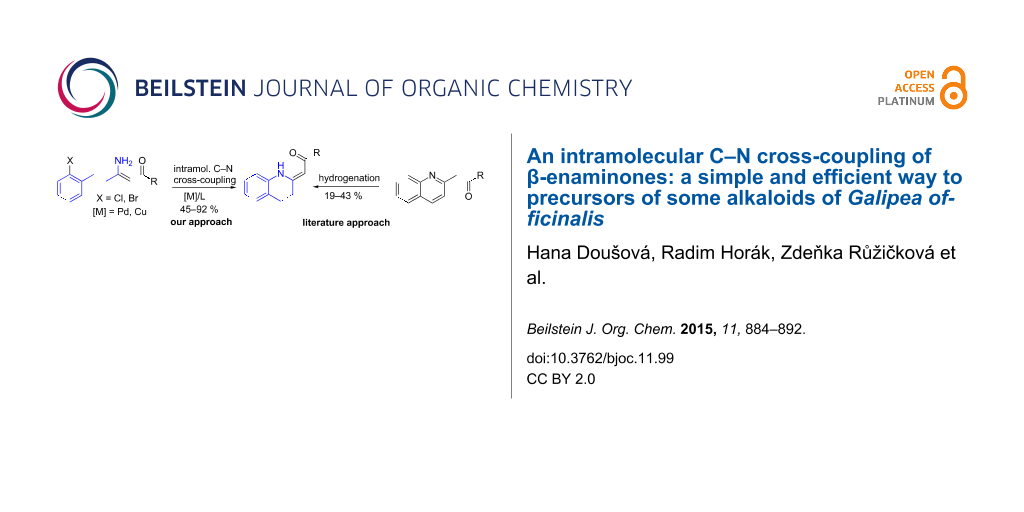
Abstract
2-Aroylmethylidene-1,2,3,4-tetrahydroquinolines with the appropriate substituents can be suitable precursors for the synthesis of alkaloids from Galipea officinalis (cuspareine, galipeine, galipinine, angustureine). However, only two, rather low-yielding procedures for their synthesis are described in the literature. We have developed a simple and efficient protocol for an intramolecular, palladium or copper-catalysed amination of both chloro- and bromo-substituted 3-amino-1,5-diphenylpent-2-en-1-ones leading to the above-mentioned tetrahydroquinoline moiety. The methodology is superior to the methods published to date.
Graphical Abstract
Introduction
Galipea officinalis Hancock is a Venezuelan shrub, the bark (angostura bark) of which is used in folk medicine for stimulation in the cure of some paralytic diseases [1] and for the treatment of fever [2]. In addition, antituberculous [3], antiplasmodial and cytotoxic [2] properties have been also described. The active constituents of the bark are mainly tetrahydroquinoline alkaloids such as galipinine, galipeine, cuspareine and angustureine (Figure 1) [1,4].
Figure 1: Tetrahydroquinoline alkaloids of Galipea officinalis.
Figure 1: Tetrahydroquinoline alkaloids of Galipea officinalis.
The synthesis of these alkaloids has attracted great interest in organic chemists and many procedures have been published to date [5-30]. As part of our ongoing interest in the preparation of polarized ethylenes and their application in organic synthesis, we have been attracted by the procedure published by Zhou [30]. Here, the authors used heterocyclic enaminone 1b as the reactant for an asymmetric reduction followed by N-methylation to give (S)-cuspareine in high yields and excellent stereoselectivity (Scheme 1).
Scheme 1: Enaminone-based synthesis of (S)-cuspareine.
Scheme 1: Enaminone-based synthesis of (S)-cuspareine.
This method, however, suffers from a low-yielding (28%) synthesis of the enaminone 1a [30]. Only two methods for the synthesis of tetrahydroquinolines 1 have been described in the literature [30-32] based on a catalytic hydrogenation of the corresponding quinolones 2 (Scheme 2) with yields not greater than 43%. In the present work, we introduce a quite different approach to the exocyclic enaminones 1 based on an intramolecular C–N cross-coupling of enaminones 3 (Scheme 2).
Scheme 2: The approaches to 2-aroylmethylidene-1,2,3,4-tetrahydroquinolines 1.
Scheme 2: The approaches to 2-aroylmethylidene-1,2,3,4-tetrahydroquinolines 1.
Results and Discussion
The synthesis of the starting substrates
A simple retrosynthesis of enaminones 3 leads to the corresponding β-diketones 4 accessible through Claisen condensation of 3-phenylpropionic ester 5 with the appropriate acetophenone 6 (Scheme 3).
Scheme 3: The retrosynthetic analysis of the starting substrates for C–N cross-coupling.
Scheme 3: The retrosynthetic analysis of the starting substrates for C–N cross-coupling.
The synthesis of ester 5 was accomplished according to Scheme 4. The classic Knoevenagel/reduction/hydrolysis/decarboxylation pathway (steps a–d in Scheme 4) to carboxylic acid 10 suffered from low yields of the last two steps (total yield 33%). The attempt to obtain 5a directly from 9 by means of Krapcho decarboxylation [33] failed, as the product was contaminated with inseparable byproducts. A better and less time-consuming route to 10 consists of the reaction of 2-halobenzaldehyde 7 with Meldrum´s acid in the presence of HCOOH/Et3N system with a total yield of 61–67% (step f in Scheme 4). The last step for both methods was the esterification of acids 10a,b (step e in Scheme 4).
Scheme 4: The synthesis of methyl 3-phenylpropionates. Conditions: (a) piperidine, PhCOOH, toluene, reflux 4 h; (b) NaBH4, MeOH/MeCN, rt, 3.5 h; (c) KOH, H2O, reflux, 8 h; (d) H2SO4, H2O, reflux, 20 h; (e) MeOH, SOCl2, reflux, 4 h; (f) Meldum’s acid, HCOOH, Et3N, 100 °C, 4 h.
Scheme 4: The synthesis of methyl 3-phenylpropionates. Conditions: (a) piperidine, PhCOOH, toluene, reflux 4 ...
