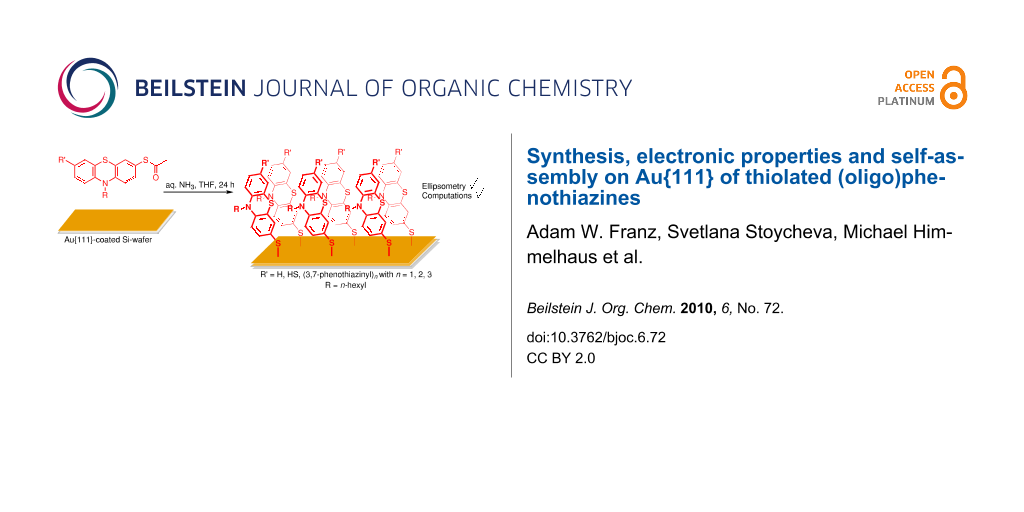
Abstract
(Oligo)phenothiazinyl thioacetates, synthesized by a one-pot sequence, are electrochemically oxidizable and highly fluorescent. SAMs can be readily formed from thiols prepared by in situ deprotection of the thioacetates in the presence of a gold-coated silicon wafer. Monolayer formation is confirmed by ellipsometry and the results compared to those obtained by force field and DFT calculations.
Graphical Abstract
Introduction
Functional organic π-systems [1] are of great relevance in the miniaturization of electronic devices particularly since they could serve as molecular switches, wires, and transistors [2-5]. As a consequence, the molecule-based bottom-up approach to nanodimensional structured self-assembled monolayers (SAMs) on well-defined metal surfaces has become a groundbreaking strategy in the development of molecular electronics [6]. In recent years, many investigations into SAMs of organic molecules on gold surfaces have been carried out [7]. Thiols, thiol esters, and disulfides can be easily chemisorbed on gold to form SAMs by exposure of well-defined gold substrates to solutions of sulfur functionalized molecules [7-14]. These “alligator-clips” [15-17] are able to bind functional molecules covalently to gold{111}-surfaces. Phenyl derivatives [18,19], conjugated bi- [18,20] and oligophenyls [18,20,21], oligothiophenes [18], porphyrin derivatives [20], phenanthrenes [22,23], fullerenes [24], and optically active naphthalenes [25] adsorbed on gold were studied in break-junction experiments and their properties on conductance, 1-bit random access memory and, especially, with regard to their ability to function as conductive molecular wires investigated. Among many heteroaromatic systems, phenothiazines, their derivatives and oligomers are interesting building blocks for rigid-rod and wire-like molecular modules for single-molecule electronics as a consequence of their electronic properties. In particular, their reversible formation of stable radical cations [26-31], their tunable redox and fluorescence properties [32-34], and their tendency to self-assemble on surfaces by π–π interactions [35] make them eligible for use as redox-switchable molecular entities. In addition, the inherent folded conformation of phenothiazines [36], with a folding angle of 158.5°, represents an intriguing new aspect for the formation of self-assembled monolayers (SAMs) of this class of compounds. Furthermore, the transformation of phenothiazines into stable planar radical cations with excellent delocalization [37] qualifies them as excellent models for switchable conductive or semiconductive molecular wires. Encouraged by successful electrode modifications with conjugated thiolated anilines [38] and SAM formation of thiolated phenylethynyl phenothiazines [39], and in continuation of our investigations directed towards the synthesis and study of (oligo)phenothiazine-based functional π-systems [40-46], we have now focused our attention on thiolated phenothiazines and (oligo)phenothiazines as “alligator-clips”. Here, we report the synthesis of phenothiazines and their oligomers bearing “alligator-clips” and their electronic properties as studied by cyclic voltammetry (CV), spectroscopic and spectrometric methods. Furthermore, their chemisorption and SAM formation on Au{111} were studied by ellipsometry.
Results and Discussion
Synthesis
The facile bromine–lithium exchange of bromo phenothiazines [47-49] and the subsequent electrophilic trapping reactions of the resulting lithio phenothiazines [50,51] with different electrophiles set the stage for a straightforward synthesis of thiolated (oligo)phenothiazines. Therefore, the synthesis of thiofunctionalized phenothiazines can be accomplished according to a standard protocol [18]. Thus, solutions of bromo phenothiazines 1 [32,52] were cooled to −78 °C and reacted with n-BuLi (1a and 1b) or t-BuLi (1c–e and 3), respectively, to give the corresponding lithio phenothiazines via bromine–lithium exchange. Subsequent addition of elemental sulfur, followed by stirring for 5 min at −78 °C, and the addition of freshly distilled acetyl chloride furnished the desired (oligo)phenothiazinyl thioacetates 2 and 4 in moderate to good yields (Scheme 1). However, in the case of dyad 1c thiolation was only accomplished by addition of acetylsulfur chloride [53] to the lithio species at low temperature, albeit the thiofunctionalized derivative 2c was obtained in only 15% yield. The structures of the (oligo)phenothiazinyl thioacetates 2 and 4 were unambiguously supported by 1H and 13C NMR spectroscopy, mass spectrometry and elemental analysis.
Scheme 1: Synthesis of (oligo)phenothiazinyl thioacetates 2 and 4.
Scheme 1: Synthesis of (oligo)phenothiazinyl thioacetates 2 and 4.
Electronic properties
The electronic properties of the (oligo)phenothiazinyl thioacetates 2 and 4 were investigated by absorption and emission spectra, and cyclic voltammetry (Table 1). Optical spectroscopy (UV–vis and fluorescence spectra) revealed that only the triad 2d and the tetrad 2e displayed considerable fluorescence with emission of greenish-blue light and large Stokes shifts (Figure 1, Δ 6400–6600 cm−1). While the absence of fluorescence of monophenothiazines 2a, 2b, and 4 with heavy atom substitution and consequently, increased spin–orbit coupling is not too surprising, the presence of a diphenothiazine unit (2c) is not sufficient. Hence, at least two covalently bound phenothiazines without an additional sulfur substituent appears to be the prerequisite for intense fluorescence of oligophenothiazinyl thioacetates.
Table 1: Selected electronic properties of (oligo)phenothiazinyl thioacetates 2 and 4 (absorptiona and emission spectraa and cyclic voltammetryb).
| Absorption λmax,abs (nm) | Emission λmax,em (nm) |
Stokes shift Δ |
E00/+1 (mV) | E0+1/+2 (mV) | E0+2/+3 (mV) | |
|---|---|---|---|---|---|---|
| 2a | 266, 310 | – | – | 800 | – | – |
| 2b | 264, 316 | – | – | 838 | – | – |
| 2c | 276, 324, 366 | – | – | 668 | 853 | – |
| 2d | 280, 326, 364 | 474 | 6400 | 608 | 765 | 876 |
| 2e | 282, 326, 362 | 476 | 6600 | 597 | 690 | 842c |
| 4 | 272, 326 | – | – | 875 | – | – |
aRecorded in CH2Cl2.
bRecorded in CH2Cl2, 20 °C, v = 100 mV/s, electrolyte: n-Bu4N+PF6−, Pt working electrode, Pt counter-electrode, Ag/AgCl reference electrode.
cThe third and fourth oxidation waves coincide.
Figure 1: Normalized absorption (solid line) and emission (dashed line) spectra of thioacetate 2d (recorded in dichloromethane, T = 298 K).
Figure 1: Normalized absorption (solid line) and emission (dashed line) spectra of thioacetate 2d (recorded i...
