d, J = 2.4 Hz, 1.26H, major rotamer), 7.29 (d, J = 8.6 Hz, 2H), 7.57 (dd, J = 8.0, 8.0 Hz, 1H), 7.81 (dd, J = 8.2, 8.0 Hz, 1H), 8.11 (d, J = 8.6 Hz, 1.24H, major rotamer), 8.12 (d, J = 8.6 Hz, 0.74H, minor rotamer), 8.44 (d, J = 8.0 Hz, 1H), 8.49 (d, J = 8.2 Hz, 1H); 13C NMR (75 MHz, CDCl3) δ 21.9, (33.2, 36.0, 1C), (36.6, 40.9, 1C), (73.1, 73.9, 1C), (77.4, 77.9, 1C), 115.0, 119.1, 123.0, 125.8, 128.3 (2C), 130.3 (2C), 133.3, 134.4, 139.9, 144.3, 146.8, 147.0, (156.6, 156.8, 1C), 164.6; HRMS–ESI (m/z): [M + Na]+ calculated for C21H17N5O3SNa, 442.0950; found, 442.0935 (100%).
General procedure D, cycloaddition of 13 to 14: A solution of the amide 13 in diglyme (0.1 M) was placed in a thick-walled microwave tube, and then the reaction mixture was subjected to microwave irradiation at 160 °C for 20 min under stirring, unless otherwise noted. After the irradiation, the solvent was removed in vacuo and the residue was purified by flash chromatography to yield the cycloadducts 14.
Representative example: 13-Methyl-8-[(4-methylbenzene)sulfonyl]8,10,13-triazatetracyclo[7.7.0.02,7.011,15]hexadeca-1(16),2(7),3,5,9,11(15)-hexaen-12-one (14a). According to general procedure D, beginning with 13a (30 mg, 0.071 mmol). Purification by flash chromatography, Rf 0.17 (DCM:EtOAc, 2:1, 27 mg, 0.069 mmol, 96% yield) gave product 14a as a white yellow solid: mp 271–272 °C; IR (neat): 3057, 2987, 1739, 1692, 1375, 1175, 667 cm−1; 1H NMR (400 MHz, CDCl3) δ 2.28 (s, 3H), 3.26 (s, 3H), 4.40 (s, 2H), 7.20 (d, J = 8.4 Hz, 2H), 7.32 (dd, J = 8.0, 7.8 Hz, 1H), 7.51 (dd, J = 7.8, 7.8 Hz, 1H), 7.86 (d, J = 7.8 Hz, 1H), 8.13 (s, 1H), 8.20 (d, J = 8.4 Hz, 2H), 8.42 (d, J = 8.0 Hz, 1H); 13C NMR (100 MHz, CDCl3) δ 21.8, 30.4, 49.7, 115.0, 120.3, 121.1, 121.7, 122.8, 123.9, 128.6 (2C), 129.3, 129.6 (2C), 130.6, 135.5, 138.8, 145.4, 147.7, 151.3, 166.3; HRMS–ESI (m/z) [M + Na]+ calcd for C21H17N3O3SNa, 414.0888; found, 414.0873 (100%).
Supporting Information
| Supporting Information File 1: Experimental details and characterization data of new compounds, 1H NMR and 13C NMR spectra. | ||
| Format: PDF | Size: 5.5 MB | Download |
Acknowledgements
This work was supported by the National Institutes of Health (P41 086180). We also thank UROP (Boston University) for support of undergraduate researchers Sie-Mun Lo and Philip Roveto, and the NSF for supporting the purchase of the NMR (CHE 0619339) and HRMS (CHE 0443618) spectrometers. We also thank Professor John Porco and Dr. Aaron Beeler (Boston University, CMLD) for helpful discussions.
References
-
Moquin, C.; Guyot, M. Tetrahedron Lett. 1984, 25, 5047–5048. doi:10.1016/S0040-4039(01)91115-3
Return to citation in text: [1] -
Moquin-Pattey, C.; Guyot, M. Tetrahedron 1989, 45, 3445–3450. doi:10.1016/S0040-4020(01)81023-1
Return to citation in text: [1] -
Abas, S. A.; Hossain, M. B.; van der Helm, D.; Schmitz, F. J.; Laney, M.; Cabuslay, R.; Schatzman, R. C. J. Org. Chem. 1996, 61, 2709–2712. doi:10.1021/jo960040e
Return to citation in text: [1] -
Kim, J.-S.; Shin-ya, K.; Furihata, K.; Hayakawa, Y.; Seto, H. Tetrahedron Lett. 1997, 38, 3431–3434. doi:10.1016/S0040-4039(97)00638-2
Return to citation in text: [1] -
Sharaf, M. H. M.; Schiff, P. L., Jr.; Tackie, A. N.; Phoebe, C. H., Jr.; Martin, G. E. J. Heterocycl. Chem. 1996, 33, 239–243. doi:10.1002/jhet.5570330204
Return to citation in text: [1] -
Cimanga, K.; De Bruyne, T.; Pieters, L.; Claeys, M.; Vlietinck, A. Tetrahedron Lett. 1996, 37, 1703–1706. doi:10.1016/0040-4039(96)00112-8
Return to citation in text: [1] -
Alajarín, M.; Molina, P.; Vidal, A. J. Nat. Prod. 1997, 60, 747–748. doi:10.1021/np970177f
Return to citation in text: [1] -
Suresh Kumar, E. V. K.; Etukala, J. R.; Ablordeppey, S. Y. Mini-Rev. Med. Chem. 2008, 8, 538–554. doi:10.2174/138955708784534418
Return to citation in text: [1] -
Cimanga, K.; De Bruyne, T.; Pieters, L.; Vlietinck, A. J. J. Nat. Prod. 1997, 60, 688–691. doi:10.1021/np9605246