β-Diketones 4 were obtained using tert-butoxide or tert-pentoxide mediated Claisen condensation of esters 5 with the appropriate acetophenones 6a–d (Scheme 5, step c). The substitution pattern on compounds 6 was chosen so that the final products 1 are the precursors for the synthesis of galipinine, galipeine and angustureine (Figure 1). 3-Hydroxy-4-methoxyacetophenone (6e) was prepared by selective deprotection of commercially available 3,4-dimethoxyacetophenone (6b) [34] (Scheme 5, step a). The hydroxy group in 6e was then protected with a benzyl group (step b). The final step d was the reaction of β-diketones 4 with ammonium surrogate (AcONH4 or NH4HCO3). The regioselectivity of the synthesis was checked by means of 2D 1H–13C HMBC (See Supporting Information File 1, page S69). The non-equivalence of NH2 protons together with the relatively high chemical shift of one resonance of the pair (δ ≈ 10 ppm) indicates the presence of an intramolecular hydrogen bond N–H···O. The enaminones 3 therefore possess Z configuration on the C=C bond.
Scheme 5: The synthesis of the starting β-enaminones. Conditions: (a) H2SO4, 65 °C, 46 h; (b) 1. t-BuOK/THF, rt, 30 min, 2. BnBr, reflux 5 h; (c) t-BuOK, t-BuONa or t-AmOK, THF, rt overnight; (d) AcONH4, MeOH, reflux 5 h (NH4HCO3, MeOH/THF, rt 24 h for 3d).
Scheme 5: The synthesis of the starting β-enaminones. Conditions: (a) H2SO4, 65 °C, 46 h; (b) 1. t-BuOK/THF, ...
The intramolecular C–N cross-coupling
β-Enaminones and related polarized ethylenes (generally enamines substituted by EWG on β-carbon atom) belong among the rather neglected molecules from a C–N cross-coupling perspective. There are relatively few works (about ten) dealing with these very useful molecules [35-46], in comparison to the hundreds of papers dedicated to the other substrates. Due to their electronic nature, β-enaminones can be considered vinylogous amides. Hence, their nucleophilicity is lowered and enaminones can be more challenging substrates for C–N cross-coupling compared to ordinary enamines.
We used 3a as the model substrate for the optimization study. The reaction conditions were surveyed from the following aspects: catalytic system [M]/[L] and base (Table 1).
Table 1: Optimisation study for C–N cross-coupling of bromo derivatives.
|
|
|||||
| Entry | [M]/% | [L]/%a | Base/equiv | Time/h | Conversionb |
|---|---|---|---|---|---|
| 1c | Pd2(dba)3/2.5 | L1/5 | Cs2CO3/1.6 | 3 | 87 |
| 2c | Pd2(dba)3/2.5 | L1/5 | Cs2CO3/1.6 | 18 | 87 |
| 3c | Pd2(dba)3/3.5 | L1/7 | Cs2CO3/1.4 | 2 | 94 (92) |
| 4c | Pd2(dba)3/3.5 | L5/7 | Cs2CO3/1.4 | 17 | 96 (88) |
| 5d | Pd(OAc)2/5 | L1/15 | Cs2CO3/1.4 | 21 | 0 |
| 6e | CuI/10 | L7/20 | Cs2CO3/1.4 | 18 | 0 |
| 7e | CuI/10 | L8/20 | Cs2CO3/1.4 | 18 | >99 (83) |
| 8e | CuI/10 | L8/20 | K3PO4/2 | 20 | 12 |
| 9e | CuI/10 | L8/20 | K2CO3/2 | 20 | 0 |
aFor ligands see Figure 2. bConversion determined by means of 1H NMR. Isolated yields in parentheses. cMethod A (See Experimental and Supporting Information File 1). dMethod B. Water-mediated pre-activation used [47]. (See Supporting Information File 1). eMethod C. (See Supporting Information File 1).
We used tris(dibenzylideneacetone)dipalladium(0) as the starting palladium source. To suppress the problems with the attenuation of its catalytic activity due to the coordination of dba ligands to the central metal, we applied preheating of Pd2(dba)3 with the corresponding ligand [L] to generate the active catalyst [48] prior to the addition to substrate 3 (see Method A in the Experimental). Dialkylbiarylphosphines currently belong among the most common ligands for palladium-catalyzed C–N cross-coupling [48,49]. The use of L1 (Figure 2) in combination with Pd2(dba)3 in toluene led to the successful formation of product 1a (Table 1, entries 1–3). Rather, a higher amount of palladium was necessary for completing the reaction in a short reaction time (Table 1, entry 3). Switching to the bidentate ligand L5 (Figure 2) gave comparable conversion but over a substantially longer period (Table 1, entry 4). Buchwald et al. [47] described a protocol for generation of the highly active Pd(0) catalyst from Pd(OAc)2 using water-mediated pre-activation. However, no conversion was observed here for L1 (Table 1, entry 5).
Figure 2: Ligands for C–N cross-coupling used in this work.
Figure 2: Ligands for C–N cross-coupling used in this work.
Besides palladium, copper is another widely applied metal for C–N bond formation [50-52]. We then applied the CuI/[L] catalytic system to 3a (see method C in Supporting Information File 1, pages S25 and S26). The choice of the ligand is crucial here, as L7 (Figure 2) had no effect (Table 1, entry 6) whereas using another common ligand L8 (Figure 2) led to the full conversion to 1a (Table 1, entry 7). Caesium carbonate was the optimal base as neither K2CO3 nor K3PO4 gave satisfactory results (Table 1, entries 8 and 9).
From the above-mentioned facts, summarized in Table 1, it follows that the optimal results were obtained using catalytic systems Pd2(dba)3/L1 (Table 1, entry 3) or CuI/L8 (Table 1, entry 7) in toluene with caesium carbonate as the base. We further preferred the first one (Table 1, entry 3) both due to the lower amount of the catalyst and shorter reaction time. Nevertheless, the copper-mediated variant could be interesting for preparations on a larger scale.