Electrochemical data for (oligo)phenothiazinyl thioacetates 2 and 4 were obtained by cyclic voltammetry in the anodic region (scan area up to 1.5 V). The reversible first oxidations to the radical cations of monophenothiazines 2a, 2b, and 4 were shifted anodically in comparison to unsubstituted monophenothiazines [54] as a consequence of the electron-withdrawing nature of the thioacetate. Due to unsymmetrical substitution, the dyad 2c showed two distinctly separated, reversible oxidations at E00/+1 = 668 mV and E0+1/+2 = 853 mV. The cyclic voltammogram of the triad 2d displayed three distinctly separated, reversible oxidations at E00/+1 = 608 mV, E0+1/+2 = 765 mV, and E0+2/+3 = 876 mV (Figure 2). However, the electrochemistry of the tetrad 2e is more complicated. Only three distinctly separated, reversible oxidations were evident. The first oxidations at E00/+1 = 597 mV and E0+1/+2 = 690 mV are in accordance with Nernstian behavior, while the third oxidation at E0 = 842 mV reveals a large difference of ΔE = 132 mV for the current peaks of the oxidation and the reduction wave. Presumably, the expected third and fourth oxidations coincide and give rise to a combined quasi-reversible peak.
Figure 2: Cyclic voltammogram of thioacetate 2d (recorded in CH2Cl2, T = 293 K; 0.1 M electrolyte [Bu4N][PF6]; ν = 100 mV/s; Pt-working electrode, Ag/AgCl-reference and Pt-counter electrode.
Figure 2: Cyclic voltammogram of thioacetate 2d (recorded in CH2Cl2, T = 293 K; 0.1 M electrolyte [Bu4N][PF6]...
Self-assembly and ellipsometry
SAMs on a Au{111}-coated silicon wafer substrate were prepared from (oligo)phenothiazinyl thioacetates 2 or 4 by in situ saponification with degassed aqueous ammonia in THF at room temperature for 24 h (Scheme 2).
Scheme 2: Preparation of SAMs from (oligo)phenothiazinyl thioacetates 2 or 4 on a Au{111}-coated silicon wafer substrate.
Scheme 2: Preparation of SAMs from (oligo)phenothiazinyl thioacetates 2 or 4 on a Au{111}-coated silicon wafe...
Based upon thorough surface analysis of the previously studied thiolated phenylethynyl phenothiazines chemisorbed on Au{111} by ellipsometry, contact angle measurements, X-ray photoelectron spectroscopy, and infrared reflection absorption spectroscopy (IRRAS) [39], we applied ellipsometry in combination with molecular modeling at the force field and DFT levels of theory for the characterization of SAMs of in situ liberated (oligo)phenothiazinyl thiols on Au{111}. The ability of the molecules to form SAMs was investigated by solution adsorption of different systems onto gold films of 100 nm thickness thermally evaporated onto Si wafers using 10 nm of Ti as adhesion promoter. This procedure is known to yield polycrystalline gold films with preferential {111} orientation [55].
The thickness of the layer was determined by ellipsometry as described above. As an estimate for the molecular dimensions of the monolayers, the structures of the (oligo)phenothiazines 2 and 4 were computed at the MM2 and DFT levels of theory (Table 2) [56].
Table 2: Measured (ellipsometry) and calculated (MM2, DFT) layer thickness of (oligo)phenothiazinyl thioacetates 2a, 2c–e, and 4 on Au{111}-coated silicon wafers.
| Compounda | Measured layer thickness dexpb (Å) | Calculated molecule length lmol | Calculated layer thickness dthd | Coverage θe | Monolayer | |||
|---|---|---|---|---|---|---|---|---|
| MM2 (Å) | DFTc (Å) | MM2 (Å) | DFTc (Å) | MM2 (%) | DFTc (%) | |||
| 2a | 9.0 ± 1.00 | 9.04 | 9.10 | 10.4 | 10.5 | 86.6 ± 9.56 | 86.1 ± 9.50 | Yes |
| 2c | 11.4 ± 0.99 | 17.5 | 15.3 | 18.2 | 16.2 | 62.6 ± 5.41 | 70.4 ± 6.09 | Poor |
| 2d | 18.0 ± 1.44 | 19.1 | 21.6 | 19.7 | 22.0 | 91.5 ± 7.32 | 81.9 ± 6.56 | Yes |
| 2e | 10.9 ± 1.61 | 25.5 | 22.4 | 25.6 | 22.7 | 42.6 ± 6.28 | 48.0 ± 7.07 | Poor |
| 4 | 12.9 ± 1.06 | 11.7 | 10.4 | 12.9 | 11.7 | 100.2 ± 8.22 | 110.5 ± 9.07 | Yes |
aThioacetate precursor.
bMeasured by ellipsometry. Errors given are the figures of merit of the least squares fitting routine as determined by the ellipsometer built-in software.
cDFT calculations (B3LYP/3-21G), the hexyl group was replaced by a methyl group [28]. ddth = lmol cos φ + lAu-S; lAu-S = 2.1 Å; φ anthracene-2-thiol = 23°. eθ = dexp/dth.
To minimize computational time in the latter calculations, the hexyl substituents were truncated to methyl groups. From these calculations, the theoretical layer thickness was calculated according to dth = lmol cos φ + lAu–S, where lmol is the calculated length of the respective molecule, φ is the molecules’ tilt angle with the surface normal, and lAu–S = 2.1 Å is the Au–S bond length [57]. For φ, we refer to a recent electron spectroscopic analysis on similar aromatic systems, which determined φ = 23° for anthracene-2-thiol [58]. Using this value, we made the reasonable assumption that the Au–S–C bond is mainly influenced by the adjacent phenyl system. Table 2 shows dth for the different molecules along with the experimental thickness dexp as determined by ellipsometry. The theoretical thicknesses are given for MM2 as well as DFT calculations. As a simple measure of monolayer formation of the different systems, the relative coverage θ obtained experimentally is calculated from θ = dexp/dth as given in Table 2. From these values it is clear that of 2, only 2a and 2d show good SAM formation, suggesting an odd–even effect on film growth, which might be related to steric hindrance during adsorption when an even number of phenothiazine units are present, e.g., because of a back bending of the thiol-bound molecule to the gold surface in these cases, supported by additional gold-π-interactions with the terminal phenothiazine, which thus would hamper the formation of a SAM with an almost parallel intermolecular orientation. In corroboration of such disorder effects, coverage seems to decrease with increasing molecule length for even-numbered molecules (cf. Table 2). The highest coverage was obtained with 4, which is not surprising, because the thiol bifunctionality allows chemisorption of the molecule at either side, which reduces the impact of steric effects on the adsorption kinetics and thus may lead to a more densely packed film. As a consequence, thiolated mono- and terphenothiazines 2 (n = 1, 3) and the dithiolated derivative 4 can be easily self-assembled to give stable monolayers on gold surfaces. This feature makes this class of redox-active molecular entities highly interesting for the fabrication of functionalized electroactive surfaces and nanostructured devices.
Conclusion
In summary we have shown a concise, general synthetic access to (oligo)phenothiazinyl thioacetates that are suitable precursors for the formation of thiol-bound (oligo)phenothiazines on gold surfaces. Whereas the first oligomers are non-fluorescent, the triad and the tetrad display intense greenish-blue fluorescence in addition to distinct multiple reversible oxidation. The in situ deprotection of the thioacetates to thiols in the presence of a gold-coated silicon wafer was used to prepare self-assembled monolayers, which were unambiguously characterized by ellipsometry and accompanying force field and DFT calculations. The chemical trigger of gradual thiol liberation enables better control of film formation and adsorption kinetics, which can be very useful, for example, for co-adsorption of the moieties with a second, nonconductive molecule, which serves as an insulating matrix. Further studies directed toward such more-complex (oligo)phenothiazine SAMs on gold and functionalized redox manipulable surfaces, the nanoscopic characterization of the monolayers as well as their manipulation with external stimuli are currently underway.
Experimental
General considerations
Reagents, catalysts, ligands, and solvents were purchased reagent grade materials and used without further purification. THF and acetyl chloride were dried and distilled according to standard procedures [59]. The bromo phenothiazines 1a [50,51], 1b [50,51], 1c and 1d [32,52] and 3 [50,51], and acetylsulfur chloride [53] were prepared according to literature procedures. Column chromatography: silica gel 60, mesh 70–230. TLC: silica gel coated plates. 1H and 13C NMR spectra: CD2Cl2, CDCl3, and [D6]-acetone (locked to Me4Si) [60]. The assignments of quaternary C, CH, CH2, and CH3 were made by using DEPT spectra. Elemental analyses were carried out in the Microanalytical Laboratories of the Organisch-Chemisches Institut, Ruprecht-Karls-Universität Heidelberg, Germany.
Electrochemistry
Cyclic voltammetry experiments (EG & G potentiostatic instrumentation) were performed under an argon atmosphere in dry and degassed CH2Cl2 at room temperature and at scan rates of 100, 250, 500, and 1000 mV/s. The electrolyte was Bu4NPF6 (0.025 M). The working electrode was a 1 mm platinum disk, the counter-electrode was a platinum wire, and the reference electrode was an Ag/AgCl electrode. The potentials were corrected to the internal standard of Fc/Fc+ in CH2Cl2 (E00/+1 = 450 mV) [61].