Return to citation in text: [1] -
Yoshida, D.; Matsumoto, T.; Yoshimura, R.; Matsuzaki, T. Biochem. Biophys. Res. Commun. 1978, 83, 915–920. doi:10.1016/0006-291X(78)91482-1
Return to citation in text: [1] -
Matsumoto, T.; Yoshida, D.; Tomita, H.; Matsushita, H. Agric. Biol. Chem. 1979, 43, 675–677. doi:10.1271/bbb1961.43.675
Return to citation in text: [1] -
Yoshida, D.; Matsumoto, T. Agric. Biol. Chem. 1979, 43, 1155–1156. doi:10.1271/bbb1961.43.1155
Return to citation in text: [1] -
Badre, A.; Boulanger, A.; Abou-Mansour, E.; Banaigs, B.; Combaut, G.; Francisco, C. J. Nat. Prod. 1994, 57, 528–533. doi:10.1021/np50106a016
Return to citation in text: [1] -
Massiot, G.; Nazabadioko, S.; Bliard, C. J. Nat. Prod. 1995, 58, 1636–1639. doi:10.1021/np50124a030
Return to citation in text: [1] -
Lawson, W.; Perkin, W. H., Jr.; Robinson, R. J. Chem. Soc., Trans. 1924, 125, 626–657. doi:10.1039/ct9242500626
Return to citation in text: [1] [2] -
Kaczmarek, Ł.; Balicki, R.; Nantka-Namirski, P.; Peczynska-Czoch, W.; Mordarski, M. Arch. Pharm. 1988, 321, 463–467. doi:10.1002/ardp.19883210807
Return to citation in text: [1] -
Peczynska-Czoch, W.; Pognan, F.; Kaczmarek, Ł.; Boratynski, J. J. Med. Chem. 1994, 37, 3503–3510. doi:10.1021/jm00047a008
Return to citation in text: [1] -
Bolton, D.; Forbes, I. T.; Hayward, C. J.; Piper, D. C.; Thomas, D. R.; Thompson, M.; Upton, N. Bioorg. Med. Chem. Lett. 1993, 3, 1941–1946. doi:10.1016/S0960-894X(01)80991-4
Return to citation in text: [1] -
Blackburn, T. P.; Bolton, D.; Forbes, I. T.; Johnson, C. N.; Martin, R. T.; Thomas, D. R.; Thompson, M.; Upton, N. Bioorg. Med. Chem. Lett. 1994, 4, 279–284. doi:10.1016/S0960-894X(01)80129-3
Return to citation in text: [1] -
Elks, J.; Webb, G. B.; Gregory, G. I.; Cocker, J. S. Novel α-carboline derivatives, the preparation thereof and compositions containing the same. GB 1268773, March 29, 1972.
Return to citation in text: [1] -
Müller, U.; Connell, R.; Bischoff, H.; Denzer, D.; Lohmer, S.; Wohlfeil, S.; Grützmann, R. Amide und Sulfonamide von heterocyclisch substituierten Benzylaminen. Eur. Pat. Appl. EP0753517 A2, Jan 15, 1997.
Return to citation in text: [1] -
Sennhenn, P.; Mantoulidis, A.; Treu, M.; Tontsch-Grunt, U.; Spevak, W.; McConnell, D.; Schoop, A.; Brueckner, R.; Jacobi, A.; Guertler, U.; Schnapp, G.; Klein, C.; Himmelsbach, F.; Pautsch, A.; Betzemeier, B.; Herfurth, L.; Mack, J.; Weidenmayer, D.; Bader, G.; Reiser, U. Alpha-carbolines as CDK-1 inhibitors. Int. Appl. WO2006131552, June 8, 2006.
Return to citation in text: [1] -
Krug, M.; Wichapong, K.; Erlenkamp, G.; Sippl, W.; Schächtele, C.; Totzke, F.; Hilgeroth, A. ChemMedChem 2011, 6, 63–72. doi:10.1002/cmdc.201000384
Return to citation in text: [1] -
Achab, S.; Guyot, M.; Potier, P. Tetrahedron Lett. 1993, 34, 2127–2130. doi:10.1016/S0040-4039(00)60362-3
Return to citation in text: [1] -
Achab, S.; Guyot, M.; Potier, P. Tetrahedron Lett. 1995, 36, 2615–2618. doi:10.1016/0040-4039(95)00323-5
Return to citation in text: [1] -
Choshi, T.; Yamada, S.; Sugino, E.; Kuwada, T.; Hibino, S. Synlett 1995, 147–148. doi:10.1055/s-1995-4910
Return to citation in text: [1] -
Choshi, T.; Yamada, S.; Sugino, E.; Kuwada, T.; Hibino, S. J. Org. Chem. 1995, 60, 5899–5904. doi:10.1021/jo00123a028
Return to citation in text: [1] -
Molina, P.; Fresneda, P. M.; Sanz, M. A. Tetrahedron Lett. 1997, 38, 6909–6912. doi:10.1016/S0040-4039(97)01592-X
Return to citation in text: [1] -
Molina, P.; Fresneda, P. M.; Sanz, M. A.; Foces-Foces, C.; Ramirez de Arellano, M. C. Tetrahedron 1998, 54, 9623–9638. doi:10.1016/S0040-4020(98)00520-1
Return to citation in text: [1] -
Miyake, F. Y.; Yakushijin, K.; Horne, D. A. Angew. Chem., Int. Ed. 2005, 44, 3280–3282. doi:10.1002/anie.200500055
Return to citation in text: [1] -
Parvatkar, P. T.; Parameswaran, P. S.; Tilve, S. G. Curr. Org. Chem. 2011, 15, 1036–1057.
