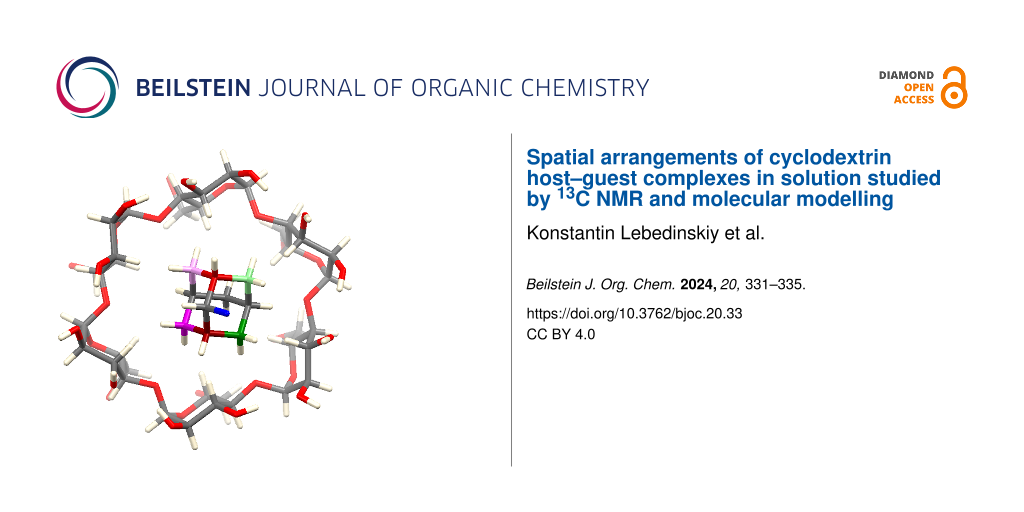
Abstract
13C NMR spectroscopic analyses of Cs symmetric guest molecules in the cyclodextrin host cavity, combined with molecular modelling and solid-state X-ray analysis, provides a detailed description of the spatial arrangement of cyclodextrin host–guest complexes in solution. The chiral cavity of the cyclodextrin molecule creates an anisotropic environment for the guest molecule resulting in a splitting of its prochiral carbon signals in 13C NMR spectra. This signal split can be correlated to the distance of the guest atoms from the wall of the host cavity and to the spatial separation of binding sites preferred by pairs of prochiral carbon atoms. These measurements complement traditional solid-state analyses, which rely on the crystallization of host–guest complexes and their crystallographic analysis.
Graphical Abstract
Introduction
Complexation of organic and inorganic compounds with α-, β-, or γ-cyclodextrins and their derivatives [1] is an established tool used in medicine for drug delivery [2-4], in analytical and preparative chemistry for compound separation [5] and in materials science for small molecule detection [6,7]. Association (binding) constants between the host and guest molecules [8-10] are typically measured by 1H NMR titration [11,12] or isothermal titration calorimetry [13]. Single crystals for many host–guest complexes have been prepared, and their structure elucidated by X-ray crystallography [14,15]. Conformations of host–guest complexes in solution have been studied by 2D NMR experiments [11] (NOESY, ROESY) or proposed computationally [16,17] based on dispersion forces and hydrogen bonding between the cyclodextrin (CD) unit and the guest molecule. Determination of the ee of chiral guests was achieved by observing the splitting of 1H NMR signals of the achiral host upon formation of diastereomeric inclusion complexes [18,19]. Shifting the H-3 and H-5 proton signals of CDs in 1H NMR or 2D NMR ROESY or NOESY experiments can indicate the spatial distances between host and guest atoms and distinguish between enantiomeric guests. However, the evaluation of such interactions is often hampered by overlapping signals [20].
Results and Discussion
In this work, we reveal the conformation (spatial arrangement) of the host–guest complex in solution spectroscopically by measuring the 13C NMR spectra of a suitable guest molecule. We decided to take Cs symmetric guest molecules, CD as a host and measure the 13C NMR spectra of these complexes. We expect that the anisotropy of the chiral cavity is expressed by differences in the magnetic shielding of prochiral atoms, resulting in signal splitting of the prochiral carbons of the guest molecule in 13C NMR spectra. Adamantan-2-amine hydrochloride was used as a model guest molecule containing three sets of prochiral carbons (Figure 1).