With the conditions chosen for the bromo derivatives, we turned our attention to the chloro derivatives. As chloro derivatives are more challenging substrates for cross-coupling reactions, we started with the successful catalytic system (Table 1, entry 3) but with an increased amount of palladium, applied to 3e as the model substrate. However these conditions failed (Table 2, entry 1). Changing the ligand to DavePhos (L3) or BINAP (L5) (Figure 2) did not improve the situation at all (Table 2, entries 2 and 3). The application of a palladacycle-based pre-catalyst L6 (Figure 2) (see method D in Supporting Information File 1, pages S25 and S26), introduced by Buchwald et al. [53] failed as well (Table 2, entries 4–6). The sterically more demanding ligand BrettPhos (L4, Figure 2), reported as a very effective ligand for N-arylations of primary amino groups [48,54], worked better but was still not satisfactory (Table 2, entry 7). The breakthrough was made after applying t-BuXPhos (L2, Figure 2), which led to 64% conversion (Table 2, entry 8). Switching from toluene to t-AmOH finally led to full conversion to the desired product (Table 2, entry 9). An attempt to reduce the amount of catalyst, however, only led to a decrease in the conversion (Table 2, entry 10). The copper-catalyzed protocol was quite unsuccessful (Table 2, entry 11). Generally, chloro derivatives (and especially non-activated ones) remain challenging substrates for copper catalysis [50]. The conditions in Table 2, entry 9 (5 mol % Pd2(dba)3, 10 mol % t-BuXPhos, Cs2CO3 in t-AmOH) were thus the best for the cyclization of chloro derivatives.
Table 2: Optimisation study for C–N cross-coupling of chloro derivatives.
|
|
|||||
| Entry | [Pd]/% | [L]/%a | Solvent | Time/h | Conversion/%b |
|---|---|---|---|---|---|
| 1c | Pd2(dba)3/5 | L1/10 | toluene | 48 | 0 |
| 2c | Pd2(dba)3/5 | L3/10 | toluene | 24 | 0 |
| 3c | Pd2(dba)3/5 | L5/10 | toluene | 48 | 0 |
| 4d,e | L6/3 | toluene | 24 | 0 | |
| 5d,e | L6/3 | DMF | 24 | 4 | |
| 6d,e | L6/3 | t-AmOH | 24 | 0 | |
| 7c | Pd2(dba)3/5 | L4/10 | toluene | 22 | 29 |
| 8c | Pd2(dba)3/5 | L2/10 | toluene | 22 | 64 |
| 9c | Pd2(dba)3/5 | L2/10 | t-AmOH | 17 | 100 (72)f |
| 10c | Pd2(dba)3/2.5 | L5/5 | t-AmOH | 24 | 47 |
| 11g | CuI/10 | L7/20 | toluene | 24 | 0 |
aFor ligands see Figure 2. bConversion determined by means of 1H NMR. Isolated yields in parentheses. cMethod A. dMethod D. eTwo equivalents of the base used. fIsolated yield. gMethod C.
These conditions, however, did not work for the bromo derivatives, despite the higher amount of both palladium and ligand and a substantially longer reaction time. Compound 3b was transformed into 1b using the above-mentioned conditions (Table 2, entry 9) only in 20% conversion.
Having the conditions for the successful cyclization of enaminones 3a,e to tetrahydroquinoline 1a in hand, we applied them to other enaminones 3. In all of these cases, the corresponding tetrahydroquinolines 1 were obtained in moderate to high yields (Table 3).
Table 3: The intramolecular C–N cross-coupling of enaminones 3: a summary of results.
|
|
||||||
| Producta | Reactant | General procedureb | Solvent | [L] | Base | Yield[%]/Conv.[%] |
|---|---|---|---|---|---|---|
|
(p. S27) |
3a |
A
C |
toluene
toluene |
L1
L8 |
Cs2CO3
Cs2CO3 |
92
83 |
| 3e | Ac | t-AmOH | L2 | Cs2CO3 | 72 | |
|
(p. S28) |
3b |
A
A A B |
toluene
toluene toluene toluene |
L1
L1 L1 L1 |
Cs2CO3
K2CO3 K3PO4 Cs2CO3 |
65
/29 /0 /0 |
| 3f | A | t-AmOH | L2 | Cs2CO3 | 72 | |
|
(p. S29) |
3c | Ad | toluene | L1 | Cs2CO3 | 85/50e |
| 3g | A | t-AmOH | L2 | Cs2CO3 | 82 | |
|
(p. S30) |
3d | A | toluene | L1 | Cs2CO3 | 45 |
| 3h | A | t-AmOH | L2 | Cs2CO3 | 61 | |
aSee Supporting Information File 1 for details. bFor procedures see Supporting Information File 1 p. S25 (methods A,B) or p. S26 (methods C,D). cFor 2.5 mol % Pd2(dba)3, 5 mol % L2 conversion 47%. d5 mol % of Pd2(dba)3 and 10 mol % L1 used. eConversion for 3.5 mol % of Pd2(dba)3 and 7 mol % L1.
The deprotection of the hydroxy group in 1c was accomplished using BCl3 in DCM (Figure 3) to obtain the precursor 1e for galipeine (Figure 1).
Figure 3: Deprotection of the hydroxy group in 1c to give the Galipein precursor 1e.
Figure 3: Deprotection of the hydroxy group in 1c to give the Galipein precursor 1e.
On the basis of relatively high chemical shifts of NH protons in compounds 1 (δ > 12), it can be assumed that all of these compounds have Z configuration on the C=C double bond (increased chemical shifts due to the presence of an intramolecular hydrogen bond C=O···H–N). This assumption was confirmed by means of X-ray characterization of the compound 1a (Figure 4).
![[1860-5397-11-99-4]](/bjoc/content/figures/1860-5397-11-99-4.png?scale=1.2&max-width=1024&background=FFFFFF)
Figure 4: ORTEP (50% probability level) view for compound 1a. For selected parameters see Supporting Information File 1.
Figure 4: ORTEP (50% probability level) view for compound 1a. For selected parameters see Supporting Information File 1.
According to the literature [55], as well as the Cambridge Structural Database, there is a plethora of compounds with an intramolecular N–H···O=C contact like 1a; on the other hand, the family of structurally related 1,2-dihydroquinolines [56] and ethanones [57] is limited to only seven examples. In the structure of 1a, some extent of π-electron delocalization is reflected in slight shortening of the formal single bond between C1 and C2 atoms, on the contrary to a slight elongation of C2–C3 and C1–O1 distances – formally the double bonds (for crystal data see Supporting Information File 1, pages S27 and S28), similarly to the situation found for peptide type of bonding [55].