7-Bromo-10,10′,10″,10′″-tetrahexyl-10H,10′H,10″H,10′″H-3,3′:7′,3″:7″,3′″-quaterphenothiazine (1e)
1.90 g (2.95 mmol) 7-Bromo-10,10′-dihexyl-10H,10′H-3,3′-biphenothiazine (1c) [32,52], 2.37 g (4.43 mmol) 10-hexyl-3,7-bis-(4,4,5,5-tetramethyl-[1,3,2]-dioxaborolan-2-yl)-10H-phenothiazine [50,51], and 2.45 g (17.7 mmol) potassium bicarbonate were dissolved in 100 mL of DME and 20 mL of water. The mixture was degassed by purging with argon gas for 20 min. After the addition of 136 mg of tetrakis(triphenylphosphan)palladium (118 μmol, 4 mol %), the reaction mixture was stirred for 12 h at 85 °C. After cooling to room temperature, 50 mg of Na2SO3 was added and the reaction mixture stirred for 14 h at room temperature. Then, 3.17 g (6.49 mmol) of 3-bromo-10-hexyl-7-iodo-10H-phenothiazine [51] was added and the mixture stirred for 4 d at 85 °C. After the addition of 100 mL of water, the crude product was extracted several times with dichloromethane. The combined organic phases were dried with magnesium sulfate and the solvents removed in vacuo. The residue was chromatographed on silica gel (hexane/acetone 50:1) to give 1.53 g (36%) of 1e as a yellow resin. 1H NMR (300 MHz, CD2Cl2): δ = 0.76–0.81 (m, 12H), 1.18–1.26 (m, 16H), 1.30–1.40 (m, 8H), 1.63–1.75 (m, 8H), 3.69–3.77 (m, 8H), 6.61–6.64 (m, 2H), 6.78–6.84 (m, 7H), 7.02–7.10 (m, 3H), 7.14–7.31 (m, 13H). 13C NMR (75 MHz, CD2Cl2): δ = 14.2 (CH3), 23.0 (CH2), 26.9 (CH2), 27.0 (CH2), 31.8 (CH2), 31.8 (CH2), 47.9 (CH2), 114.5 (Cquat), 115.9 (CH), 116.0 (CH), 116.9 (CH), 122.7 (CH), 124.6 (CH), 125.2 (CH), 125.4 (CH), 125.5 (CH), 125.7 (CH), 126.9 (Cquat), 127.6 (CH), 129.7 (CH), 130.2 (Cquat), 134.6 (Cquat), 144.7 (Cquat). MS (MALDI-TOF) m/z (%): 1206.3 (M+, 100), 1126.4 (M+ − Br), 1121.2 (M+ − C6H13, 6), 1041.2 (M+ − Br − C6H13, 6). MS (FAB+) m/z (%): 1206.2 (M+, 100), 1121.1 (M+ − C6H13, 45), 1037.0 (M+ − 2C6H13, 10), 951.0 (M+ − 3C6H13, 9), 865.9 (M+ − 4C6H13, 23). IR (KBr): ν = 2953, 2927, 2868, 2854, 1604, 1457, 1415, 1378, 1332, 1295, 1252, 1240, 1193, 1147, 806, 746 cm−1. UV–vis (CH2Cl2): λmax (ε) = 238 (6700), 282 (10500), 326 (3500), 376 nm (4300). Anal. Calcd for C72H77BrN4S4 (1206.6): C, 71.67; H, 6.43; N, 4.64; Br, 6.62; S, 10.63. Found: C, 71.54; H, 6.53; N, 4.64; Br, 6.91; S, 10.58.
Thioacetic acid S-(10-hexyl-10H-phenothiazin-3-yl) ester (2a)
To a cooled solution of 500 mg (1.38 mmol) of 3-bromo-10H-hexylphenothiazine (1a) in 25 mL of dry THF, 0.55 mL (1.52 mmol, 1.1 equiv) of 2.5 M n-butyllithium in hexanes was added dropwise over 5 min at −78 °C (dry ice/acetone). After stirring for 5 min at −78 °C, 49 mg (1.52 mmol, 1.1 equiv) of sulfur was added to the reaction mixture. After stirring for a further 5 min at −78 °C, 0.11 mL (1.52 mmol, 1.1 equiv) of acetyl chloride was added dropwise over 5 min. The solution was allowed to come to room temperature and stirred overnight. Then, 50 mL of water was added and the aqueous phase was extracted several times with small portions of dichloromethane. The combined organic phases were dried with magnesium sulfate and the solvents removed in vacuo. The residue was chromatographed on silica gel (hexane/acetone 10:1) to give 279 mg (57%) of 2a as a yellow oil. Rf (hexane/acetone 5:1) = 0.45. 1H NMR (D6-acetone, 300 MHz): δ = 0.88 (t, 3J = 6.9 Hz, 3H), 1.28 (m, 4H), 1.46 (m, 2H), 1.77 (m, 2H), 2.35 (s, 3H), 3.97 (t, 3J = 7.2 Hz, 2H), 6.94 (m, 1H), 7.08 (m, 2H), 7.13 (m, 2H), 7.24 (m, 2H). 13C NMR (D6-acetone, 75 MHz): δ = 14.2 (CH3), 23.2 (CH2), 27.1 (CH2), 27.4 (CH2), 29.9 (CH3), 32.1 (CH2), 47.9 (CH2), 116.9 (CH), 116.9 (CH), 121.9 (Cquat), 123.8 (CH), 126.2 (Cquat), 126.7 (Cquat), 128.1 (CH), 128.5 (CH), 133.7 (CH), 134.7 (CH), 145.6 (Cquat), 147.5 (Cquat), 194.2 (Cquat). MS (FAB+) m/z (%): 357.3 (M+, 100), 314.3 (M+ − COCH3, 18). IR (film): ν = 3061, 2954, 2928, 2855, 1708, 1593, 1486, 1462, 1393, 1377, 1126, 878, 812, 749, 615 cm−1. UV–vis (CH2Cl2): λmax (ε) = 240 (11600), 266 (24800), 310 nm (6200). Anal. Calcd for C20H23NOS2 (357.1): C, 67.19; H, 6.48; N, 3.92. Found: C, 67.16; H, 6.52; N, 3.86.
Thioacetic acid S-(10-methyl-10H-phenothiazin-3-yl) ester (2b)
To a cooled solution of 292 mg (1.00 mmol) of 3-bromo-10H-methylphenothiazine (1b) in 10 mL of dry THF, 0.7 mL (1.1 mmol, 1.1 equiv) of 1.58 M n-butyllithium in hexanes was added dropwise over 5 min at −78 °C (dry ice/acetone). After stirring for 5 min at −78 °C, 35 mg (1.1 mmol, 1.1 equiv) of sulfur was added to the reaction mixture. After stirring for a further 5 min at −78 °C, 0.07 mL (1.1 mmol, 1.1 equiv) of acetyl chloride was added dropwise over 5 min. The solution was allowed to come to room temperature and stirred overnight. Then, 50 mL of water was added and the aqueous phase extracted several times with small portions of dichloromethane. The combined organic phases were dried with magnesium sulfate and the solvents removed in vacuo. The residue was chromatographed on silica gel (hexane/acetone 50:1) to give 141 mg (49%) of 2b as a yellow oil. Rf (hexane/acetone 5:1) = 0.29. 1H NMR (CD2Cl2, 300 MHz): δ = 2.38 (s, 3H), 3.36 (s, 3H), 6.84 (m, 2H), 6.96 (dt, dJ = 1.2 Hz, tJ = 4.5 Hz, 1H), 7.13 (m, 2H), 7.20 (m, 2H). 13C NMR (CD2Cl2, 75 MHz): δ = 30.2 (CH3), 35.7 (CH3), 114.7 (CH), 114.8 (CH), 121.2 (Cquat), 122.9 (Cquat), 123.1 (CH), 124.5 (Cquat), 127.4 (CH), 127.9 (CH), 133.1 (CH), 134.4 (CH), 145.6 (Cquat), 147.4 (Cquat), 194.8 (Cquat). MS (EI+) m/z (%): 287.0 (M+, 100), 245.0 (M+ − COCH3, 62), 230.3 (M+ − COCH3 − CH3, 68), 212.0 (M+ − SCOCH3, 16). HR-MS (EI+) m/z Calcd for C15H13NOS2: 287.0438. Found: 287.0458. IR (KBr): ν = 3057, 2965, 2883, 2819, 1598 cm−1. UV–vis (CH2Cl2): λmax (ε) = 264 (71200), 316 nm (12800). Anal. Calcd for C15H13NOS2 (287.0): C, 62.69; H, 4.56; N, 4.87. Found: C, 62.68; H, 4.78; N, 4.81.