Return to citation in text: [1] -
Kraus, G. A.; Guo, H. Tetrahedron Lett. 2010, 51, 4137–4139. doi:10.1016/j.tetlet.2010.05.141
Return to citation in text: [1] -
Parvatkar, P. T.; Tilve, S. G. Tetrahedron Lett. 2011, 52, 6594–6596. doi:10.1016/j.tetlet.2011.09.135
Return to citation in text: [1] -
Haddadin, M. J.; Zerdan, R. M. B.; Kurth, M. J.; Fettinger, J. C. Org. Lett. 2010, 12, 5502–5505. doi:10.1021/ol102376a
Return to citation in text: [1] -
Hostyn, S.; Tehrani, K. A.; Lemière, F.; Smout, V.; Maes, B. U. W. Tetrahedron 2011, 67, 655–659. doi:10.1016/j.tet.2010.10.077
Return to citation in text: [1] -
Freak, R. H.; Robinson, R. J. Chem. Soc. 1938, 2013–2015. doi:10.1039/jr9380002013
Return to citation in text: [1] -
Witkop, B. J. Am. Chem. Soc. 1953, 75, 3361–3370. doi:10.1021/ja01110a021
Return to citation in text: [1] -
Stephenson, L.; Warburton, W. K. J. Chem. Soc. C 1970, 1355–1364. doi:10.1039/j39700001355
Return to citation in text: [1] -
Rivalle, C.; Ducrocq, C.; Lhoste, J.-M.; Wendling, F.; Bisagni, E.; Chermann, J.-C. Tetrahedron 1981, 37, 2097–2103. doi:10.1016/S0040-4020(01)97964-5
Return to citation in text: [1] -
Mehta, L. K.; Parrick, J.; Payne, F. J. Chem. Soc., Perkin Trans. 1 1993, 11, 1261–1267. doi:10.1039/p19930001261
Return to citation in text: [1] -
Abramovitch, R. A.; Hey, D. H.; Mulley, R. D. J. Chem. Soc. 1954, 4263–4266. doi:10.1039/jr9540004263
Return to citation in text: [1] -
Vera-Luque, P.; Alajarín, R.; Alvarez-Builla, J.; Vaquero, J. J. Org. Lett. 2006, 8, 415–418. doi:10.1021/ol052552y
Return to citation in text: [1] -
Smith, P. A. S.; Boyer, J. H. J. Am. Chem. Soc. 1951, 73, 2626–2629. doi:10.1021/ja01150a061
Return to citation in text: [1] -
Bunyan, P. Y.; Cadogan, J. I. G. J. Chem. Soc. 1963, 42–49. doi:10.1039/JR9630000042
Return to citation in text: [1] -
Laha, J. K.; Petrou, P.; Cuny, G. D. J. Org. Chem. 2009, 74, 3152–3155. doi:10.1021/jo802776m
Return to citation in text: [1] -
Laha, J. K.; Barolo, S. M.; Rossi, R. A.; Cuny, G. D. J. Org. Chem. 2011, 76, 6421–6425. doi:10.1021/jo200923n
Return to citation in text: [1] -
Rocca, P.; Marsais, F.; Godard, A.; Queguiner, G. Tetrahedron 1993, 49, 49–64. doi:10.1016/S0040-4020(01)80505-6
Return to citation in text: [1] -
Okuda, S.; Robinson, M. M. J. Am. Chem. Soc. 1959, 81, 740–743. doi:10.1021/ja01512a057
Return to citation in text: [1] -
Kurihara, T.; Tani, T.; Imai, H.; Nasu, K. Chem. Pharm. Bull. 1980, 28, 2972–2979. doi:10.1248/cpb.28.2972
Return to citation in text: [1] -
Forbes, I. T.; Johnson, C. N.; Thompson, M. J. Chem. Soc., Perkin Trans. 1 1992, 92, 275–281. doi:10.1039/p19920000275
Return to citation in text: [1] -
Levacher, V.; Boussad, N.; Dupas, G.; Bourguignon, J.; Quéguiner, G. Tetrahedron 1992, 48, 831–840. doi:10.1016/S0040-4020(01)88187-4
Return to citation in text: [1] -
Meyer, M.; Guyot, M. Tetrahedron Lett. 1996, 37, 4931–4932. doi:10.1016/0040-4039(96)00950-1
Return to citation in text: [1] -
Erba, E.; Gelmi, M. L.; Pocar, D. Tetrahedron 2000, 56, 9991–9997. doi:10.1016/S0040-4020(00)00950-9
Return to citation in text: [1] -
Sharma, S. K.; Gupta, S.; Saifuddin, M.; Mandadapu, A. K.; Agarwal, P. K.; Gauniyal, H. M.; Kundu, B. Tetrahedron Lett. 2011, 52, 65–68. doi:10.1016/j.tetlet.2010.10.147
Return to citation in text: [1] -
Majumder, S.; Bhuyan, P. J. Synlett 2011, 173–176. doi:10.1055/s-0030-1259298
Return to citation in text: [1] -
Kumar, A. S.; Nagarajan, R. Org. Lett. 2011, 13, 1398–1401. doi:10.1021/ol2000827
Return to citation in text: [1] -
Barun, O.; Patra, P. K.; Ila, H.; Junjappa, H. Tetrahedron Lett. 1999, 40, 3797–3800. doi:10.1016/S0040-4039(99)00530-4
Return to citation in text: [1] -
Beccalli, E. M.; Clerici, F.; Marchesini, A. Tetrahedron 2001, 57, 4787–4792. doi:10.1016/S0040-4020(01)00404-5
Return to citation in text: [1] -
Abouabdellah, A.; Dodd, R. H. Tetrahedron Lett. 1998, 39, 2119–2122. doi:10.1016/S0040-4039(98)00065-3
Return to citation in text: [1] [2] -
Ono, A.; Narasaka, K. Chem. Lett. 2001, 146–147. doi:10.1246/cl.2001.146
Return to citation in text: [1] -
Tanaka, K.; Kitamura, M.; Narasaka, K. Bull. Chem. Soc. Jpn. 2005, 78, 1659–1664. doi:10.1246/bcsj.78.1659
Return to citation in text: [1] -
Molina, P.; Fresneda, P. M. Synthesis 1989, 878–880. doi:10.1055/s-1989-27422
Return to citation in text: [1] -
Hibino, S.; Sugino, E.; Kuwada, T.; Ogura, N.; Shintani, Y.; Satoh, K. Chem. Pharm. Bull. 1991, 39, 79–80. doi:10.1248/cpb.39.79
Return to citation in text: [1] -
Bonini, C.; Funicello, M.; Spagnolo, P. Synlett 2006, 1574–1576. doi:10.1055/s-2006-944182
Return to citation in text: [1] -
Chavan, N. L.; Nayak, S. K.; Kusurkar, R. S. Tetrahedron 2010, 66, 1827–1831. doi:10.1016/j.tet.2010.01.042
Return to citation in text: [1] -
Majumder, S.; Bhuyan, P. J. Synlett 2011, 1547–1550. doi:10.1055/s-0030-1260787
Return to citation in text: [1] -
Molina, P.; Alajarin, M.; Vidal, A.; Sanchez-Andrada, P. J. Org. Chem. 1992, 57, 929–939. doi:10.1021/jo00029a026
Return to citation in text: [1] -
Saito, T.; Ohmori, H.; Furuno, E.; Motoki, S. J. Chem. Soc., Chem. Commun. 1992, 22–24. doi:10.1039/C39920000022
Return to citation in text: [1] -
Schneider, C.; Goyard, D.; Gueyrard, D.; Joseph, B.; Goekjian, P. G. Eur. J. Org. Chem. 2010, 6665–6677. doi:10.1002/ejoc.201000795
See for an example of diversification of the α-carboline scaffold amenable to library synthesis.