Conclusion
An asymmetric reduction of suitably substituted 2-aroylmethylidene-1,2,3,4-tetrahydroquinolines is one of the possible routes to tetrahydroquinoline alkaloids of Galipea officinalis. The methodology, however, suffered from the unsatisfactory source of the 2-aroylmethylidene reactants. In this work, we have established a novel, simple protocol for the synthesis of the above-mentioned 2-aroylmethylidene-1,2,3,4-tetrahydroquinolines, which is superior to the methods published so far. The methodology is based on an intramolecular C–N cross-coupling of acyclic β-enaminones. The reaction conditions are described for the successful cyclization of both bromo and chloro derivatives. The crucial factors here are the ligand and the solvent. The best system for bromo derivatives is Pd2(dba)3/XPhos/Cs2CO3 in toluene, although the transformation is also feasible using CuI/DESA/Cs2CO3 in toluene, albeit for a substantially longer time than in the palladium-catalyzed version. The more challenging chloro derivatives required higher amounts of palladium, different ligands and solvents, together with much longer reaction times. Pd2(dba)3/t-BuXPhos/Cs2CO3 in t-AmOH worked best. Due to the importance of polarized ethylenes, the extension of the methodology to other substrates and substituents could be useful and is the subject of thorough examination nowadays.
Experimental
For analytical and synthetic procedures as well as characterization data of individual compounds and copies of their NMR spectra see Supporting Information File 1.
Representative procedure for palladium-catalyzed synthesis of tetrahydroquinolines 1a–d (Method A)
An oven-dried vial equipped with a magnetic stir bar and fitted with a Teflon septum was charged with Pd2(dba)3 and the corresponding ligand. The vessel was evacuated three times and backfilled with argon. Subsequently, the solvent (3 mL) was added via a syringe and the mixture was preheated at 100 °C for 30 min. Another oven-dried vial was charged with Cs2CO3 and substrate 3. Also, this vessel was evacuated three times and backfilled with argon. The solution of the activated catalyst was transferred from the first vial into the second one via a syringe. The vessel was then heated at 100 °C until the starting component was fully consumed (control by TLC). The mixture was then diluted with EtOAc and filtered through a small plug of Celite® S which was subsequently thoroughly washed with EtOAc. The filtrate was concentrated in vacuo and the residue was purified by recrystallization or column chromatography (see details at individual compounds).
1-Phenyl-2-((2Z)-1,2,3,4-tetrahydroquinolin-2-ylidene)ethan-1-one (1a): Method A: from 3a, 3.5 mol % Pd2(dba)3, 7 mol % XPhos, toluene, 2 h. Column chromatography (silica gel; DCM/EtOAc 10:1). Yield 92%. From 3e, 5 mol % Pd2(dba)3, 10 mol % t-BuXPhos, t-AmOH, 17 h. Column chromatography (silica gel; DCM). Yield 72%. Yellow solid, mp 103–105 °C (ref. [31] reports 105–106 °C). Proton NMR data are in accordance with [32]. 1H NMR (400.13 MHz) δ 12.85 (brs, 1H), 7.94–7.91 (m, 2H), 7.50–7.42 (m, 3H), 7.21–7.17 (m, 1H), 7.12–7.10 (m, 1H), 6.99–6.94 (m, 2H), 5.88 (s, 1H), 2.89–2.86 (m, 2H), 2.75–2.71 (m, 2H) ppm; 13C NMR (100.62 MHz) δ 189.7, 159.0, 139.9, 136.7, 131.3, 128.5, 128.4, 127.9, 127.3, 125.3, 123.3, 116.8, 92.6, 28.8, 24.4 ppm.
Supporting Information
| Supporting Information File 1: Experimental procedures, characterization data and copies of NMR spectra. | ||
| Format: PDF | Size: 7.9 MB | Download |
References
-
Jacquemond-Collet, I.; Hannedouche, S.; Fabre, N.; Fourasté, I.; Moulis, C. Phytochemistry 1999, 51, 1167–1169. doi:10.1016/S0031-9422(99)00032-1
Return to citation in text: [1] [2] -
Jacquemond-Collet, I.; Benoit-Vical, S.; Mustofa; Valentin, A.; Stanislas, E.; Mallié, M.; Fourasté, I. Planta Med. 2002, 68, 68–69. doi:10.1055/s-2002-19869
Return to citation in text: [1] [2] -
Houghton, P. J.; Woldemariam, T. Z.; Watanabe, Y.; Yates, M. Planta Med. 1999, 65, 250–254. doi:10.1055/s-1999-13988
Return to citation in text: [1] -
Rakotoson, J. H.; Fabre, N.; Jacquemond-Collet, I.; Hannedouche, S.; Fourasté, I.; Moulis, C. Planta Med. 1998, 64, 762–763. doi:10.1055/s-2006-957578
Return to citation in text: [1] -
Avemaria, F.; Vanderheiden, S.; Bräse, S. Tetrahedron 2003, 59, 6785–6796. doi:10.1016/S0040-4020(03)00915-3
Return to citation in text: [1] -
Bentley, S. A.; Davies, S. G.; Lee, J. A.; Roberts, P. M.; Thomson, J. E. Org. Lett. 2011, 13, 2544–2547. doi:10.1021/ol200625h
Return to citation in text: [1] -