![[1860-5397-20-33-1]](/bjoc/content/figures/1860-5397-20-33-1.png?scale=2.0&max-width=1024&background=FFFFFF)
Figure 1: Model system for the elucidation of the solution state conformations of the host–guest complexes. 13C NMR splits of prochiral carbons of the guest molecule 1 (adamantan-2-amine hydrochloride) in the anisotropic cavity of the host α-CD in D2O solution. Data were acquired at 13C Larmor frequency of 150 MHz.
Figure 1: Model system for the elucidation of the solution state conformations of the host–guest complexes. 13...
The degree of the signal split correlates with the distance of prochiral atoms (carbons) to the host cavity and thus gives information about the conformation of the guest molecule in the host cavity. First, we measured simple 1D 13C NMR spectra and indeed observed the split of the peaks of all prochiral carbon signals of the guest upon complexation with α-CD. The biggest split, 22.5 Hz, was observed for prochiral carbons 8 and 10, depicted in Figure 1 in green color, followed by a split of carbons 3 and 9 (in blue color) and the smallest difference in the magnetic field shielding was detected for carbons 1 and 3 (in red color).
The same 1D 13C NMR spectra were measured for guest molecule 1 in host cavities of β-CD and γ-CD, showing almost no split of prochiral carbon peaks and suggesting a higher degree of conformational flexibility of the host–guest complexes than for the complex of 1 with α-CD. Interatomic distances within the host–guest complex were measured using rotating-frame nuclear Overhauser effect spectroscopy (ROESY) measurements. For the complex of 1 with α-CD, the cross-peak in the 2D ROESY spectrum between proton 5 of the glucose moiety of the host α-CD and the protons 6 of molecule 1 reveals deep penetration of the guest into the cavity of α-cyclodextrin (Figure 2). All protons of 1 show ROESY cross-peaks with proton 3 of the glucose unit (see page S73 in Supporting Information File 1).
![[1860-5397-20-33-2]](/bjoc/content/figures/1860-5397-20-33-2.png?scale=2.0&max-width=1024&background=FFFFFF)
Figure 2: 2D ROESY NMR measurements to estimate the depth of the guest molecule 1 (adamantan-2-amine hydrochloride) in the α-CD cavity.
Figure 2: 2D ROESY NMR measurements to estimate the depth of the guest molecule 1 (adamantan-2-amine hydrochl...
For complexes of ligands with α-CD, we always performed a series of ten classical molecular dynamics (MD) simulations [16] (each lasting 100 ns, Figure 3). Then, we superimposed α-CD structures from different snapshots of each MD run. Further, the 3D densities, showing the spatial distribution of prochiral atoms of ligands (that rotate and wobble towards α-CD), were calculated. If two atoms have the same spatial density, then the splitting of their signals in 13C NMR spectra cannot occur. On the contrary, if these densities, which belong to a pair of prochiral atoms, are well separated, then NMR splitting can be expected due to the anisotropic environment of the chiral α-CD cavity.
![[1860-5397-20-33-3]](/bjoc/content/figures/1860-5397-20-33-3.png?scale=2.0&max-width=1024&background=FFFFFF)
Figure 3: Molecular model of the host (α-CD–guest 1 (adamantan-2-amine hydrochloride)) complex. The 3D densities show the spatial distribution of prochiral atoms within MD simulations.
Figure 3: Molecular model of the host (α-CD–guest 1 (adamantan-2-amine hydrochloride)) complex. The 3D densit...
The NMR splitting is generally larger if the prochiral guest atoms are located closer to the cavity of the α-CD. In other words, the splitting is larger, if the radius of a density is bigger or the density runs deeper into the α-CD cavity. In Figure 3, the green and blue densities with well-separated light and dark clouds (belonging to different prochiral carbons from a pair) have larger radii and run deeper into the α-CD cavity than the red density with mixed light and dark clouds. Accordingly, our NMR experiment only showed splitting for the green and blue atoms (see Figure 1).