Return to citation in text: [1] -
Stolle, W. A. W.; Marcelis, A. T. M.; Koetsier, A.; van der Plas, H. C. Tetrahedron 1989, 45, 6511–6518. doi:10.1016/S0040-4020(01)89527-2
Return to citation in text: [1] -
Forbes, I. T.; Johnson, C. N.; Thompson, M. Synth. Commun. 1993, 23, 715–723. doi:10.1080/00397919308009831
Return to citation in text: [1] -
Tahri, A.; Buysens, K. J.; van der Eycken, E. V.; Vandenberghe, D. M.; Hoornaert, G. J. Tetrahedron 1998, 54, 13211–13226. doi:10.1016/S0040-4020(98)00802-3
Return to citation in text: [1] -
Repič, O.; Mattner, P. G.; Shapiro, M. J. J. Heterocycl. Chem. 1982, 19, 1201–1204. doi:10.1002/jhet.5570190542
Return to citation in text: [1] -
Doleschall, G.; Lempert, K. Tetrahedron 1974, 30, 3997–4012. doi:10.1016/S0040-4020(01)97375-2
Return to citation in text: [1] -
Boger, D. L.; Duff, S. R.; Panek, J. S.; Yasuda, M. J. Org. Chem. 1985, 50, 5782–5789. doi:10.1021/jo00350a069
Return to citation in text: [1] -
Boger, D. L.; Panek, J. S.; Yasuda, M. Org. Synth. 1988, 66, 142–150.
Return to citation in text: [1] [2] -
Benson, S. C.; Gross, J. L.; Snyder, J. K. J. Org. Chem. 1990, 55, 3257–3269. doi:10.1021/jo00297a050
Return to citation in text: [1] -
Soenen, D. R.; Zimpleman, J. M.; Boger, D. L. J. Org. Chem. 2003, 68, 3593–3598. doi:10.1021/jo020713v
Return to citation in text: [1] -
Lahue, B. R.; Lo, S.-M.; Wan, Z.-K.; Woo, G. H. C.; Snyder, J. K. J. Org. Chem. 2004, 69, 7171–7182. doi:10.1021/jo040193z
Return to citation in text: [1] -
Lahue, B. R.; Wan, Z.-K.; Snyder, J. K. J. Org. Chem. 2003, 68, 4345–4354. doi:10.1021/jo030049y
Return to citation in text: [1] -
Basha, A.; Lipton, M.; Weinreb, S. M. Tetrahedron Lett. 1977, 18, 4171–4172. doi:10.1016/S0040-4039(01)83457-2
Return to citation in text: [1] -
Levin, J. I.; Turos, E.; Weinreb, S. M. Synth. Commun. 1982, 12, 989–993. doi:10.1080/00397918208061938
Return to citation in text: [1] -
Sidler, D. R.; Lovelace, T. C.; McNamara, J. M.; Reider, P. J. J. Org. Chem. 1994, 59, 1231–1233. doi:10.1021/jo00085a001
Return to citation in text: [1] -
Nomak, R.; Snyder, J. K. Tetrahedron Lett. 2001, 42, 7929–7933. doi:10.1016/S0040-4039(01)01678-1
Return to citation in text: [1] -
Han, C.; Porco, J. A., Jr. Org. Lett. 2007, 9, 1517–1520. doi:10.1021/ol0702728
Return to citation in text: [1] -
Girardot, M.; Nomak, R.; Snyder, J. K. J. Org. Chem. 1998, 63, 10063–10068. doi:10.1021/jo9815352
Return to citation in text: [1] -
Gutmann, B.; Obermayer, D.; Reichart, B.; Prekodravac, B.; Irfan, M.; Kremsner, J. M.; Kappe, C. O. Chem.–Eur. J. 2010, 16, 12182–12194. doi:10.1002/chem.201001703
Return to citation in text: [1] -
Beesley, R. M.; Ingold, C. K.; Thorpe, J. F. J. Chem. Soc., Trans. 1915, 107, 1080–1106. doi:10.1039/CT9150701080
Return to citation in text: [1] -
Ringer, A. L.; Magers, D. H. J. Org. Chem. 2007, 72, 2533–2537. doi:10.1021/jo0624647
Return to citation in text: [1] -
Fürstner, A.; Langemann, K. J. Org. Chem. 1996, 61, 8746–8749. doi:10.1021/jo961600c
Return to citation in text: [1] -
Kondaiah, G. C. M.; Reddy, L. A.; Babu, K. S.; Gurav, V. M.; Huge, K. G.; Bandichhor, R.; Reddy, P. P.; Bhattacharya, A.; Anand, R. V. Tetrahedron Lett. 2008, 49, 106–109. doi:10.1016/j.tetlet.2007.11.008
Return to citation in text: [1] -
Ranu, B. C.; Dutta, P.; Sarkar, A. J. Org. Chem. 1998, 63, 6027–6028. doi:10.1021/jo980157y
Return to citation in text: [1] -
Shapiro, G.; Marzi, M. J. Org. Chem. 1997, 62, 7096–7097. doi:10.1021/jo971498z
Return to citation in text: [1] -