Cruz-Lopez, O.; Nunez, M. C.; Conejo-Garcia, A.; Kimatrai, M.; Campos, J. M. Curr. Org. Chem. 2011, 15, 869–887. doi:10.2174/138527211794518925
Return to citation in text: [1] -
Diaz, G.; Diaz, M. A. N.; Reis, M. A. J. Braz. Chem. Soc. 2013, 24, 1497–1503. doi:10.5935/0103-5053.20130189
Return to citation in text: [1] -
Chen, B.-L.; Wang, B.; Lin, G.-Q. J. Org. Chem. 2009, 75, 941–944. doi:10.1021/jo902424m
Return to citation in text: [1] -
Kothandaraman, P.; Foo, S. J.; Chan, P. W. H. J. Org. Chem. 2010, 74, 5947–5952. doi:10.1021/jo900917q
Return to citation in text: [1] -
Lin, X.-F.; Li, Y.; Ma, D.-W. Chin. J. Chem. 2004, 22, 932–934. doi:10.1002/cjoc.20040220911
Return to citation in text: [1] -
Lu, S.-M.; Wang, Y.-Q.; Han, X.-W.; Zhou, Y.-G. Angew. Chem., Int. Ed. 2006, 45, 2260–2263. doi:10.1002/anie.200503073
Return to citation in text: [1] -
Melzig, L.; Dennenwaldt, T.; Gavryushin, A.; Knochel, P. J. Org. Chem. 2011, 76, 8891–8906. doi:10.1021/jo201630e
Return to citation in text: [1] -
O'Byrne, A.; Evans, P. Tetrahedron 2008, 64, 8067–8072. doi:10.1016/j.tet.2008.06.073
Return to citation in text: [1] -
Patil, N. T.; Wu, H.; Yamamoto, Y. J. Org. Chem. 2007, 72, 6577–6579. doi:10.1021/jo0708137
Return to citation in text: [1] -
Rueping, M.; Antonchick, A. P.; Theissmann, T. Angew. Chem., Int. Ed. 2006, 45, 3683–3686. doi:10.1002/anie.200600191
Return to citation in text: [1] -
Ryu, J.-S. Bull. Korean Chem. Soc. 2006, 27, 631–632. doi:10.5012/bkcs.2006.27.5.631
Return to citation in text: [1] -
Satyanarayana, G.; Pflästerer, D.; Helmchen, G. Eur. J. Org. Chem. 2011, 6877–6886. doi:10.1002/ejoc.201100981
Return to citation in text: [1] -
Shahane, S.; Louafi, F.; Moreau, J.; Hurvois, J.-P.; Renaud, J.-L.; van de Weghe, P.; Roisnel, T. Eur. J. Org. Chem. 2008, 4622–4631. doi:10.1002/ejoc.200800512
Return to citation in text: [1] -
Tang, W.-J.; Tan, J.; Xu, L.-J.; Lam, K.-H.; Fan, Q.-H.; Chan, A. S. C. Adv. Synth. Catal. 2010, 352, 1055–1062. doi:10.1002/adsc.200900870
Return to citation in text: [1] -
Taylor, L. L.; Goldberg, F. W.; Hii, K. K. Org. Biomol. Chem. 2012, 10, 4424–4432. doi:10.1039/c2ob25122a
Return to citation in text: [1] -
Theeraladanon, C.; Arisawa, M.; Nakagawa, M.; Nishida, A. Tetrahedron: Asymmetry 2005, 16, 827–831. doi:10.1016/j.tetasy.2004.12.022
Return to citation in text: [1] -
Tummatorn, J.; Muñoz, G. D.; Dudley, G. B. Tetrahedron Lett. 2013, 54, 1312–1314. doi:10.1016/j.tetlet.2012.12.122
Return to citation in text: [1] -
Wang, D.-W.; Zeng, W.; Zhou, Y.-G. Tetrahedron: Asymmetry 2007, 18, 1103–1107. doi:10.1016/j.tetasy.2007.04.028
Return to citation in text: [1] -
Wang, T.; Zhuo, L.-G.; Li, Z.; Chen, F.; Ding, Z.; He, Y.; Fan, Q.-H.; Xiang, J.; Yu, Z.-X.; Chan, A. S. C. J. Am. Chem. Soc. 2011, 133, 9878–9891. doi:10.1021/ja2023042
Return to citation in text: [1] -
Wang, W.-B.; Lu, S.-M.; Yang, P.-Y.; Han, X.-W.; Zhou, Y.-G. J. Am. Chem. Soc. 2003, 125, 10536–10537. doi:10.1021/ja0353762
Return to citation in text: [1] -
Wang, Z.-J.; Zhou, H.-F.; Wang, T.-L.; He, Y.-M.; Fan, Q.-H. Green Chem. 2009, 11, 767–769. doi:10.1039/b822822a
Return to citation in text: [1] -
Ye, K.-Y.; He, H.; Liu, W.-B.; Dai, L.-X.; Helmchen, G.; You, S.-L. J. Am. Chem. Soc. 2011, 133, 19006–19014. doi:10.1021/ja2092954
Return to citation in text: [1] -
Fustero, S.; Moscardó, J.; Jiménez, D.; Pérez-Carrión, M. D.; Sánchez-Roselló, M.; del Pozo, C. Chem. – Eur. J. 2008, 14, 9868–9872. doi:10.1002/chem.200801480
Return to citation in text: [1] -
Wang, X.-B.; Wang, D.-W.; Lu, S.-M.; Yu, C.-B.; Zhou, Y.-G. Tetrahedron: Asymmetry 2009, 20, 1040–1045. doi:10.1016/j.tetasy.2009.03.037
Return to citation in text: [1] [2] [3] [4] -
Greenhill, J. V.; Loghmani-Kkhouzani, H.; Maitland, D. J. Tetrahedron 1988, 44, 3319–3326. doi:10.1016/s0040-4020(01)85965-2
Return to citation in text: [1] [2] -
Wang, D.-W.; Wang, X.-B.; Wang, D.-S.; Lu, S.-M.; Zhou, Y.-G.; Li, Y.-X. J. Org. Chem. 2009, 74, 2780–2787. doi:10.1021/jo900073z
Return to citation in text: [1] [2] -
Krapcho, A. P. ARKIVOC 2007, No. ii, 1–53.
Return to citation in text: [1] -
Rosiak, A.; Frey, W.; Christoffers, J. Eur. J. Org. Chem. 2006, 4044–4054. doi:10.1002/ejoc.200600372
Return to citation in text: [1] -
Bernini, R.; Cacchi, S.; Fabrizi, G.; Sferrazza, A. Synthesis 2009, 1209–1219. doi:10.1055/s-0028-1087990
Return to citation in text: [1] -
Edmondson, S. D.; Mastracchio, A.; Parmee, E. R. Org. Lett. 2000, 2, 1109–1112. doi:10.1021/ol000031z
Return to citation in text: [1] -
Liu, Y.; Yu, C.-Y.; Wang, M.-X. ARKIVOC 2003, No. ii, 146–154.