S-(10,10′-Dihexyl-10H,10′H-3,3′-biphenothiazin-7-yl) ethanethioate (2c)
To a cooled solution of 500 mg (0.78 mmol) of 7-bromo-10,10′-dihexyl-10H,10′H-3,3′-biphenothiazine (1c) in 25 mL of dry THF, 0.91 mL (1.55 mmol, 2.0 equiv) of 1.7 M t-butyllithium in pentane was added dropwise over 5 min at −78 °C (dry ice/acetone). After stirring for 10 min at −78 °C, 95 mg (0.78 mmol, 1.0 equiv) of acetylsulfur chloride [25] was added to the reaction mixture. The solution was allowed to come to room temperature and stirred overnight. Then 30 mL of water was added and the aqueous phase was extracted several times with small portions of dichloromethane. The combined organic phases were dried with magnesium sulfate and the solvents were removed in vacuo. The residue was chromatographed on silica gel (hexane/acetone 50:1) to give 76 mg (15%) of 2c as a yellow resin. Rf (hexane/acetone 5:1) = 0.42. 1H NMR (CD2Cl2, 300 MHz): δ = 0.87 (t, 3J = 6.6 Hz, 6H), 1.31 (m, 8H), 1.43 (m, 4H), 1.79 (m, 4H), 2.36 (s, 3H), 3.85 (t, 3J = 6.9 Hz, 4H), 6.86 (m, 1H), 6.90 (m, 4H), 7.14 (m, 3H), 7.29 (m, 4H), 7.31 (m, 1H). 13C NMR (CD2Cl2, 75 MHz): δ = 14.2 (CH3), 23.0 (CH2), 27.1 (CH2), 27.1 (CH2), 30.1 (CH3), 31.9 (CH2), 48.1 (CH2), 115.8 (CH), 116.0 (CH), 116.2 (CH), 121.2 (Cquat), 122.7 (CH), 124.7 (Cquat), 124.9 (Cquat), 125.3 (Cquat), 125.3 (CH), 125.4 (Cquat), 125.6 (CH), 127.7 (CH), 133.4 (CH), 134.2 (CH), 134.4 (Cquat), 135.1 (Cquat), 143.9 (Cquat), 144.7 (Cquat), 146.9 (Cquat), 194.9 (Cquat). MS (FAB+) m/z (%): 638.6 (M+, 100), 595.5 (M+ − COCH3, 12), 553.4 (M+ − C6H13, 14). HR-MS (FAB+) m/z Calcd for C38H42N2OS3: 638.2459. Found: 683.2445. IR (film): ν = 2960, 2927, 2854, 1706, 1601, 1575, 1459, 1416, 1393, 1377, 1334, 1040, 876, 809, 745, 616 cm−1. UV–vis (CH2Cl2): λmax (ε) = 276 nm (44900), 324 (18900), 366 nm (15400).
S-[10,10′-Dihexyl-7′-(10-hexyl-10H-phenothiazin-3-yl)-10H,10′H-3,3′-biphenothiazin-7-yl] ethanethioate (2d)
To a cooled solution of 800 mg (0.87 mmol) of 7-bromo-10,10′-dihexyl-7′-(10-hexyl-10H-phenothiazin-3-yl)-10H,10′H-3,3′-biphenothiazine (1d) in 25 mL of dry THF, 1.07 mL (1.82 mmol, 2.1 equiv) of 1.7 M t-butyllithium in pentane was added dropwise over 5 min at °C (dry ice/acetone). After stirring for 10 min at −78 °C, 29 mg (0.91 mmol, 1.1 equiv) of sulfur was added to the reaction mixture. After stirring for a further 10 min at −78 °C, 0.07 mL (0.95 mmol, 1.1 equiv) of acetyl chloride was added dropwise over 5 min. The solution was allowed to come to room temperature and stirred overnight. Then, 50 mL of water was added and the aqueous phase extracted several times with small portions of dichloromethane. The combined organic phases were dried with magnesium sulfate and the solvents removed in vacuo. The residue was chromatographed on silica gel (hexane to hexane/acetone 10:1) to give 536 mg (67%) of 2d as a yellow resin. Rf (hexane/acetone 5:1) = 0.35. 1H NMR (CD2Cl2, 300 MHz): δ = 0.88 (t, 3J = 3 Hz, 9H), 1.31 (m, 12H), 1.44 (m, 6H), 1.80 (m, 6H), 2.36 (s, 3H), 3.85 (t, 3J = 6 Hz, 6H), 6.90 (m, 7H), 7.14 (m, 5H), 7.29 (m, 7H). 13C NMR (CD2Cl2, 75 MHz): δ = 14.2 (CH3), 23.0 (CH2), 27.0 (CH2), 27.1 (CH2), 30.2 (CH3), 31.9 (CH2), 36.3 (CH2), 48.0 (CH2), 115.8 (Cquat), 116.0 (CH), 116.2 (CH), 121.1 (Cquat), 122.7 (CH), 124.8 (Cquat), 125.2 (CH), 125.6 (CH), 127.7 (CH), 133.4 (CH), 134.2 (CH), 143.9 (Cquat), 144.7 (Cquat), 146.8 (Cquat), 194.9 (Cquat). MS (MALDI-TOF) m/z (%): 919.4 (M+, 100), 877.4 (M+ − COCH3, 4). IR (KBr): ν = 2954, 2928, 2855, 1700, 1635, 1458, 1416, 1379, 1241, 1193, 873, 807, 747 cm−1. UV–vis (CH2Cl2): λmax (ε) = 280 (101000), 326 (38100), 364 nm (31400). Anal. calcd. for C56H61N3OS4: C, 73.08; H, 6.68; N, 4.57; S, 13.94. Found: C, 73.08; H, 6.60; N, 4.69; S, 13.99.
S-[10,10′,10″-Trihexyl-7′-(10-hexyl-10H-phenothiazin-3-yl)-10H,10′H,10″H-3,3′:7′,3″-terphenothiazin-7-yl] ethanethioate (2e)
To a cooled solution of 200 mg (0.17 mmol) of 7-(7′-bromo-10,10′-dihexyl-10H,10′H-3,3′-biphenothiazin-7-yl)-10,10′-dihexyl-10H,10′H-3,3′-biphenothiazine (1e) in 10 mL of dry THF, 0.20 mL (0.35 mmol, 2.1 equiv) of 1.7 M t-butyllithium in pentane was added dropwise over 5 min at −78 °C (dry ice/acetone). After stirring for 10 min at −78 °C, 6.0 mg (0.17 mmol, 1.0 equiv) of sulfur was added to the reaction mixture. After stirring for a further 10 min at −78 °C, 0.013 mL (0.18 mmol, 1.1 equiv) of acetyl chloride was added dropwise over 5 min. The solution was allowed to come to room temperature and stirred overnight. Then, 20 mL of water was added and the aqueous phase extracted several times with small portions of dichloromethane. The combined organic phases were dried with magnesium sulfate and the solvents removed in vacuo. The residue was chromatographed on silica gel (hexane to hexane/acetone 50:1) to give 132 mg (66%) of 2e as a yellow resin. Rf (hexane/acetone 5:1) = 0.24. 1H NMR (CD2Cl2, 300 MHz): δ = 0.87 (m, 12H), 1.31 (m, 16H), 1.43 (m, 8H), 1.79 (m, 8H), 2.36 (s, 3H), 3.86 (m, 8H), 6.89 (m, 8H), 7.12 (m, 7H), 7.31 (m, 10H). 13C NMR (CD2Cl2, 75 MHz): δ = 14.2 (CH3), 23.0 (CH2), 27.0 (CH2), 30.2 (CH3), 31.9 (CH2), 48.3 (CH2), 115.9 (CH), 121.8 (Cquat), 125.5 (Cquat), 127.8 (Cquat), 131.1 (CH), 132.8 (CH), 133.5 (CH), 134.3 (CH), 140.1 (Cquat), 140.8 (Cquat), 146.3 (Cquat), 148.1 (Cquat), 194.9 (Cquat). MS (MALDI-TOF) m/z: 1200.5 (M+), 1158.5 (M+ − COCH3), 1126.5 (M+ − SCOCH3), 1116.4 (M+ − C6H13). IR (KBr): ν = 2955, 2928, 2854, 1706, 1634, 1604, 1575, 1459, 1415, 1379, 1333, 1241, 1192, 1106, 1062, 874, 807, 746, 616 cm−1. UV/vis (CH2Cl2): λmax (ε) = 282 (92900), 326 (33800), 362 nm (31000).