Otera, J.; Danoh, N.; Nozaki, H. J. Org. Chem. 1991, 56, 5307–5311. doi:10.1021/jo00018a019
Return to citation in text: [1] -
Taylor, E. C.; Macor, J. E. Tetrahedron Lett. 1986, 27, 2107–2110. doi:10.1016/S0040-4039(00)84460-3
Return to citation in text: [1] -
Taylor, E. C.; Macor, J. E. J. Org. Chem. 1987, 52, 4280–4287. doi:10.1021/jo00228a023
Return to citation in text: [1] -
Taylor, E. C.; Macor, J. E. J. Org. Chem. 1989, 54, 4984–4989. doi:10.1021/jo00282a006
Return to citation in text: [1] -
Taylor, E. C.; Macor, J. E.; French, L. G. J. Org. Chem. 1991, 56, 1807–1812. doi:10.1021/jo00005a027
Return to citation in text: [1] -
Page, M. I.; Jencks, W. P. Proc. Natl. Acad. Sci. U. S. A. 1971, 68, 1678–1683. doi:10.1073/pnas.68.8.1678
Return to citation in text: [1] -
Casadei, M. A.; Galli, C.; Mandolini, L. J. Am. Chem. Soc. 1984, 106, 1051–1056. doi:10.1021/ja00316a039
Return to citation in text: [1]
| 45. | Laha, J. K.; Petrou, P.; Cuny, G. D. J. Org. Chem. 2009, 74, 3152–3155. doi:10.1021/jo802776m |
| 46. | Laha, J. K.; Barolo, S. M.; Rossi, R. A.; Cuny, G. D. J. Org. Chem. 2011, 76, 6421–6425. doi:10.1021/jo200923n |
| 47. | Rocca, P.; Marsais, F.; Godard, A.; Queguiner, G. Tetrahedron 1993, 49, 49–64. doi:10.1016/S0040-4020(01)80505-6 |
| 65. | Chavan, N. L.; Nayak, S. K.; Kusurkar, R. S. Tetrahedron 2010, 66, 1827–1831. doi:10.1016/j.tet.2010.01.042 |
| 66. | Majumder, S.; Bhuyan, P. J. Synlett 2011, 1547–1550. doi:10.1055/s-0030-1260787 |
| 60. | Ono, A.; Narasaka, K. Chem. Lett. 2001, 146–147. doi:10.1246/cl.2001.146 |
| 61. | Tanaka, K.; Kitamura, M.; Narasaka, K. Bull. Chem. Soc. Jpn. 2005, 78, 1659–1664. doi:10.1246/bcsj.78.1659 |
| 62. | Molina, P.; Fresneda, P. M. Synthesis 1989, 878–880. doi:10.1055/s-1989-27422 |
| 63. | Hibino, S.; Sugino, E.; Kuwada, T.; Ogura, N.; Shintani, Y.; Satoh, K. Chem. Pharm. Bull. 1991, 39, 79–80. doi:10.1248/cpb.39.79 |
| 64. | Bonini, C.; Funicello, M.; Spagnolo, P. Synlett 2006, 1574–1576. doi:10.1055/s-2006-944182 |
| 57. | Barun, O.; Patra, P. K.; Ila, H.; Junjappa, H. Tetrahedron Lett. 1999, 40, 3797–3800. doi:10.1016/S0040-4039(99)00530-4 |
| 58. | Beccalli, E. M.; Clerici, F.; Marchesini, A. Tetrahedron 2001, 57, 4787–4792. doi:10.1016/S0040-4020(01)00404-5 |
| 59. | Abouabdellah, A.; Dodd, R. H. Tetrahedron Lett. 1998, 39, 2119–2122. doi:10.1016/S0040-4039(98)00065-3 |
| 48. | Okuda, S.; Robinson, M. M. J. Am. Chem. Soc. 1959, 81, 740–743. doi:10.1021/ja01512a057 |
| 49. | Kurihara, T.; Tani, T.; Imai, H.; Nasu, K. Chem. Pharm. Bull. 1980, 28, 2972–2979. doi:10.1248/cpb.28.2972 |
| 50. | Forbes, I. T.; Johnson, C. N.; Thompson, M. J. Chem. Soc., Perkin Trans. 1 1992, 92, 275–281. doi:10.1039/p19920000275 |
| 51. | Levacher, V.; Boussad, N.; Dupas, G.; Bourguignon, J.; Quéguiner, G. Tetrahedron 1992, 48, 831–840. doi:10.1016/S0040-4020(01)88187-4 |
| 52. | Meyer, M.; Guyot, M. Tetrahedron Lett. 1996, 37, 4931–4932. doi:10.1016/0040-4039(96)00950-1 |
| 53. | Erba, E.; Gelmi, M. L.; Pocar, D. Tetrahedron 2000, 56, 9991–9997. doi:10.1016/S0040-4020(00)00950-9 |
| 54. | Sharma, S. K.; Gupta, S.; Saifuddin, M.; Mandadapu, A. K.; Agarwal, P. K.; Gauniyal, H. M.; Kundu, B. Tetrahedron Lett. 2011, 52, 65–68. doi:10.1016/j.tetlet.2010.10.147 |
| 55. | Majumder, S.; Bhuyan, P. J. Synlett 2011, 173–176. doi:10.1055/s-0030-1259298 |
| 56. | Kumar, A. S.; Nagarajan, R. Org. Lett. 2011, 13, 1398–1401. doi:10.1021/ol2000827 |
| 67. | Molina, P.; Alajarin, M.; Vidal, A.; Sanchez-Andrada, P. J. Org. Chem. 1992, 57, 929–939. doi:10.1021/jo00029a026 |