Return to citation in text: [1] -
Melkonyan, F.; Topolyan, A.; Yurovskaya, M.; Karchava, A. Eur. J. Org. Chem. 2008, 5952–5956. doi:10.1002/ejoc.200800777
Return to citation in text: [1] -
Melkonyan, F. S.; Karchava, A. V.; Yurovskaya, M. A. J. Org. Chem. 2008, 73, 4275–4278. doi:10.1021/jo800630v
Return to citation in text: [1] -
Melkonyan, F. S.; Topolyan, A. P.; Karchava, A. V.; Yurovskaya, M. A. Chem. Heterocycl. Compd. 2008, 44, 1288–1290. doi:10.1007/s10593-009-0183-0
Return to citation in text: [1] -
Melkonyan, F. S.; Topolyan, A. P.; Karchava, A. V.; Yurovskaya, M. A. Chem. Heterocycl. Compd. 2010, 46, 1158–1160. doi:10.1007/s10593-010-0648-1
Return to citation in text: [1] -
Mori, M.; Uozumi, Y.; Shibasaki, M. Heterocycles 1992, 33, 819–830. doi:10.3987/COM-91-S88
Return to citation in text: [1] -
Uozumi, Y.; Mori, M.; Shibasaki, M. J. Chem. Soc., Chem. Commun. 1991, 81–83. doi:10.1039/c39910000081
Return to citation in text: [1] -
Vaswani, R. G.; Albrecht, B. K.; Audia, J. E.; Côté, A.; Dakin, L. A.; Duplessis, M.; Gehling, V. S.; Harmange, J. C.; Hewitt, M. C.; Leblanc, Y.; Nasveschuk, C. G.; Taylor, A. M. Org. Lett. 2014, 16, 4114–4117. doi:10.1021/ol5018118
Return to citation in text: [1] -
Wu, X.-J.; Jiang, R.; Wu, B.; Su, X.-M.; Xu, X.-P.; Ji, S.-J. Adv. Synth. Catal. 2009, 351, 3150–3156. doi:10.1002/adsc.200900481
Return to citation in text: [1] -
Wu, X.-J.; Xu, X.-P.; Su, X.-M.; Chen, G.; Zhang, Y.; Ji, S.-J. Eur. J. Org. Chem. 2009, 4963–4970. doi:10.1002/ejoc.200900451
Return to citation in text: [1] -
Fors, B. P.; Krattiger, P.; Strieter, E.; Buchwald, S. L. Org. Lett. 2008, 10, 3505–3508. doi:10.1021/ol801285g
Return to citation in text: [1] [2] -
Surry, D. S.; Buchwald, S. L. Chem. Sci. 2011, 2, 27–50. doi:10.1039/c0sc00331j
Return to citation in text: [1] [2] [3] -
Surry, D. S.; Buchwald, S. L. Angew. Chem., Int. Ed. 2008, 47, 6338–6361. doi:10.1002/anie.200800497
Return to citation in text: [1] -
Beletskaya, I. P.; Cheprakov, A. V. Organometallics 2012, 31, 7753–7808. doi:10.1021/om300683c
Return to citation in text: [1] [2] -
Ley, S. V.; Thomas, A. W. Angew. Chem., Int. Ed. 2003, 42, 5400–5449. doi:10.1002/anie.200300594
Return to citation in text: [1] -
Surry, D. S.; Buchwald, S. L. Chem. Sci. 2010, 1, 13–31. doi:10.1039/c0sc00107d
Return to citation in text: [1] -
Kinzel, T.; Zhang, Y.; Buchwald, S. L. J. Am. Chem. Soc. 2010, 132, 14073–14075. doi:10.1021/ja1073799
Return to citation in text: [1] -
Maiti, D.; Fors, B. P.; Henderson, J. L.; Nakamura, Y.; Buchwald, S. L. Chem. Sci. 2011, 2, 57–68. doi:10.1039/c0sc00330a
Return to citation in text: [1] -
Allen, F. H.; Kennard, O.; Watson, D. G.; Brammer, L.; Orpen, A. G.; Taylor, R. J. Chem. Soc., Perkin Trans. 2 1987, S1–S19. doi:10.1039/p298700000s1
Return to citation in text: [1] [2] -
Kolehmainen, E.; Ośmiałowski, B.; Krygowski, T. M.; Kauppinen, R.; Nissinen, M.; Gawinecki, R. J. Chem. Soc., Perkin Trans. 2 2000, 1259–1266. doi:10.1039/A908874A
Return to citation in text: [1] -
Mehrabi, H.; Loghmani-Khouzani, H.; Sadeghi, M. M.; Charmant, J. P. H.; Gunter, C. M. M.; Ghorbani, M. M. Anal. Sci.: X-Ray Struct. Anal. Online 2007, 23, x161–x162. doi:10.2116/analscix.23.x161
Return to citation in text: [1]
| 1. | Jacquemond-Collet, I.; Hannedouche, S.; Fabre, N.; Fourasté, I.; Moulis, C. Phytochemistry 1999, 51, 1167–1169. doi:10.1016/S0031-9422(99)00032-1 |
| 1. | Jacquemond-Collet, I.; Hannedouche, S.; Fabre, N.; Fourasté, I.; Moulis, C. Phytochemistry 1999, 51, 1167–1169. doi:10.1016/S0031-9422(99)00032-1 |
| 4. | Rakotoson, J. H.; Fabre, N.; Jacquemond-Collet, I.; Hannedouche, S.; Fourasté, I.; Moulis, C. Planta Med. 1998, 64, 762–763. doi:10.1055/s-2006-957578 |
| 48. | Surry, D. S.; Buchwald, S. L. Chem. Sci. 2011, 2, 27–50. doi:10.1039/c0sc00331j |
| 49. | Surry, D. S.; Buchwald, S. L. Angew. Chem., Int. Ed. 2008, 47, 6338–6361. doi:10.1002/anie.200800497 |
| 2. | Jacquemond-Collet, I.; Benoit-Vical, S.; Mustofa; Valentin, A.; Stanislas, E.; Mallié, M.; Fourasté, I. Planta Med. 2002, 68, 68–69. doi:10.1055/s-2002-19869 |
| 47. | Fors, B. P.; Krattiger, P.; Strieter, E.; Buchwald, S. L. Org. Lett. 2008, 10, 3505–3508. doi:10.1021/ol801285g |