Having established the method, we chose various cyclic compounds (noradamantane, adamantane, cyclohexane derivatives) with desired symmetry and measured NMR spectra of these guest molecules (2–8) in α-cyclodextrin and cyclodextrins with a larger cavity (β-CD and γ-CD, Figure 4).
Figure 4: Studied host–guest complexes and splitting of guests’ prochiral carbons in their 13C NMR spectra.
Figure 4: Studied host–guest complexes and splitting of guests’ prochiral carbons in their 13C NMR spectra.
In all cases, the 1H NMR spectra indicated the interaction of the guest molecule with the host. In cases where the split of prochiral carbons was observed, 13C NMR spectra showed which pair of prochiral carbons in the guest molecule is close to the wall of the host’s chiral cavity, creating an anisotropic environment. For compound 4, we were thus able to select representative conformations of the guest molecule in all types of cyclodextrins (Figure 5) using the spatial densities gained from classical MD simulations.
![[1860-5397-20-33-5]](/bjoc/content/figures/1860-5397-20-33-5.png?scale=2.0&max-width=1024&background=FFFFFF)
Figure 5: Molecular modelling of the host–guest complexes of compound 4 with α-CD, β-CD and γ-CD.
Figure 5: Molecular modelling of the host–guest complexes of compound 4 with α-CD, β-CD and γ-CD.
Compounds 1–8 (as hydrochloride salts or free bases), were attempted to co-crystallize with α-, β- and γ-cyclodextrins. Compounds 1, 6 and 8 are commercial, compounds 2–5 [21] and 7 [22] were prepared according to published procedures. Compound 2 (as a free amine) crystallized in the supramolecular capsule of two α-cyclodextrins, and the mono-crystal was subjected to X-ray analysis. This experiment was performed to compare the solid-state structure of the guest 2–α-CD host complex with its proposed solution state conformations (Figure 6).
![[1860-5397-20-33-6]](/bjoc/content/figures/1860-5397-20-33-6.png?scale=2.0&max-width=1024&background=FFFFFF)
Figure 6: X-ray analysis of the α-CD supramolecular capsule with noradamantane-3-methyleneamine (2) as guest molecule (side and top view).
Figure 6: X-ray analysis of the α-CD supramolecular capsule with noradamantane-3-methyleneamine (2) as guest ...
Conclusion
We have demonstrated that simple 13C NMR analyses of properly chosen Cs symmetric compounds varying in size can be used to estimate the host–guest spatial arrangement in solution and extrapolated for compounds with similar size and bonding nature.
Supporting Information
| Supporting Information File 1: General information, NMR spectra, NMR study, computational study, crystallographic data collection and refinement details. | ||
| Format: PDF | Size: 6.8 MB | Download |
Data Availability Statement
All data that supports the findings of this study is available in the published article and/or the supporting information to this article.
References
-
Crini, G. Chem. Rev. 2014, 114, 10940–10975. doi:10.1021/cr500081p
Return to citation in text: [1] -
Challa, R.; Ahuja, A.; Ali, J.; Khar, R. K. AAPS PharmSciTech 2005, 6, E329–E357. doi:10.1208/pt060243
Return to citation in text: [1] -