| 68. | Saito, T.; Ohmori, H.; Furuno, E.; Motoki, S. J. Chem. Soc., Chem. Commun. 1992, 22–24. doi:10.1039/C39920000022 |
| 69. |
Schneider, C.; Goyard, D.; Gueyrard, D.; Joseph, B.; Goekjian, P. G. Eur. J. Org. Chem. 2010, 6665–6677. doi:10.1002/ejoc.201000795
See for an example of diversification of the α-carboline scaffold amenable to library synthesis. |
| 70. | Stolle, W. A. W.; Marcelis, A. T. M.; Koetsier, A.; van der Plas, H. C. Tetrahedron 1989, 45, 6511–6518. doi:10.1016/S0040-4020(01)89527-2 |
| 71. | Forbes, I. T.; Johnson, C. N.; Thompson, M. Synth. Commun. 1993, 23, 715–723. doi:10.1080/00397919308009831 |
| 85. | Han, C.; Porco, J. A., Jr. Org. Lett. 2007, 9, 1517–1520. doi:10.1021/ol0702728 |
| 86. | Girardot, M.; Nomak, R.; Snyder, J. K. J. Org. Chem. 1998, 63, 10063–10068. doi:10.1021/jo9815352 |
| 78. | Soenen, D. R.; Zimpleman, J. M.; Boger, D. L. J. Org. Chem. 2003, 68, 3593–3598. doi:10.1021/jo020713v |
| 79. | Lahue, B. R.; Lo, S.-M.; Wan, Z.-K.; Woo, G. H. C.; Snyder, J. K. J. Org. Chem. 2004, 69, 7171–7182. doi:10.1021/jo040193z |
| 80. | Lahue, B. R.; Wan, Z.-K.; Snyder, J. K. J. Org. Chem. 2003, 68, 4345–4354. doi:10.1021/jo030049y |
| 81. | Basha, A.; Lipton, M.; Weinreb, S. M. Tetrahedron Lett. 1977, 18, 4171–4172. doi:10.1016/S0040-4039(01)83457-2 |
| 82. | Levin, J. I.; Turos, E.; Weinreb, S. M. Synth. Commun. 1982, 12, 989–993. doi:10.1080/00397918208061938 |
| 83. | Sidler, D. R.; Lovelace, T. C.; McNamara, J. M.; Reider, P. J. J. Org. Chem. 1994, 59, 1231–1233. doi:10.1021/jo00085a001 |
| 84. | Nomak, R.; Snyder, J. K. Tetrahedron Lett. 2001, 42, 7929–7933. doi:10.1016/S0040-4039(01)01678-1 |
| 73. | Repič, O.; Mattner, P. G.; Shapiro, M. J. J. Heterocycl. Chem. 1982, 19, 1201–1204. doi:10.1002/jhet.5570190542 |
| 74. | Doleschall, G.; Lempert, K. Tetrahedron 1974, 30, 3997–4012. doi:10.1016/S0040-4020(01)97375-2 |
| 75. | Boger, D. L.; Duff, S. R.; Panek, J. S.; Yasuda, M. J. Org. Chem. 1985, 50, 5782–5789. doi:10.1021/jo00350a069 |
| 76. | Boger, D. L.; Panek, J. S.; Yasuda, M. Org. Synth. 1988, 66, 142–150. |
| 77. | Benson, S. C.; Gross, J. L.; Snyder, J. K. J. Org. Chem. 1990, 55, 3257–3269. doi:10.1021/jo00297a050 |
| 72. | Tahri, A.; Buysens, K. J.; van der Eycken, E. V.; Vandenberghe, D. M.; Hoornaert, G. J. Tetrahedron 1998, 54, 13211–13226. doi:10.1016/S0040-4020(98)00802-3 |
| 59. | Abouabdellah, A.; Dodd, R. H. Tetrahedron Lett. 1998, 39, 2119–2122. doi:10.1016/S0040-4039(98)00065-3 |
| 88. | Beesley, R. M.; Ingold, C. K.; Thorpe, J. F. J. Chem. Soc., Trans. 1915, 107, 1080–1106. doi:10.1039/CT9150701080 |
| 89. | Ringer, A. L.; Magers, D. H. J. Org. Chem. 2007, 72, 2533–2537. doi:10.1021/jo0624647 |
| 90. | Fürstner, A.; Langemann, K. J. Org. Chem. 1996, 61, 8746–8749. doi:10.1021/jo961600c |
| 91. | Kondaiah, G. C. M.; Reddy, L. A.; Babu, K. S.; Gurav, V. M.; Huge, K. G.; Bandichhor, R.; Reddy, P. P.; Bhattacharya, A.; Anand, R. V. Tetrahedron Lett. 2008, 49, 106–109. doi:10.1016/j.tetlet.2007.11.008 |
| 87. | Gutmann, B.; Obermayer, D.; Reichart, B.; Prekodravac, B.; Irfan, M.; Kremsner, J. M.; Kappe, C. O. Chem.–Eur. J. 2010, 16, 12182–12194. doi:10.1002/chem.201001703 |
| 1. | Moquin, C.; Guyot, M. Tetrahedron Lett. 1984, 25, 5047–5048. doi:10.1016/S0040-4039(01)91115-3 |
| 2. | Moquin-Pattey, C.; Guyot, M. Tetrahedron 1989, 45, 3445–3450. doi:10.1016/S0040-4020(01)81023-1 |
| 6. | Cimanga, K.; De Bruyne, T.; Pieters, L.; Claeys, M.; Vlietinck, A. Tetrahedron Lett. 1996, 37, 1703–1706. doi:10.1016/0040-4039(96)00112-8 |