S,S′-(10-Hexyl-10H-phenothiazine-3,7-diyl) bis(ethanethioate) (4)
To a cooled solution of 800 mg (1.82 mmol) of 3,7-dibromo-10H-hexylphenothiazine (3) in 25 mL of dry THF, 4.38 mL (7.44 mmol, 4.1 equiv) of 1.7 M t-butyllithium in pentane was added dropwise over 5 min at −78 °C (dry ice/acetone). After stirring for 5 min at −78 °C, 122 mg (3.81 mmol, 2.1 equiv) sulfur was added to the reaction mixture. After stirring for a further 10 min at −78 °C, 0.27 mL (3.72 mmol, 2.1 equiv) of acetyl chloride was added dropwise over 5 min. The solution was allowed to come to room temperature and stirred overnight. Then, 50 mL of water was added and the aqueous phase extracted several times with small portions of dichloromethane. The combined organic phases were dried with magnesium sulfate and the solvents removed in vacuo. The residue was chromatographed on silica gel (hexane to hexane/acetone 50:1) to give 317 mg (41%) of 4 as a yellow oil. Rf (hexane/acetone 5:1) = 0.18. 1H NMR (CD2Cl2, 300 MHz): δ = 0.91 (t, 3J = 6.9 Hz, 3H), 1.33 (m, 4H), 1.42 (m, 2H), 1.80 (m, 2H), 2.38 (s, 6H), 3.85 (t, 3J = 7.2 Hz, 2H), 6.88 (m, 1H), 6.90 (m, 1H), 7.12 (m, 2H), 7.17 (m, 1H), 7.20 (m, 1H). 13C NMR (CD2Cl2, 75 MHz): δ = 14.2 (CH3), 23.0 (CH2), 26.9 (CH2), 27.0 (CH2), 30.2 (CH3), 31.8 (CH2), 48.2 (CH2), 116.3 (CH), 121.8 (Cquat), 125.2 (Cquat), 133.5 (CH), 134.3 (CH), 146.3 (Cquat), 194.7 (Cquat). MS (MALDI-TOF) m/z: 431.0 (M+), 388 (M+ − COCH3), 356 (M+ − SCOCH3). IR (film): ν = 2955, 2927, 2858, 1707, 1590, 1564, 1463, 1393, 1352, 1332, 1265, 1250, 1124, 949, 813, 615 cm−1. UV–vis (CH2Cl2): λmax (ε) = 272 (36000), 326 nm (7100). Anal. Calcd for C20H25NO2S3 (431.1): C, 61.22; H, 5.84; N, 3.24; S, 22.29. Found: C, 61.27; H, 5.95; N, 3.25; S, 20.70.
SAM preparation and ellipsometry
The (oligo)phenothiazinyl thioacetates 2a, 2c–e, and 4 were dissolved under an argon atmosphere in dry THF to give a 10−4 M solutions. Au-coated silicon wafers (surface area: 1 cm2) were placed in these solutions. Upon the addition of a few drops of a concentrated solution of aqueous ammonia the thioacetates were saponified to liberate the thiol functionality necessary for chemisorption and SAM formation on gold. After 24 h the wafers were removed from the solution and rinsed several times with dry THF.
The thickness of the formed organic layers was determined by means of spectral ellipsometry (M-44, J.A. Woollam, USA) applying a 3-layer model consisting of gold substrate, organic layer, and ambient [62]. The organic layer was described by means of a Cauchy model, with the first two Cauchy parameters chosen such to yield a refractive index of 1.490 at 500 nm, which resulted from a study on biphenylthiolates on gold in excellent agreement with theory [63].
Supporting Information
| Supporting Information File 1: Molecular modeling coordinates of 2a, 2c, 2d, 2e, and 4, cyclic voltammograms of 2a, 2b, 2c, 2d, 2e, and 4, and absorption and emission spectra of 2d and 2e. | ||
| Format: PDF | Size: 407.4 KB | Download |
References
-
Functional Organic Materials – Synthesis, Strategies, and Applications; Müller, T. J. J.; Bunz, U. H. F., Eds.; Wiley-VCH: Weinheim, 2007.
(see e.g., for a monography).
Return to citation in text: [1] -
Timp, G. L. Nanotechnology; AIP Press/Springer: New York, 1999.
Return to citation in text: [1] -
Waser, R. Nanoelectronics and Information Technology: Advanced Electronic Materials and Novel Devices Information Technology; Wiley-VCH: Weinheim, Germany, 2003.
Return to citation in text: [1] -
Joachim, C.; Gimzewski, J. K.; Aviram, A. Nature 2000, 408, 541–548. doi:10.1038/35046000
Return to citation in text: [1] -
Forrest, S. R. Nature 2004, 428, 911–918. doi:10.1038/nature02498
Return to citation in text: [1] -
Tour, J. M. Molecular Electronics: Commercial Insights, Chemistry, Devices, Architecture and Programming; World Scientific: River Edge, NJ, 2003.
Return to citation in text: [1] -
Love, J. C.; Estroff, L. A.; Kriebel, J. K.; Nuzzo, R. G.; Whitesides, G. M. Chem. Rev. 2005, 105, 1103–1170. doi:10.1021/cr0300789
(and references therein).
Return to citation in text: [1] [2] -
Tao, F.; Bernasek, S. L. Chem. Rev. 2007, 107, 1408–1453. doi:10.1021/cr050258d
Return to citation in text: [1] -
Kriegisch, V.; Lambert, C. Top. Curr. Chem. 2005, 258, 257–313. doi:10.1007/b135682
Return to citation in text: [1] -
Otsubo, T.; Aso, Y.; Takimiya, K. J. Mater. Chem. 2002, 12, 2565–2575. doi:10.1039/b203780g
Return to citation in text: [1] -
Ulman, A. Acc. Chem. Res. 2001, 34, 855–863. doi:10.1021/ar0001564
Return to citation in text: [1] -
Ulman, A. Chem. Rev. 1996, 96, 1533–1554. doi:10.1021/cr9502357
Return to citation in text: [1] -
Kumar, A.; Abbott, N. L.; Kim, E.; Biebuyck, H. A.; Whitesides, G. M. Acc. Chem. Res. 1995, 28, 219–226. doi:10.1021/ar00053a003
Return to citation in text: [1] -
Berry, J. F.; Cotton, F. A.; Murillo, C. A. Organometallics 2004, 23, 2503–2506. doi:10.1021/om049979d
Return to citation in text: [1] -
Maya, F.; Flatt, A. K.; Stewart, M. P.; Shen, D. E.; Tour, J. M. Chem. Mater. 2004, 16, 2987–2997. doi:10.1021/cm049504c
Return to citation in text: [1] -
Seminario, J. M.; Zacarias, A. G.; Tour, J. M. J. Am. Chem. Soc. 1999, 121, 411–416. doi:10.1021/ja982234c
Return to citation in text: [1] -
Pearson, D. L.; Tour, J. M. J. Org. Chem. 1997, 62, 1376–1387. doi:10.1021/jo962335y
Return to citation in text: [1] -
Tour, J. M.; Jones, L., II; Pearson, D. L.; Lamba, J. J. S.; Burgin, T. P.; Whitesides, G. M.; Allara, D. L.; Parikh, A. N.; Atre, S. J. Am. Chem. Soc. 1995, 117, 9529–9534. doi:10.1021/ja00142a021
Return to citation in text: [1] [2] [3] [4] [5] -
Reed, M. A.; Zhou, C.; Muller, C. J.; Burgin, T. P.; Tour, J. M. Science 1997, 278, 252–254. doi:10.1126/science.278.5336.252
Return to citation in text: [1] -
Tour, J. M.; Rawlett, A. M.; Kozaki, M.; Yao, Y.; Jagessar, R. C.; Dirk, S. M.; Price, D. W.; Reed, M. A.; Zhou, C.-W.; Chen, J.; Wang, W.; Campbell, I. Chem.–Eur. J. 2001, 7, 5118–5134. doi:10.1002/1521-3765(20011203)7:23<5118::AID-CHEM5118>3.0.CO;2-1
Return to citation in text: [1] [2] [3] -
Bumm, L. A.; Arnold, J. J.; Cygan, M. T.; Dunbar, T. D.; Burgin, T. P.; Jones, L., II; Allara, D. L.; Tour, J. M.; Weiss, P. S. Science 1996, 271, 1705–1707. doi:10.1126/science.271.5256.1705
Return to citation in text: [1] -
Dameron, A. A.; Ciszek, J. W.; Tour, J. M.; Weiss, P. S. J. Phys. Chem. B 2004, 108, 16761–16767. doi:10.1021/jp049442d
Return to citation in text: [1] -
Ciszek, J. W.; Tour, J. M. Tetrahedron Lett. 2004, 45, 2801–2803. doi:10.1016/j.tetlet.2004.02.028
Return to citation in text: [1] -
Shirai, Y.; Cheng, L.; Chen, B.; Tour, J. M. J. Am. Chem. Soc. 2006, 128, 13479–13489. doi:10.1021/ja063451d
Return to citation in text: [1] -
Zhu, Y.; Gergel, N.; Majumdar, N.; Harriott, L. R.; Bean, J. C.; Pu, L. Org. Lett. 2006, 8, 355–358. doi:10.1021/ol0517168
Return to citation in text: [1] [2] -
Oka, H. J. Mater. Chem. 2008, 18, 1927–1934. doi:10.1039/b716919a
Return to citation in text: [1] -
Okamoto, T.; Kuratsu, M.; Kozaki, M.; Hirotsu, K.; Ichimura, A.; Matsushita, T.; Okada, K. Org. Lett. 2004, 6, 3493–3496. doi:10.1021/ol048698z
Return to citation in text: [1] -
Sun, D.; Rosokha, S. V.; Kochi, J. K. J. Am. Chem. Soc. 2004, 126, 1388–1401. doi:10.1021/ja038746v
Return to citation in text: [1] [2] -
Kochi, J. K.; Rathore, R.; Le Maguères, P. J. Org. Chem. 2000, 65, 6826–6836. doi:10.1021/jo000570h
Return to citation in text: [1] -
Nishinaga, T.; Inoue, R.; Matsuura, A.; Komatsu, K. Org. Lett. 2002, 4, 1435–1438. doi:10.1021/ol0255662
Return to citation in text: [1] -
Pan, D.; Phillips, D. L. J. Phys. Chem. A 1999, 103, 4737–4743. doi:10.1021/jp990399h
Return to citation in text: [1] -
Sailer, M.; Franz, A. W.; Müller, T. J. J. Chem.–Eur. J. 2008, 14, 2602–2614. doi:10.1002/chem.200701341
Return to citation in text: [1] [2] [3] [4] -
Franz, A. W.; Popa, L. N.; Müller, T. J. J. Tetrahedron Lett. 2008, 49, 3300–3303. doi:10.1016/j.tetlet.2008.03.071
Return to citation in text: [1] -
Franz, A. W.; Popa, L. N.; Rominger, F.; Müller, T. J. J. Org. Biomol. Chem. 2009, 7, 469–475. doi:10.1039/b814850c
Return to citation in text: [1] -
Barkschat, C. S.; Guckenberger, R.; Müller, T. J. J. Z. Naturforsch. 2009, 64b, 707–718.