| 3. | Houghton, P. J.; Woldemariam, T. Z.; Watanabe, Y.; Yates, M. Planta Med. 1999, 65, 250–254. doi:10.1055/s-1999-13988 |
| 47. | Fors, B. P.; Krattiger, P.; Strieter, E.; Buchwald, S. L. Org. Lett. 2008, 10, 3505–3508. doi:10.1021/ol801285g |
| 2. | Jacquemond-Collet, I.; Benoit-Vical, S.; Mustofa; Valentin, A.; Stanislas, E.; Mallié, M.; Fourasté, I. Planta Med. 2002, 68, 68–69. doi:10.1055/s-2002-19869 |
| 48. | Surry, D. S.; Buchwald, S. L. Chem. Sci. 2011, 2, 27–50. doi:10.1039/c0sc00331j |
| 30. | Wang, X.-B.; Wang, D.-W.; Lu, S.-M.; Yu, C.-B.; Zhou, Y.-G. Tetrahedron: Asymmetry 2009, 20, 1040–1045. doi:10.1016/j.tetasy.2009.03.037 |
| 31. | Greenhill, J. V.; Loghmani-Kkhouzani, H.; Maitland, D. J. Tetrahedron 1988, 44, 3319–3326. doi:10.1016/s0040-4020(01)85965-2 |
| 32. | Wang, D.-W.; Wang, X.-B.; Wang, D.-S.; Lu, S.-M.; Zhou, Y.-G.; Li, Y.-X. J. Org. Chem. 2009, 74, 2780–2787. doi:10.1021/jo900073z |
| 34. | Rosiak, A.; Frey, W.; Christoffers, J. Eur. J. Org. Chem. 2006, 4044–4054. doi:10.1002/ejoc.200600372 |
| 30. | Wang, X.-B.; Wang, D.-W.; Lu, S.-M.; Yu, C.-B.; Zhou, Y.-G. Tetrahedron: Asymmetry 2009, 20, 1040–1045. doi:10.1016/j.tetasy.2009.03.037 |
| 35. | Bernini, R.; Cacchi, S.; Fabrizi, G.; Sferrazza, A. Synthesis 2009, 1209–1219. doi:10.1055/s-0028-1087990 |
| 36. | Edmondson, S. D.; Mastracchio, A.; Parmee, E. R. Org. Lett. 2000, 2, 1109–1112. doi:10.1021/ol000031z |
| 37. | Liu, Y.; Yu, C.-Y.; Wang, M.-X. ARKIVOC 2003, No. ii, 146–154. |
| 38. | Melkonyan, F.; Topolyan, A.; Yurovskaya, M.; Karchava, A. Eur. J. Org. Chem. 2008, 5952–5956. doi:10.1002/ejoc.200800777 |
| 39. | Melkonyan, F. S.; Karchava, A. V.; Yurovskaya, M. A. J. Org. Chem. 2008, 73, 4275–4278. doi:10.1021/jo800630v |
| 40. | Melkonyan, F. S.; Topolyan, A. P.; Karchava, A. V.; Yurovskaya, M. A. Chem. Heterocycl. Compd. 2008, 44, 1288–1290. doi:10.1007/s10593-009-0183-0 |
| 41. | Melkonyan, F. S.; Topolyan, A. P.; Karchava, A. V.; Yurovskaya, M. A. Chem. Heterocycl. Compd. 2010, 46, 1158–1160. doi:10.1007/s10593-010-0648-1 |
| 42. | Mori, M.; Uozumi, Y.; Shibasaki, M. Heterocycles 1992, 33, 819–830. doi:10.3987/COM-91-S88 |
| 43. | Uozumi, Y.; Mori, M.; Shibasaki, M. J. Chem. Soc., Chem. Commun. 1991, 81–83. doi:10.1039/c39910000081 |
| 44. | Vaswani, R. G.; Albrecht, B. K.; Audia, J. E.; Côté, A.; Dakin, L. A.; Duplessis, M.; Gehling, V. S.; Harmange, J. C.; Hewitt, M. C.; Leblanc, Y.; Nasveschuk, C. G.; Taylor, A. M. Org. Lett. 2014, 16, 4114–4117. doi:10.1021/ol5018118 |
| 45. | Wu, X.-J.; Jiang, R.; Wu, B.; Su, X.-M.; Xu, X.-P.; Ji, S.-J. Adv. Synth. Catal. 2009, 351, 3150–3156. doi:10.1002/adsc.200900481 |
| 46. | Wu, X.-J.; Xu, X.-P.; Su, X.-M.; Chen, G.; Zhang, Y.; Ji, S.-J. Eur. J. Org. Chem. 2009, 4963–4970. doi:10.1002/ejoc.200900451 |
| 30. | Wang, X.-B.; Wang, D.-W.; Lu, S.-M.; Yu, C.-B.; Zhou, Y.-G. Tetrahedron: Asymmetry 2009, 20, 1040–1045. doi:10.1016/j.tetasy.2009.03.037 |
| 5. | Avemaria, F.; Vanderheiden, S.; Bräse, S. Tetrahedron 2003, 59, 6785–6796. doi:10.1016/S0040-4020(03)00915-3 |
| 6. | Bentley, S. A.; Davies, S. G.; Lee, J. A.; Roberts, P. M.; Thomson, J. E. Org. Lett. 2011, 13, 2544–2547. doi:10.1021/ol200625h |
| 7. | Cruz-Lopez, O.; Nunez, M. C.; Conejo-Garcia, A.; Kimatrai, M.; Campos, J. M. Curr. Org. Chem. 2011, 15, 869–887. doi:10.2174/138527211794518925 |
| 8. | Diaz, G.; Diaz, M. A. N.; Reis, M. A. J. Braz. Chem. Soc. 2013, 24, 1497–1503. doi:10.5935/0103-5053.20130189 |
| 9. | Chen, B.-L.; Wang, B.; Lin, G.-Q. J. Org. Chem. 2009, 75, 941–944. doi:10.1021/jo902424m |
| 10. | Kothandaraman, P.; Foo, S. J.; Chan, P. W. H. J. Org. Chem. 2010, 74, 5947–5952. doi:10.1021/jo900917q |
| 11. | Lin, X.-F.; Li, Y.; Ma, D.-W. Chin. J. Chem. 2004, 22, 932–934. doi:10.1002/cjoc.20040220911 |
| 12. | Lu, S.-M.; Wang, Y.-Q.; Han, X.-W.; Zhou, Y.-G. Angew. Chem., Int. Ed. 2006, 45, 2260–2263. doi:10.1002/anie.200503073 |
| 13. | Melzig, L.; Dennenwaldt, T.; Gavryushin, A.; Knochel, P. J. Org. Chem. 2011, 76, 8891–8906. doi:10.1021/jo201630e |
| 14. | O'Byrne, A.; Evans, P. Tetrahedron 2008, 64, 8067–8072. doi:10.1016/j.tet.2008.06.073 |
| 15. | Patil, N. T.; Wu, H.; Yamamoto, Y. J. Org. Chem. 2007, 72, 6577–6579. doi:10.1021/jo0708137 |