| 17. | Peczynska-Czoch, W.; Pognan, F.; Kaczmarek, Ł.; Boratynski, J. J. Med. Chem. 1994, 37, 3503–3510. doi:10.1021/jm00047a008 |
| 5. | Sharaf, M. H. M.; Schiff, P. L., Jr.; Tackie, A. N.; Phoebe, C. H., Jr.; Martin, G. E. J. Heterocycl. Chem. 1996, 33, 239–243. doi:10.1002/jhet.5570330204 |
| 18. | Bolton, D.; Forbes, I. T.; Hayward, C. J.; Piper, D. C.; Thomas, D. R.; Thompson, M.; Upton, N. Bioorg. Med. Chem. Lett. 1993, 3, 1941–1946. doi:10.1016/S0960-894X(01)80991-4 |
| 19. | Blackburn, T. P.; Bolton, D.; Forbes, I. T.; Johnson, C. N.; Martin, R. T.; Thomas, D. R.; Thompson, M.; Upton, N. Bioorg. Med. Chem. Lett. 1994, 4, 279–284. doi:10.1016/S0960-894X(01)80129-3 |
| 4. | Kim, J.-S.; Shin-ya, K.; Furihata, K.; Hayakawa, Y.; Seto, H. Tetrahedron Lett. 1997, 38, 3431–3434. doi:10.1016/S0040-4039(97)00638-2 |
| 15. | Lawson, W.; Perkin, W. H., Jr.; Robinson, R. J. Chem. Soc., Trans. 1924, 125, 626–657. doi:10.1039/ct9242500626 |
| 99. | Page, M. I.; Jencks, W. P. Proc. Natl. Acad. Sci. U. S. A. 1971, 68, 1678–1683. doi:10.1073/pnas.68.8.1678 |
| 3. | Abas, S. A.; Hossain, M. B.; van der Helm, D.; Schmitz, F. J.; Laney, M.; Cabuslay, R.; Schatzman, R. C. J. Org. Chem. 1996, 61, 2709–2712. doi:10.1021/jo960040e |
| 16. | Kaczmarek, Ł.; Balicki, R.; Nantka-Namirski, P.; Peczynska-Czoch, W.; Mordarski, M. Arch. Pharm. 1988, 321, 463–467. doi:10.1002/ardp.19883210807 |
| 100. | Casadei, M. A.; Galli, C.; Mandolini, L. J. Am. Chem. Soc. 1984, 106, 1051–1056. doi:10.1021/ja00316a039 |
| 10. | Yoshida, D.; Matsumoto, T.; Yoshimura, R.; Matsuzaki, T. Biochem. Biophys. Res. Commun. 1978, 83, 915–920. doi:10.1016/0006-291X(78)91482-1 |
| 11. | Matsumoto, T.; Yoshida, D.; Tomita, H.; Matsushita, H. Agric. Biol. Chem. 1979, 43, 675–677. doi:10.1271/bbb1961.43.675 |
| 13. | Badre, A.; Boulanger, A.; Abou-Mansour, E.; Banaigs, B.; Combaut, G.; Francisco, C. J. Nat. Prod. 1994, 57, 528–533. doi:10.1021/np50106a016 |
| 94. | Otera, J.; Danoh, N.; Nozaki, H. J. Org. Chem. 1991, 56, 5307–5311. doi:10.1021/jo00018a019 |
| 9. | Cimanga, K.; De Bruyne, T.; Pieters, L.; Vlietinck, A. J. J. Nat. Prod. 1997, 60, 688–691. doi:10.1021/np9605246 |
| 14. | Massiot, G.; Nazabadioko, S.; Bliard, C. J. Nat. Prod. 1995, 58, 1636–1639. doi:10.1021/np50124a030 |
| 95. | Taylor, E. C.; Macor, J. E. Tetrahedron Lett. 1986, 27, 2107–2110. doi:10.1016/S0040-4039(00)84460-3 |
| 96. | Taylor, E. C.; Macor, J. E. J. Org. Chem. 1987, 52, 4280–4287. doi:10.1021/jo00228a023 |
| 97. | Taylor, E. C.; Macor, J. E. J. Org. Chem. 1989, 54, 4984–4989. doi:10.1021/jo00282a006 |
| 98. | Taylor, E. C.; Macor, J. E.; French, L. G. J. Org. Chem. 1991, 56, 1807–1812. doi:10.1021/jo00005a027 |
| 8. | Suresh Kumar, E. V. K.; Etukala, J. R.; Ablordeppey, S. Y. Mini-Rev. Med. Chem. 2008, 8, 538–554. doi:10.2174/138955708784534418 |
| 92. | Ranu, B. C.; Dutta, P.; Sarkar, A. J. Org. Chem. 1998, 63, 6027–6028. doi:10.1021/jo980157y |
| 7. | Alajarín, M.; Molina, P.; Vidal, A. J. Nat. Prod. 1997, 60, 747–748. doi:10.1021/np970177f |
| 12. | Yoshida, D.; Matsumoto, T. Agric. Biol. Chem. 1979, 43, 1155–1156. doi:10.1271/bbb1961.43.1155 |
| 93. | Shapiro, G.; Marzi, M. J. Org. Chem. 1997, 62, 7096–7097. doi:10.1021/jo971498z |
| 22. | Sennhenn, P.; Mantoulidis, A.; Treu, M.; Tontsch-Grunt, U.; Spevak, W.; McConnell, D.; Schoop, A.; Brueckner, R.; Jacobi, A.; Guertler, U.; Schnapp, G.; Klein, C.; Himmelsbach, F.; Pautsch, A.; Betzemeier, B.; Herfurth, L.; Mack, J.; Weidenmayer, D.; Bader, G.; Reiser, U. Alpha-carbolines as CDK-1 inhibitors. Int. Appl. WO2006131552, June 8, 2006. |