Return to citation in text: [1] -
McDowell, J. J. H. Acta Crystallogr., Sect. B: Struct. Crystallogr. Cryst. Chem. 1976, B32, 5–10. doi:10.1107/S0567740876002215
Return to citation in text: [1] -
Uchida, T.; Ito, M.; Kozawa, K. Bull. Chem. Soc. Jpn. 1983, 56, 577–582. doi:10.1246/bcsj.56.577
Return to citation in text: [1] -
Lambert, C.; Kriegisch, V. Langmuir 2006, 22, 8807–8812. doi:10.1021/la061404t
Return to citation in text: [1] -
Barkschat, C. S.; Stoycheva, S.; Himmelhaus, M.; Müller, T. J. J. Chem. Mater. 2010, 22, 52–63. doi:10.1021/cm901514t
Return to citation in text: [1] [2] -
Müller, T. J. J. Tetrahedron Lett. 1999, 40, 6563–6566. doi:10.1016/S0040-4039(99)01402-1
Return to citation in text: [1] -
Krämer, C. S.; Zeitler, K.; Müller, T. J. J. Org. Lett. 2000, 2, 3723–3726. doi:10.1021/ol0066328
Return to citation in text: [1] -
Krämer, C. S.; Müller, T. J. J. Eur. J. Org. Chem. 2003, 3534–3548. doi:10.1002/ejoc.200300250
Return to citation in text: [1] -
Sailer, M.; Nonnenmacher, M.; Oeser, T.; Müller, T. J. J. Eur. J. Org. Chem. 2006, 423–435. doi:10.1002/ejoc.200500539
Return to citation in text: [1] -
Bucci, N.; Müller, T. J. J. Tetrahedron Lett. 2006, 47, 8323–8327. doi:10.1016/j.tetlet.2006.09.076
Return to citation in text: [1] -
Bucci, N.; Müller, T. J. J. Tetrahedron Lett. 2006, 47, 8329–8332. doi:10.1016/j.tetlet.2006.09.075
Return to citation in text: [1] -
Hauck, M.; Schönhaber, J.; Zucchero, A. J.; Hardcastle, K. I.; Müller, T. J. J.; Bunz, U. H. F. J. Org. Chem. 2007, 72, 6714–6725. doi:10.1021/jo070922l
Return to citation in text: [1] -
Ebdrup, S. Synthesis 1998, 1107–1109. doi:10.1055/s-1998-2121
Return to citation in text: [1] -
Ebdrup, S. J. Chem. Soc., Perkin Trans. 1 1998, 1147–1150. doi:10.1039/a705813f
Return to citation in text: [1] -
Ebdrup, S.; Schou Jensen, M.; Vedsø, P. J. Chem. Soc., Perkin Trans. 1 1998, 351–354. doi:10.1039/a704120i
Return to citation in text: [1] -
Krämer, C. S.; Zimmermann, T. J.; Sailer, M.; Müller, T. J. J. Synthesis 2002, 1163–1170. doi:10.1055/s-2002-32527
Return to citation in text: [1] [2] [3] [4] [5] -
Sailer, M.; Gropeanu, R.-A.; Müller, T. J. J. J. Org. Chem. 2003, 68, 7509–7512. doi:10.1021/jo034555z
Return to citation in text: [1] [2] [3] [4] [5] [6] -
Franz, A. W.; Müller, T. J. J. Synthesis 2008, 1121–1125. doi:10.1055/s-2008-1032118
Return to citation in text: [1] [2] [3] -
Grassi, G.; Tyblewski, M.; Bauder, A. Helv. Chim. Acta 1985, 68, 1876–1879. doi:10.1002/hlca.19850680710
Return to citation in text: [1] [2] -
Krämer, C. S.; Zeitler, K.; Müller, T. J. J. Tetrahedron Lett. 2001, 42, 8619–8624. doi:10.1016/S0040-4039(01)01848-2
Return to citation in text: [1] -
Stoycheva, S.; Himmelhaus, M.; Fick, J.; Korniakov, A.; Grunze, M.; Ulman, A. Langmuir 2006, 22, 4170–4178. doi:10.1021/la0531188
Return to citation in text: [1] -
Gaussian 03, Revision C.02; Gaussian, Inc.: Wallingford, CT, 2004.
Return to citation in text: [1] -
Leung, T. Y. B.; Schwartz, P.; Scoles, G.; Schreiber, F.; Ulman, A. Surf. Sci. 2000, 458, 34–52. doi:10.1016/S0039-6028(00)00385-X
Return to citation in text: [1] -
Frey, S.; Stadler, V.; Heister, K.; Eck, W.; Zharnikov, M.; Grunze, M.; Zeysing, B.; Terfort, A. Langmuir 2001, 17, 2408–2415. doi:10.1021/la001540c
Return to citation in text: [1] -
Becker, H. G. O.; Berger, W.; Domschke, G.; Fanghänel, E.; Faust, J.; Fischer, M.; Gentz, F.; Gewald, K.; Gluch, R.; Mayer, R.; Müller, K.; Pavel, D.; Schmidt, H.; Schollberg, K.; Schwetlick, K.; Seiler, E.; Zeppenfeld, G. Organikum, 20th ed.; Johann Ambrosius Barth Verlag: Heidelberg, Leipzig, 1996.
Return to citation in text: [1] -
Hesse, M.; Meier, H.; Zeeh, B. Spektroskopische Methoden in der organischen Chemie; Georg Thieme Verlag: Stuttgart, New York, 1991; p 69.
Return to citation in text: [1] -
Zanello, P. In Ferrocenes; Togni, A.; Hayashi, T., Eds.; VCH: Weinheim, New York, Basel, Cambridge, Tokyo, 1995; pp 317–430.
Return to citation in text: [1] -
Tompkins, H. G.; McGahan, W. A. Spectroscopic Ellipsometry and Reflectivity, A User’s Guide; John Wiley & Sons, Inc., 1999.
Return to citation in text: [1] -
Gilchrist, V. A.; Lu, J. R.; Keddie, J. L.; Staples, E.; Garrett, P. Langmuir 2000, 16, 740–748. doi:10.1021/la9906572
Return to citation in text: [1]
| 54. | Krämer, C. S.; Zeitler, K.; Müller, T. J. J. Tetrahedron Lett. 2001, 42, 8619–8624. doi:10.1016/S0040-4039(01)01848-2 |
| 39. | Barkschat, C. S.; Stoycheva, S.; Himmelhaus, M.; Müller, T. J. J. Chem. Mater. 2010, 22, 52–63. doi:10.1021/cm901514t |
| 55. | Stoycheva, S.; Himmelhaus, M.; Fick, J.; Korniakov, A.; Grunze, M.; Ulman, A. Langmuir 2006, 22, 4170–4178. doi:10.1021/la0531188 |
| 1. |
Functional Organic Materials – Synthesis, Strategies, and Applications; Müller, T. J. J.; Bunz, U. H. F., Eds.; Wiley-VCH: Weinheim, 2007.