| 16. | Rueping, M.; Antonchick, A. P.; Theissmann, T. Angew. Chem., Int. Ed. 2006, 45, 3683–3686. doi:10.1002/anie.200600191 |
| 17. | Ryu, J.-S. Bull. Korean Chem. Soc. 2006, 27, 631–632. doi:10.5012/bkcs.2006.27.5.631 |
| 18. | Satyanarayana, G.; Pflästerer, D.; Helmchen, G. Eur. J. Org. Chem. 2011, 6877–6886. doi:10.1002/ejoc.201100981 |
| 19. | Shahane, S.; Louafi, F.; Moreau, J.; Hurvois, J.-P.; Renaud, J.-L.; van de Weghe, P.; Roisnel, T. Eur. J. Org. Chem. 2008, 4622–4631. doi:10.1002/ejoc.200800512 |
| 20. | Tang, W.-J.; Tan, J.; Xu, L.-J.; Lam, K.-H.; Fan, Q.-H.; Chan, A. S. C. Adv. Synth. Catal. 2010, 352, 1055–1062. doi:10.1002/adsc.200900870 |
| 21. | Taylor, L. L.; Goldberg, F. W.; Hii, K. K. Org. Biomol. Chem. 2012, 10, 4424–4432. doi:10.1039/c2ob25122a |
| 22. | Theeraladanon, C.; Arisawa, M.; Nakagawa, M.; Nishida, A. Tetrahedron: Asymmetry 2005, 16, 827–831. doi:10.1016/j.tetasy.2004.12.022 |
| 23. | Tummatorn, J.; Muñoz, G. D.; Dudley, G. B. Tetrahedron Lett. 2013, 54, 1312–1314. doi:10.1016/j.tetlet.2012.12.122 |
| 24. | Wang, D.-W.; Zeng, W.; Zhou, Y.-G. Tetrahedron: Asymmetry 2007, 18, 1103–1107. doi:10.1016/j.tetasy.2007.04.028 |
| 25. | Wang, T.; Zhuo, L.-G.; Li, Z.; Chen, F.; Ding, Z.; He, Y.; Fan, Q.-H.; Xiang, J.; Yu, Z.-X.; Chan, A. S. C. J. Am. Chem. Soc. 2011, 133, 9878–9891. doi:10.1021/ja2023042 |
| 26. | Wang, W.-B.; Lu, S.-M.; Yang, P.-Y.; Han, X.-W.; Zhou, Y.-G. J. Am. Chem. Soc. 2003, 125, 10536–10537. doi:10.1021/ja0353762 |
| 27. | Wang, Z.-J.; Zhou, H.-F.; Wang, T.-L.; He, Y.-M.; Fan, Q.-H. Green Chem. 2009, 11, 767–769. doi:10.1039/b822822a |
| 28. | Ye, K.-Y.; He, H.; Liu, W.-B.; Dai, L.-X.; Helmchen, G.; You, S.-L. J. Am. Chem. Soc. 2011, 133, 19006–19014. doi:10.1021/ja2092954 |
| 29. | Fustero, S.; Moscardó, J.; Jiménez, D.; Pérez-Carrión, M. D.; Sánchez-Roselló, M.; del Pozo, C. Chem. – Eur. J. 2008, 14, 9868–9872. doi:10.1002/chem.200801480 |
| 30. | Wang, X.-B.; Wang, D.-W.; Lu, S.-M.; Yu, C.-B.; Zhou, Y.-G. Tetrahedron: Asymmetry 2009, 20, 1040–1045. doi:10.1016/j.tetasy.2009.03.037 |
| 48. | Surry, D. S.; Buchwald, S. L. Chem. Sci. 2011, 2, 27–50. doi:10.1039/c0sc00331j |
| 54. | Maiti, D.; Fors, B. P.; Henderson, J. L.; Nakamura, Y.; Buchwald, S. L. Chem. Sci. 2011, 2, 57–68. doi:10.1039/c0sc00330a |
| 50. | Beletskaya, I. P.; Cheprakov, A. V. Organometallics 2012, 31, 7753–7808. doi:10.1021/om300683c |
| 51. | Ley, S. V.; Thomas, A. W. Angew. Chem., Int. Ed. 2003, 42, 5400–5449. doi:10.1002/anie.200300594 |
| 52. | Surry, D. S.; Buchwald, S. L. Chem. Sci. 2010, 1, 13–31. doi:10.1039/c0sc00107d |
| 53. | Kinzel, T.; Zhang, Y.; Buchwald, S. L. J. Am. Chem. Soc. 2010, 132, 14073–14075. doi:10.1021/ja1073799 |
| 32. | Wang, D.-W.; Wang, X.-B.; Wang, D.-S.; Lu, S.-M.; Zhou, Y.-G.; Li, Y.-X. J. Org. Chem. 2009, 74, 2780–2787. doi:10.1021/jo900073z |
| 55. | Allen, F. H.; Kennard, O.; Watson, D. G.; Brammer, L.; Orpen, A. G.; Taylor, R. J. Chem. Soc., Perkin Trans. 2 1987, S1–S19. doi:10.1039/p298700000s1 |
| 31. | Greenhill, J. V.; Loghmani-Kkhouzani, H.; Maitland, D. J. Tetrahedron 1988, 44, 3319–3326. doi:10.1016/s0040-4020(01)85965-2 |
| 56. | Kolehmainen, E.; Ośmiałowski, B.; Krygowski, T. M.; Kauppinen, R.; Nissinen, M.; Gawinecki, R. J. Chem. Soc., Perkin Trans. 2 2000, 1259–1266. doi:10.1039/A908874A |
| 57. | Mehrabi, H.; Loghmani-Khouzani, H.; Sadeghi, M. M.; Charmant, J. P. H.; Gunter, C. M. M.; Ghorbani, M. M. Anal. Sci.: X-Ray Struct. Anal. Online 2007, 23, x161–x162. doi:10.2116/analscix.23.x161 |
| 50. | Beletskaya, I. P.; Cheprakov, A. V. Organometallics 2012, 31, 7753–7808. doi:10.1021/om300683c |
| 55. | Allen, F. H.; Kennard, O.; Watson, D. G.; Brammer, L.; Orpen, A. G.; Taylor, R. J. Chem. Soc., Perkin Trans. 2 1987, S1–S19. doi:10.1039/p298700000s1 |
© 2015 Doušová et al; licensee Beilstein-Institut.
This is an Open Access article under the terms of the Creative Commons Attribution License (http://creativecommons.org/licenses/by/2.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
The license is subject to the Beilstein Journal of Organic Chemistry terms and conditions: (http://www.beilstein-journals.org/bjoc)