| 20. | Elks, J.; Webb, G. B.; Gregory, G. I.; Cocker, J. S. Novel α-carboline derivatives, the preparation thereof and compositions containing the same. GB 1268773, March 29, 1972. |
| 21. | Müller, U.; Connell, R.; Bischoff, H.; Denzer, D.; Lohmer, S.; Wohlfeil, S.; Grützmann, R. Amide und Sulfonamide von heterocyclisch substituierten Benzylaminen. Eur. Pat. Appl. EP0753517 A2, Jan 15, 1997. |
| 42. | Vera-Luque, P.; Alajarín, R.; Alvarez-Builla, J.; Vaquero, J. J. Org. Lett. 2006, 8, 415–418. doi:10.1021/ol052552y |
| 43. | Smith, P. A. S.; Boyer, J. H. J. Am. Chem. Soc. 1951, 73, 2626–2629. doi:10.1021/ja01150a061 |
| 44. | Bunyan, P. Y.; Cadogan, J. I. G. J. Chem. Soc. 1963, 42–49. doi:10.1039/JR9630000042 |
| 37. | Witkop, B. J. Am. Chem. Soc. 1953, 75, 3361–3370. doi:10.1021/ja01110a021 |
| 38. | Stephenson, L.; Warburton, W. K. J. Chem. Soc. C 1970, 1355–1364. doi:10.1039/j39700001355 |
| 39. | Rivalle, C.; Ducrocq, C.; Lhoste, J.-M.; Wendling, F.; Bisagni, E.; Chermann, J.-C. Tetrahedron 1981, 37, 2097–2103. doi:10.1016/S0040-4020(01)97964-5 |
| 40. | Mehta, L. K.; Parrick, J.; Payne, F. J. Chem. Soc., Perkin Trans. 1 1993, 11, 1261–1267. doi:10.1039/p19930001261 |
| 41. | Abramovitch, R. A.; Hey, D. H.; Mulley, R. D. J. Chem. Soc. 1954, 4263–4266. doi:10.1039/jr9540004263 |
| 31. | Parvatkar, P. T.; Parameswaran, P. S.; Tilve, S. G. Curr. Org. Chem. 2011, 15, 1036–1057. |
| 32. | Kraus, G. A.; Guo, H. Tetrahedron Lett. 2010, 51, 4137–4139. doi:10.1016/j.tetlet.2010.05.141 |
| 33. | Parvatkar, P. T.; Tilve, S. G. Tetrahedron Lett. 2011, 52, 6594–6596. doi:10.1016/j.tetlet.2011.09.135 |
| 34. | Haddadin, M. J.; Zerdan, R. M. B.; Kurth, M. J.; Fettinger, J. C. Org. Lett. 2010, 12, 5502–5505. doi:10.1021/ol102376a |
| 35. | Hostyn, S.; Tehrani, K. A.; Lemière, F.; Smout, V.; Maes, B. U. W. Tetrahedron 2011, 67, 655–659. doi:10.1016/j.tet.2010.10.077 |
| 15. | Lawson, W.; Perkin, W. H., Jr.; Robinson, R. J. Chem. Soc., Trans. 1924, 125, 626–657. doi:10.1039/ct9242500626 |
| 36. | Freak, R. H.; Robinson, R. J. Chem. Soc. 1938, 2013–2015. doi:10.1039/jr9380002013 |
| 23. | Krug, M.; Wichapong, K.; Erlenkamp, G.; Sippl, W.; Schächtele, C.; Totzke, F.; Hilgeroth, A. ChemMedChem 2011, 6, 63–72. doi:10.1002/cmdc.201000384 |
| 24. | Achab, S.; Guyot, M.; Potier, P. Tetrahedron Lett. 1993, 34, 2127–2130. doi:10.1016/S0040-4039(00)60362-3 |
| 25. | Achab, S.; Guyot, M.; Potier, P. Tetrahedron Lett. 1995, 36, 2615–2618. doi:10.1016/0040-4039(95)00323-5 |
| 26. | Choshi, T.; Yamada, S.; Sugino, E.; Kuwada, T.; Hibino, S. Synlett 1995, 147–148. doi:10.1055/s-1995-4910 |
| 27. | Choshi, T.; Yamada, S.; Sugino, E.; Kuwada, T.; Hibino, S. J. Org. Chem. 1995, 60, 5899–5904. doi:10.1021/jo00123a028 |
| 28. | Molina, P.; Fresneda, P. M.; Sanz, M. A. Tetrahedron Lett. 1997, 38, 6909–6912. doi:10.1016/S0040-4039(97)01592-X |
| 29. | Molina, P.; Fresneda, P. M.; Sanz, M. A.; Foces-Foces, C.; Ramirez de Arellano, M. C. Tetrahedron 1998, 54, 9623–9638. doi:10.1016/S0040-4020(98)00520-1 |
| 30. | Miyake, F. Y.; Yakushijin, K.; Horne, D. A. Angew. Chem., Int. Ed. 2005, 44, 3280–3282. doi:10.1002/anie.200500055 |
© 2012 Ma et al; licensee Beilstein-Institut.
This is an Open Access article under the terms of the Creative Commons Attribution License (http://creativecommons.org/licenses/by/2.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
The license is subject to the Beilstein Journal of Organic Chemistry terms and conditions: (http://www.beilstein-journals.org/bjoc)