(see e.g., for a monography). |
| 7. |
Love, J. C.; Estroff, L. A.; Kriebel, J. K.; Nuzzo, R. G.; Whitesides, G. M. Chem. Rev. 2005, 105, 1103–1170. doi:10.1021/cr0300789
(and references therein). |
| 8. | Tao, F.; Bernasek, S. L. Chem. Rev. 2007, 107, 1408–1453. doi:10.1021/cr050258d |
| 9. | Kriegisch, V.; Lambert, C. Top. Curr. Chem. 2005, 258, 257–313. doi:10.1007/b135682 |
| 10. | Otsubo, T.; Aso, Y.; Takimiya, K. J. Mater. Chem. 2002, 12, 2565–2575. doi:10.1039/b203780g |
| 11. | Ulman, A. Acc. Chem. Res. 2001, 34, 855–863. doi:10.1021/ar0001564 |
| 12. | Ulman, A. Chem. Rev. 1996, 96, 1533–1554. doi:10.1021/cr9502357 |
| 13. | Kumar, A.; Abbott, N. L.; Kim, E.; Biebuyck, H. A.; Whitesides, G. M. Acc. Chem. Res. 1995, 28, 219–226. doi:10.1021/ar00053a003 |
| 14. | Berry, J. F.; Cotton, F. A.; Murillo, C. A. Organometallics 2004, 23, 2503–2506. doi:10.1021/om049979d |
| 26. | Oka, H. J. Mater. Chem. 2008, 18, 1927–1934. doi:10.1039/b716919a |
| 27. | Okamoto, T.; Kuratsu, M.; Kozaki, M.; Hirotsu, K.; Ichimura, A.; Matsushita, T.; Okada, K. Org. Lett. 2004, 6, 3493–3496. doi:10.1021/ol048698z |
| 28. | Sun, D.; Rosokha, S. V.; Kochi, J. K. J. Am. Chem. Soc. 2004, 126, 1388–1401. doi:10.1021/ja038746v |
| 29. | Kochi, J. K.; Rathore, R.; Le Maguères, P. J. Org. Chem. 2000, 65, 6826–6836. doi:10.1021/jo000570h |
| 30. | Nishinaga, T.; Inoue, R.; Matsuura, A.; Komatsu, K. Org. Lett. 2002, 4, 1435–1438. doi:10.1021/ol0255662 |
| 31. | Pan, D.; Phillips, D. L. J. Phys. Chem. A 1999, 103, 4737–4743. doi:10.1021/jp990399h |
| 50. | Krämer, C. S.; Zimmermann, T. J.; Sailer, M.; Müller, T. J. J. Synthesis 2002, 1163–1170. doi:10.1055/s-2002-32527 |
| 51. | Sailer, M.; Gropeanu, R.-A.; Müller, T. J. J. J. Org. Chem. 2003, 68, 7509–7512. doi:10.1021/jo034555z |
| 7. |
Love, J. C.; Estroff, L. A.; Kriebel, J. K.; Nuzzo, R. G.; Whitesides, G. M. Chem. Rev. 2005, 105, 1103–1170. doi:10.1021/cr0300789
(and references therein). |
| 32. | Sailer, M.; Franz, A. W.; Müller, T. J. J. Chem.–Eur. J. 2008, 14, 2602–2614. doi:10.1002/chem.200701341 |
| 33. | Franz, A. W.; Popa, L. N.; Müller, T. J. J. Tetrahedron Lett. 2008, 49, 3300–3303. doi:10.1016/j.tetlet.2008.03.071 |
| 34. | Franz, A. W.; Popa, L. N.; Rominger, F.; Müller, T. J. J. Org. Biomol. Chem. 2009, 7, 469–475. doi:10.1039/b814850c |
| 32. | Sailer, M.; Franz, A. W.; Müller, T. J. J. Chem.–Eur. J. 2008, 14, 2602–2614. doi:10.1002/chem.200701341 |
| 52. | Franz, A. W.; Müller, T. J. J. Synthesis 2008, 1121–1125. doi:10.1055/s-2008-1032118 |
| 6. | Tour, J. M. Molecular Electronics: Commercial Insights, Chemistry, Devices, Architecture and Programming; World Scientific: River Edge, NJ, 2003. |
| 24. | Shirai, Y.; Cheng, L.; Chen, B.; Tour, J. M. J. Am. Chem. Soc. 2006, 128, 13479–13489. doi:10.1021/ja063451d |
| 59. | Becker, H. G. O.; Berger, W.; Domschke, G.; Fanghänel, E.; Faust, J.; Fischer, M.; Gentz, F.; Gewald, K.; Gluch, R.; Mayer, R.; Müller, K.; Pavel, D.; Schmidt, H.; Schollberg, K.; Schwetlick, K.; Seiler, E.; Zeppenfeld, G. Organikum, 20th ed.; Johann Ambrosius Barth Verlag: Heidelberg, Leipzig, 1996. |
| 2. | Timp, G. L. Nanotechnology; AIP Press/Springer: New York, 1999. |
| 3. | Waser, R. Nanoelectronics and Information Technology: Advanced Electronic Materials and Novel Devices Information Technology; Wiley-VCH: Weinheim, Germany, 2003. |
| 4. | Joachim, C.; Gimzewski, J. K.; Aviram, A. Nature 2000, 408, 541–548. doi:10.1038/35046000 |
| 5. | Forrest, S. R. Nature 2004, 428, 911–918. doi:10.1038/nature02498 |
| 25. | Zhu, Y.; Gergel, N.; Majumdar, N.; Harriott, L. R.; Bean, J. C.; Pu, L. Org. Lett. 2006, 8, 355–358. doi:10.1021/ol0517168 |
| 50. | Krämer, C. S.; Zimmermann, T. J.; Sailer, M.; Müller, T. J. J. Synthesis 2002, 1163–1170. doi:10.1055/s-2002-32527 |
| 51. | Sailer, M.; Gropeanu, R.-A.; Müller, T. J. J. J. Org. Chem. 2003, 68, 7509–7512. doi:10.1021/jo034555z |
| 18. | Tour, J. M.; Jones, L., II; Pearson, D. L.; Lamba, J. J. S.; Burgin, T. P.; Whitesides, G. M.; Allara, D. L.; Parikh, A. N.; Atre, S. J. Am. Chem. Soc. 1995, 117, 9529–9534. doi:10.1021/ja00142a021 |
| 20. | Tour, J. M.; Rawlett, A. M.; Kozaki, M.; Yao, Y.; Jagessar, R. C.; Dirk, S. M.; Price, D. W.; Reed, M. A.; Zhou, C.-W.; Chen, J.; Wang, W.; Campbell, I. Chem.–Eur. J. 2001, 7, 5118–5134. doi:10.1002/1521-3765(20011203)7:23<5118::AID-CHEM5118>3.0.CO;2-1 |
| 21. | Bumm, L. A.; Arnold, J. J.; Cygan, M. T.; Dunbar, T. D.; Burgin, T. P.; Jones, L., II; Allara, D. L.; Tour, J. M.; Weiss, P. S. Science 1996, 271, 1705–1707. doi:10.1126/science.271.5256.1705 |
| 20. | Tour, J. M.; Rawlett, A. M.; Kozaki, M.; Yao, Y.; Jagessar, R. C.; Dirk, S. M.; Price, D. W.; Reed, M. A.; Zhou, C.-W.; Chen, J.; Wang, W.; Campbell, I. Chem.–Eur. J. 2001, 7, 5118–5134. doi:10.1002/1521-3765(20011203)7:23<5118::AID-CHEM5118>3.0.CO;2-1 |
| 57. | Leung, T. Y. B.; Schwartz, P.; Scoles, G.; Schreiber, F.; Ulman, A. Surf. Sci. 2000, 458, 34–52. doi:10.1016/S0039-6028(00)00385-X |
| 18. | Tour, J. M.; Jones, L., II; Pearson, D. L.; Lamba, J. J. S.; Burgin, T. P.; Whitesides, G. M.; Allara, D. L.; Parikh, A. N.; Atre, S. J. Am. Chem. Soc. 1995, 117, 9529–9534. doi:10.1021/ja00142a021 |
| 20. | Tour, J. M.; Rawlett, A. M.; Kozaki, M.; Yao, Y.; Jagessar, R. C.; Dirk, S. M.; Price, D. W.; Reed, M. A.; Zhou, C.-W.; Chen, J.; Wang, W.; Campbell, I. Chem.–Eur. J. 2001, 7, 5118–5134. doi:10.1002/1521-3765(20011203)7:23<5118::AID-CHEM5118>3.0.CO;2-1 |
| 22. | Dameron, A. A.; Ciszek, J. W.; Tour, J. M.; Weiss, P. S. J. Phys. Chem. B 2004, 108, 16761–16767. doi:10.1021/jp049442d |