Zhang, J.; Ma, P. X. Adv. Drug Delivery Rev. 2013, 65, 1215–1233. doi:10.1016/j.addr.2013.05.001
Return to citation in text: [1] -
Wankar, J.; Kotla, N. G.; Gera, S.; Rasala, S.; Pandit, A.; Rochev, Y. A. Adv. Funct. Mater. 2020, 30, 1909049. doi:10.1002/adfm.201909049
Return to citation in text: [1] -
Szente, L.; Szemán, J. Anal. Chem. (Washington, DC, U. S.) 2013, 85, 8024–8030. doi:10.1021/ac400639y
Return to citation in text: [1] -
Ogoshi, T.; Harada, A. Sensors 2008, 8, 4961–4982. doi:10.3390/s8084961
Return to citation in text: [1] -
Mako, T. L.; Racicot, J. M.; Levine, M. Chem. Rev. 2019, 119, 322–477. doi:10.1021/acs.chemrev.8b00260
Return to citation in text: [1] -
Rekharsky, M. V.; Inoue, Y. Chem. Rev. 1998, 98, 1875–1918. doi:10.1021/cr970015o
Return to citation in text: [1] -
Mura, P. J. Pharm. Biomed. Anal. 2015, 113, 226–238. doi:10.1016/j.jpba.2015.01.058
Return to citation in text: [1] -
Mura, P. J. Pharm. Biomed. Anal. 2014, 101, 238–250. doi:10.1016/j.jpba.2014.02.022
Return to citation in text: [1] -
Schneider, H.-J.; Hacket, F.; Rüdiger, V.; Ikeda, H. Chem. Rev. 1998, 98, 1755–1786. doi:10.1021/cr970019t
Return to citation in text: [1] [2] -
Fielding, L. Tetrahedron 2000, 56, 6151–6170. doi:10.1016/s0040-4020(00)00492-0
Return to citation in text: [1] -
Bouchemal, K.; Mazzaferro, S. Drug Discovery Today 2012, 17, 623–629. doi:10.1016/j.drudis.2012.01.023
Return to citation in text: [1] -
Lindner, K.; Saenger, W. Acta Crystallogr., Sect. B: Struct. Crystallogr. Cryst. Chem. 1982, 38, 203–210. doi:10.1107/s0567740882002386
Return to citation in text: [1] -
Groom, C. R.; Bruno, I. J.; Lightfoot, M. P.; Ward, S. C. Acta Crystallogr., Sect. B: Struct. Sci., Cryst. Eng. Mater. 2016, 72, 171–179. doi:10.1107/s2052520616003954
Return to citation in text: [1] -
Gebhardt, J.; Kleist, C.; Jakobtorweihen, S.; Hansen, N. J. Phys. Chem. B 2018, 122, 1608–1626. doi:10.1021/acs.jpcb.7b11808
Return to citation in text: [1] [2] -
Schönbeck, C. J. Phys. Chem. B 2018, 122, 4821–4827. doi:10.1021/acs.jpcb.8b02579
Return to citation in text: [1] -
Shundo, A.; Labuta, J.; Hill, J. P.; Ishihara, S.; Ariga, K. J. Am. Chem. Soc. 2009, 131, 9494–9495. doi:10.1021/ja903371d
Return to citation in text: [1] -
Labuta, J.; Ishihara, S.; Šikorský, T.; Futera, Z.; Shundo, A.; Hanyková, L.; Burda, J. V.; Ariga, K.; Hill, J. P. Nat. Commun. 2013, 4, 2188. doi:10.1038/ncomms3188
Return to citation in text: [1] -
Dodziuk, H.; Koźmiński, W.; Ejchart, A. Chirality 2004, 16, 90–105. doi:10.1002/chir.10304
Return to citation in text: [1] -
Hrdina, R.; Holovko-Kamoshenkova, O. M.; Císařová, I.; Koucký, F.; Machalický, O. RSC Adv. 2022, 12, 31056–31060. doi:10.1039/d2ra06402b
Return to citation in text: [1] -
Torres, E.; Fernández, R.; Miquet, S.; Font-Bardia, M.; Vanderlinden, E.; Naesens, L.; Vázquez, S. ACS Med. Chem. Lett. 2012, 3, 1065–1069. doi:10.1021/ml300279b
Return to citation in text: [1]
| 8. | Rekharsky, M. V.; Inoue, Y. Chem. Rev. 1998, 98, 1875–1918. doi:10.1021/cr970015o |
| 9. | Mura, P. J. Pharm. Biomed. Anal. 2015, 113, 226–238. doi:10.1016/j.jpba.2015.01.058 |
| 10. | Mura, P. J. Pharm. Biomed. Anal. 2014, 101, 238–250. doi:10.1016/j.jpba.2014.02.022 |