| 23. | Ciszek, J. W.; Tour, J. M. Tetrahedron Lett. 2004, 45, 2801–2803. doi:10.1016/j.tetlet.2004.02.028 |
| 58. | Frey, S.; Stadler, V.; Heister, K.; Eck, W.; Zharnikov, M.; Grunze, M.; Zeysing, B.; Terfort, A. Langmuir 2001, 17, 2408–2415. doi:10.1021/la001540c |
| 18. | Tour, J. M.; Jones, L., II; Pearson, D. L.; Lamba, J. J. S.; Burgin, T. P.; Whitesides, G. M.; Allara, D. L.; Parikh, A. N.; Atre, S. J. Am. Chem. Soc. 1995, 117, 9529–9534. doi:10.1021/ja00142a021 |
| 19. | Reed, M. A.; Zhou, C.; Muller, C. J.; Burgin, T. P.; Tour, J. M. Science 1997, 278, 252–254. doi:10.1126/science.278.5336.252 |
| 15. | Maya, F.; Flatt, A. K.; Stewart, M. P.; Shen, D. E.; Tour, J. M. Chem. Mater. 2004, 16, 2987–2997. doi:10.1021/cm049504c |
| 16. | Seminario, J. M.; Zacarias, A. G.; Tour, J. M. J. Am. Chem. Soc. 1999, 121, 411–416. doi:10.1021/ja982234c |
| 17. | Pearson, D. L.; Tour, J. M. J. Org. Chem. 1997, 62, 1376–1387. doi:10.1021/jo962335y |
| 18. | Tour, J. M.; Jones, L., II; Pearson, D. L.; Lamba, J. J. S.; Burgin, T. P.; Whitesides, G. M.; Allara, D. L.; Parikh, A. N.; Atre, S. J. Am. Chem. Soc. 1995, 117, 9529–9534. doi:10.1021/ja00142a021 |
| 28. | Sun, D.; Rosokha, S. V.; Kochi, J. K. J. Am. Chem. Soc. 2004, 126, 1388–1401. doi:10.1021/ja038746v |
| 37. | Uchida, T.; Ito, M.; Kozawa, K. Bull. Chem. Soc. Jpn. 1983, 56, 577–582. doi:10.1246/bcsj.56.577 |
| 35. | Barkschat, C. S.; Guckenberger, R.; Müller, T. J. J. Z. Naturforsch. 2009, 64b, 707–718. |
| 50. | Krämer, C. S.; Zimmermann, T. J.; Sailer, M.; Müller, T. J. J. Synthesis 2002, 1163–1170. doi:10.1055/s-2002-32527 |
| 51. | Sailer, M.; Gropeanu, R.-A.; Müller, T. J. J. J. Org. Chem. 2003, 68, 7509–7512. doi:10.1021/jo034555z |
| 36. | McDowell, J. J. H. Acta Crystallogr., Sect. B: Struct. Crystallogr. Cryst. Chem. 1976, B32, 5–10. doi:10.1107/S0567740876002215 |
| 53. | Grassi, G.; Tyblewski, M.; Bauder, A. Helv. Chim. Acta 1985, 68, 1876–1879. doi:10.1002/hlca.19850680710 |
| 60. | Hesse, M.; Meier, H.; Zeeh, B. Spektroskopische Methoden in der organischen Chemie; Georg Thieme Verlag: Stuttgart, New York, 1991; p 69. |
| 32. | Sailer, M.; Franz, A. W.; Müller, T. J. J. Chem.–Eur. J. 2008, 14, 2602–2614. doi:10.1002/chem.200701341 |
| 52. | Franz, A. W.; Müller, T. J. J. Synthesis 2008, 1121–1125. doi:10.1055/s-2008-1032118 |
| 63. | Gilchrist, V. A.; Lu, J. R.; Keddie, J. L.; Staples, E.; Garrett, P. Langmuir 2000, 16, 740–748. doi:10.1021/la9906572 |
| 53. | Grassi, G.; Tyblewski, M.; Bauder, A. Helv. Chim. Acta 1985, 68, 1876–1879. doi:10.1002/hlca.19850680710 |
| 50. | Krämer, C. S.; Zimmermann, T. J.; Sailer, M.; Müller, T. J. J. Synthesis 2002, 1163–1170. doi:10.1055/s-2002-32527 |
| 51. | Sailer, M.; Gropeanu, R.-A.; Müller, T. J. J. J. Org. Chem. 2003, 68, 7509–7512. doi:10.1021/jo034555z |
| 25. | Zhu, Y.; Gergel, N.; Majumdar, N.; Harriott, L. R.; Bean, J. C.; Pu, L. Org. Lett. 2006, 8, 355–358. doi:10.1021/ol0517168 |
| 18. | Tour, J. M.; Jones, L., II; Pearson, D. L.; Lamba, J. J. S.; Burgin, T. P.; Whitesides, G. M.; Allara, D. L.; Parikh, A. N.; Atre, S. J. Am. Chem. Soc. 1995, 117, 9529–9534. doi:10.1021/ja00142a021 |
| 62. | Tompkins, H. G.; McGahan, W. A. Spectroscopic Ellipsometry and Reflectivity, A User’s Guide; John Wiley & Sons, Inc., 1999. |
| 40. | Müller, T. J. J. Tetrahedron Lett. 1999, 40, 6563–6566. doi:10.1016/S0040-4039(99)01402-1 |
| 41. | Krämer, C. S.; Zeitler, K.; Müller, T. J. J. Org. Lett. 2000, 2, 3723–3726. doi:10.1021/ol0066328 |
| 42. | Krämer, C. S.; Müller, T. J. J. Eur. J. Org. Chem. 2003, 3534–3548. doi:10.1002/ejoc.200300250 |
| 43. | Sailer, M.; Nonnenmacher, M.; Oeser, T.; Müller, T. J. J. Eur. J. Org. Chem. 2006, 423–435. doi:10.1002/ejoc.200500539 |
| 44. | Bucci, N.; Müller, T. J. J. Tetrahedron Lett. 2006, 47, 8323–8327. doi:10.1016/j.tetlet.2006.09.076 |
| 45. | Bucci, N.; Müller, T. J. J. Tetrahedron Lett. 2006, 47, 8329–8332. doi:10.1016/j.tetlet.2006.09.075 |
| 46. | Hauck, M.; Schönhaber, J.; Zucchero, A. J.; Hardcastle, K. I.; Müller, T. J. J.; Bunz, U. H. F. J. Org. Chem. 2007, 72, 6714–6725. doi:10.1021/jo070922l |
| 50. | Krämer, C. S.; Zimmermann, T. J.; Sailer, M.; Müller, T. J. J. Synthesis 2002, 1163–1170. doi:10.1055/s-2002-32527 |
| 51. | Sailer, M.; Gropeanu, R.-A.; Müller, T. J. J. J. Org. Chem. 2003, 68, 7509–7512. doi:10.1021/jo034555z |
| 47. | Ebdrup, S. Synthesis 1998, 1107–1109. doi:10.1055/s-1998-2121 |
| 48. | Ebdrup, S. J. Chem. Soc., Perkin Trans. 1 1998, 1147–1150. doi:10.1039/a705813f |
| 49. | Ebdrup, S.; Schou Jensen, M.; Vedsø, P. J. Chem. Soc., Perkin Trans. 1 1998, 351–354. doi:10.1039/a704120i |
| 51. | Sailer, M.; Gropeanu, R.-A.; Müller, T. J. J. J. Org. Chem. 2003, 68, 7509–7512. doi:10.1021/jo034555z |
| 38. | Lambert, C.; Kriegisch, V. Langmuir 2006, 22, 8807–8812. doi:10.1021/la061404t |
| 61. | Zanello, P. In Ferrocenes; Togni, A.; Hayashi, T., Eds.; VCH: Weinheim, New York, Basel, Cambridge, Tokyo, 1995; pp 317–430. |
| 39. | Barkschat, C. S.; Stoycheva, S.; Himmelhaus, M.; Müller, T. J. J. Chem. Mater. 2010, 22, 52–63. doi:10.1021/cm901514t |
| 32. | Sailer, M.; Franz, A. W.; Müller, T. J. J. Chem.–Eur. J. 2008, 14, 2602–2614. doi:10.1002/chem.200701341 |
| 52. | Franz, A. W.; Müller, T. J. J. Synthesis 2008, 1121–1125. doi:10.1055/s-2008-1032118 |
© 2010 Franz et al; licensee Beilstein-Institut.
This is an Open Access article under the terms of the Creative Commons Attribution License (http://creativecommons.org/licenses/by/2.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
The license is subject to the Beilstein Journal of Organic Chemistry terms and conditions: (http://www.beilstein-journals.org/bjoc)