| 22. | Torres, E.; Fernández, R.; Miquet, S.; Font-Bardia, M.; Vanderlinden, E.; Naesens, L.; Vázquez, S. ACS Med. Chem. Lett. 2012, 3, 1065–1069. doi:10.1021/ml300279b |
| 6. | Ogoshi, T.; Harada, A. Sensors 2008, 8, 4961–4982. doi:10.3390/s8084961 |
| 7. | Mako, T. L.; Racicot, J. M.; Levine, M. Chem. Rev. 2019, 119, 322–477. doi:10.1021/acs.chemrev.8b00260 |
| 5. | Szente, L.; Szemán, J. Anal. Chem. (Washington, DC, U. S.) 2013, 85, 8024–8030. doi:10.1021/ac400639y |
| 16. | Gebhardt, J.; Kleist, C.; Jakobtorweihen, S.; Hansen, N. J. Phys. Chem. B 2018, 122, 1608–1626. doi:10.1021/acs.jpcb.7b11808 |
| 2. | Challa, R.; Ahuja, A.; Ali, J.; Khar, R. K. AAPS PharmSciTech 2005, 6, E329–E357. doi:10.1208/pt060243 |
| 3. | Zhang, J.; Ma, P. X. Adv. Drug Delivery Rev. 2013, 65, 1215–1233. doi:10.1016/j.addr.2013.05.001 |
| 4. | Wankar, J.; Kotla, N. G.; Gera, S.; Rasala, S.; Pandit, A.; Rochev, Y. A. Adv. Funct. Mater. 2020, 30, 1909049. doi:10.1002/adfm.201909049 |
| 21. | Hrdina, R.; Holovko-Kamoshenkova, O. M.; Císařová, I.; Koucký, F.; Machalický, O. RSC Adv. 2022, 12, 31056–31060. doi:10.1039/d2ra06402b |
| 11. | Schneider, H.-J.; Hacket, F.; Rüdiger, V.; Ikeda, H. Chem. Rev. 1998, 98, 1755–1786. doi:10.1021/cr970019t |
| 18. | Shundo, A.; Labuta, J.; Hill, J. P.; Ishihara, S.; Ariga, K. J. Am. Chem. Soc. 2009, 131, 9494–9495. doi:10.1021/ja903371d |
| 19. | Labuta, J.; Ishihara, S.; Šikorský, T.; Futera, Z.; Shundo, A.; Hanyková, L.; Burda, J. V.; Ariga, K.; Hill, J. P. Nat. Commun. 2013, 4, 2188. doi:10.1038/ncomms3188 |
| 14. | Lindner, K.; Saenger, W. Acta Crystallogr., Sect. B: Struct. Crystallogr. Cryst. Chem. 1982, 38, 203–210. doi:10.1107/s0567740882002386 |
| 15. | Groom, C. R.; Bruno, I. J.; Lightfoot, M. P.; Ward, S. C. Acta Crystallogr., Sect. B: Struct. Sci., Cryst. Eng. Mater. 2016, 72, 171–179. doi:10.1107/s2052520616003954 |
| 20. | Dodziuk, H.; Koźmiński, W.; Ejchart, A. Chirality 2004, 16, 90–105. doi:10.1002/chir.10304 |
| 13. | Bouchemal, K.; Mazzaferro, S. Drug Discovery Today 2012, 17, 623–629. doi:10.1016/j.drudis.2012.01.023 |
| 11. | Schneider, H.-J.; Hacket, F.; Rüdiger, V.; Ikeda, H. Chem. Rev. 1998, 98, 1755–1786. doi:10.1021/cr970019t |
| 12. | Fielding, L. Tetrahedron 2000, 56, 6151–6170. doi:10.1016/s0040-4020(00)00492-0 |
| 16. | Gebhardt, J.; Kleist, C.; Jakobtorweihen, S.; Hansen, N. J. Phys. Chem. B 2018, 122, 1608–1626. doi:10.1021/acs.jpcb.7b11808 |
| 17. | Schönbeck, C. J. Phys. Chem. B 2018, 122, 4821–4827. doi:10.1021/acs.jpcb.8b02579 |
© 2024 Lebedinskiy et al.; licensee Beilstein-Institut.
This is an open access article licensed under the terms of the Beilstein-Institut Open Access License Agreement (https://www.beilstein-journals.org/bjoc/terms), which is identical to the Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0). The reuse of material under this license requires that the author(s), source and license are credited. Third-party material in this article could be subject to other licenses (typically indicated in the credit line), and in this case, users are required to obtain permission from the license holder to reuse the material.